Label: CALCIUM CHLORIDE- calcium chloride injection
- NDC Code(s): 76329-3304-1
- Packager: International Medication Systems, Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 7, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
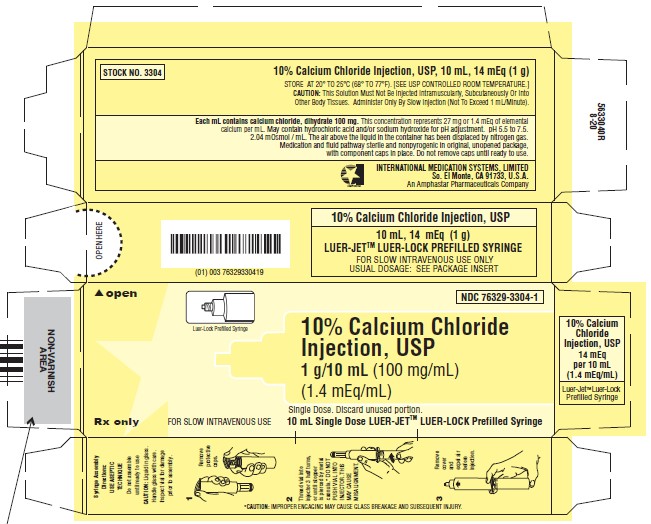
10% Calcium Chloride
Injection, USP
Rx Only
1 g/10 mL (100 mg/mL)
Represents 27 mg (1.4 mEq) Ca++/mL
A HYPERTONIC SOLUTION IN A 10 ML SINGLE-DOSE SYRINGE FOR PROMPT INTRAVENOUS INJECTION.
CAUTION: This solution must not be injected intramuscularly or subcutaneously. Administer only by slow injection (not to exceed 1 mL/minute) -
DESCRIPTION
10% Calcium Chloride Injection, USP is a sterile, nonpyrogenic, hypertonic solution. Each mL contains 100 mg (1.4 mEq/mL) of calcium chloride, dihydrate (1.4 mEq each of Ca++ and Cl-) in water for injection. It is provided in a 10 mL Single-Dose Syringe to facilitate prompt intravenous injection. The solution contains no bacteriostat, antimicrobial agent or added buffer and is intended for use only as a single-dose injection. The pH of 10% Calcium Chloride Injection, USP is 5.5 to 7.5 when diluted with water for injection to make a 5% solution. May contain hydrochloric acid and/or sodium hydroxide for pH adjustment. The osmolar concentration is 2.04 mOsmol/mL (calc.). 10% Calcium Chloride Injection, USP is oxygen sensitive.
Calcium Chloride, USP dihydrate is chemically designated CaCl2 • 2H2O (dihydrate) and is described as white, odorless fragments or granules freely soluble in water.
-
CLINICAL PHARMACOLOGY
Calcium chloride in water dissociates to provide calcium (Ca++) and chloride (Cl-) ions. They are normal constituents of the body fluids and are dependent on various physiological mechanisms for maintenance of balance between intake and output. Approximately 80% of body calcium is excreted in the feces as insoluble salts; urinary excretion accounts for the remaining 20%.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
10% Calcium Chloride Injection, USP is irritating to veins and must not be injected into tissues, since severe necrosis and sloughing may occur. Great care should be taken to avoid extravasation or accidental injection into perivascular tissues.
This product contains aluminum that may be toxic. Aluminum may reach toxic levels with prolonged parenteral administration if kidney function is impaired. Premature neonates are particularly at risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum.
Research indicates that patients with impaired kidney function, including premature neonates, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration.
-
PRECAUTIONS
Do not administer unless solution is clear and seal is intact. Discard unused portion.
Because of its additive effect, calcium should be administered very cautiously to a patient who is digitalized or who is taking effective doses of digitalis or digitalis-like preparations.
Injections should be made slowly through a small needle into a large vein to minimize venous irritation and avoid undesirable reactions. It is particularly important to prevent a high concentration of calcium from reaching the heart because of the danger of cardiac syncope.
Pediatric Use: Safety and effectiveness are based on similar clinical conditions in children and adults.
Pregnancy: Animal reproduction studies have not been conducted with calcium chloride. It also is not known whether calcium chloride can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. Calcium chloride should be given to a pregnant woman only if clearly needed.
Geriatric Use: An evaluation of current literature revealed no clinical experience identifying differences in response between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
-
ADVERSE REACTIONS
Rapid injection may cause the patient to complain of tingling sensations, a calcium taste, a sense of oppression or “heat wave”.
Injections of calcium chloride are accompanied by peripheral vasodilatation as well as a local “burning” sensation and there may be a moderate fall in blood pressure.
Should perivascular infiltration occur, I.V. administration at that site should be discontinued at once. Local infiltration of the affected area with 1% procaine hydrochloride, to which hyaluronidase may be added, will often reduce venospasm and dilute the calcium remaining in the tissues locally. Local application of heat may also be helpful.
- DRUG ABUSE AND DEPENDENCE
-
OVERDOSAGE
Too rapid injection may produce lowering of blood pressure and cardiac syncope. Persistent hypercalcemia from overdosage of calcium is unlikely because of rapid excretion. In the event of untoward effects from excessive calcium administration, the drug should be discontinued promptly, the patient re-evaluated and appropriate countermeasures instituted, if necessary. See PRECAUTIONS and ADVERSE REACTIONS.
-
DOSAGE AND ADMINISTRATION
10% Calcium Chloride Injection, USP is administered only by slow intravenous injection (not to exceed 1 mL/min), preferably in a central or deep vein.
The usual precautions for intravenous therapy should be observed. If time permits, the solution should be warmed to body temperature. The injection should be halted if the patient complains of any discomfort; it may be resumed when symptoms disappear. Following injection, the patient should remain recumbent for a short time.
The usual adult dosage in hypocalcemic disorders ranges from 200 mg to 1 g (2 -10 mL) at intervals of 1 to 3 days depending on the response of the patient and/or results of serum ionized calcium determinations. Repeated injections may be required because of rapid excretion of calcium.
The pediatric dosage in hypocalcemic disorders ranges from 2.7 to 5.0 mg/kg hydrated calcium chloride (or 0.136 to 0.252 mEq elemental calcium per kg, or 0.027 to 0.05 mL of 10% Calcium Chloride Injection per kg). No data from clinical trials is available about repeated dosages, though textbook references recommend repeat dosages q 4 to 6 hours.
Caution: 10% Calcium Chloride Injection consists of 1 gram of calcium chloride in a 10 mL syringe, or 100 mg/mL. This concentration represents 27 mg or 1.4 mEq of elemental calcium per mL. Thus, one 10 mL syringe provides 270 mg of elemental calcium. The dosage recommendation in various references is given either as amount of calcium chloride or amount of elemental calcium, and often it is not specified. Ionized calcium concentrations should be measured, to assist in dosage adjustment.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. See PRECAUTIONS.
To prevent needle-stick injuries, needles should not be recapped, purposely bent or broken by hand.
-
HOW SUPPLIED
10% Calcium Chloride Injection, USP is supplied in single-dose containers as follows:
NDC No. Stock No. Container Size Needle 76329-3304-1 3304 10 mL None One shrink wrapped package containing 10 unit cartons, each containing a Luer-Jet™ Luer-Lock Prefilled Syringe.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F). [See USP Controlled Room Temperature.]Syringe Assembly Directions:
USE ASEPTIC TECHNIQUE
Do not assemble until ready to use.
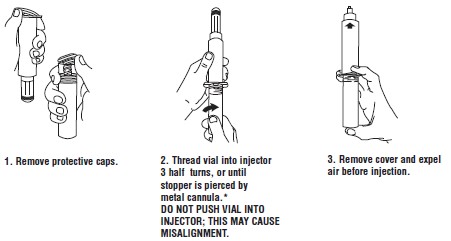
*CAUTION: IMPROPER ENGAGING MAY CAUSE GLASS BREAKAGE AND SUBSEQUENT INJURY.
- SPL UNCLASSIFIED SECTION
- 10% Calcium Chloride Injection, USP - Carton
-
INGREDIENTS AND APPEARANCE
CALCIUM CHLORIDE
calcium chloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:76329-3304 Route of Administration INTRAVENOUS, INTRAVENTRICULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CHLORIDE 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength Hydrochloric Acid (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76329-3304-1 10 in 1 PACKAGE 07/08/2018 1 10 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA203477 07/08/2018 Labeler - International Medication Systems, Limited (055750020) Establishment Name Address ID/FEI Business Operations International Medication Systems, Limited 055750020 analysis(76329-3304) , manufacture(76329-3304)