Label: HYDROCORTISONE (antipruritic- anti-itch cream
-
NDC Code(s):
67777-004-01,
67777-004-02,
67777-004-03,
67777-004-04, view more67777-004-05, 67777-004-06, 67777-004-07, 67777-004-08
- Packager: Dynarex Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 23, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
-
Warnings
For External Use Only
Do not use
- In the eyes
- For diaper rash
- For external genital or feminine itching if you have a vaginal discharge
- More than the recommended daily dosage unless directed by a doctor
- In the rectum by using fingers or any mechanical device or applicator
-
Directions
- Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
- Children under 2 years of age: Consult a doctor.
- When used for anal itching, cleanse the affected area with mild soap and warm water and rinse thoroughly, gently dry by patting or blotting with bathroom tissue or soft cloth before applying.
- Children under 12: Consult a doctor before using for anal itching.
- Other Information
- Inactive Ingredients
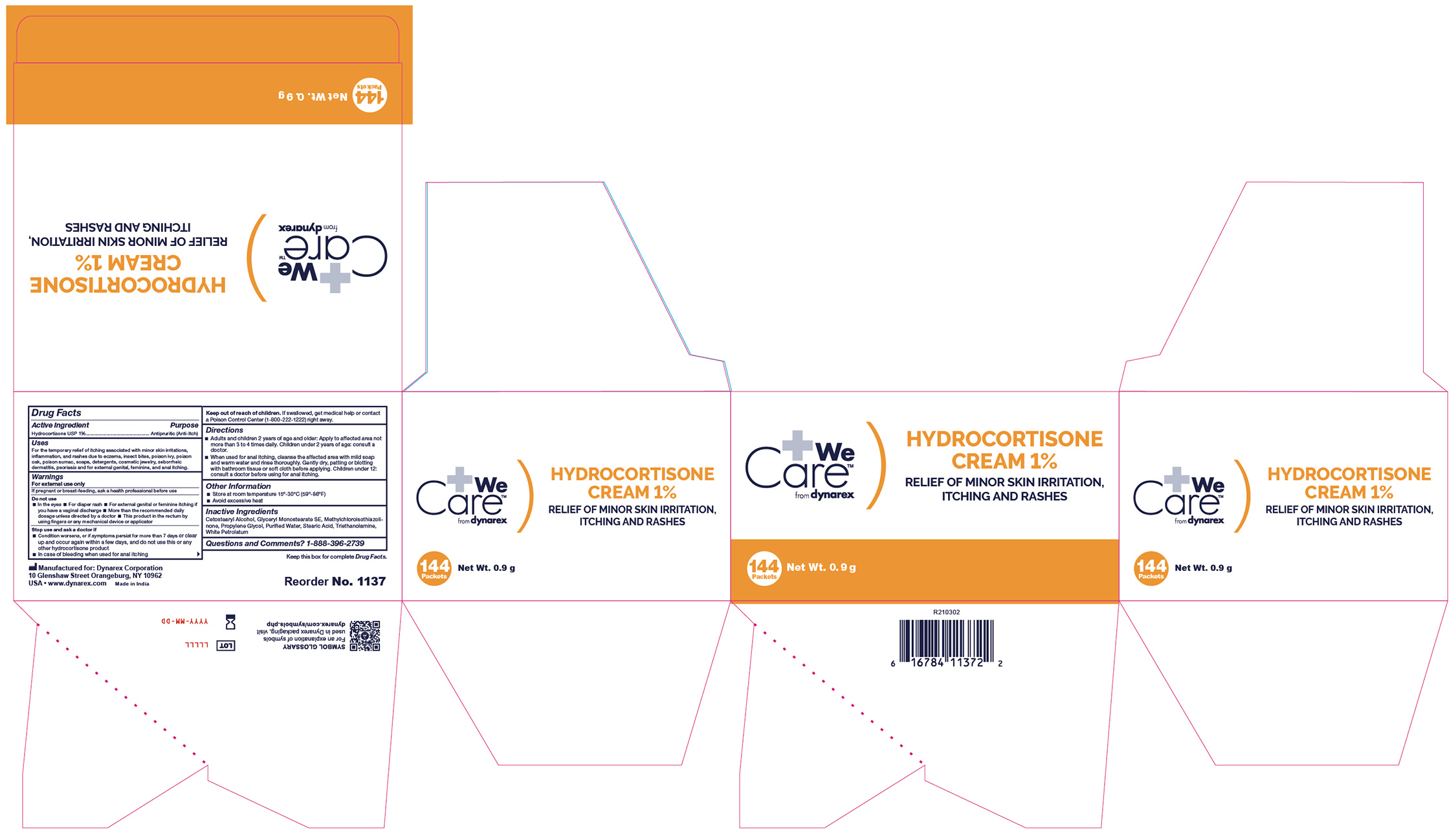
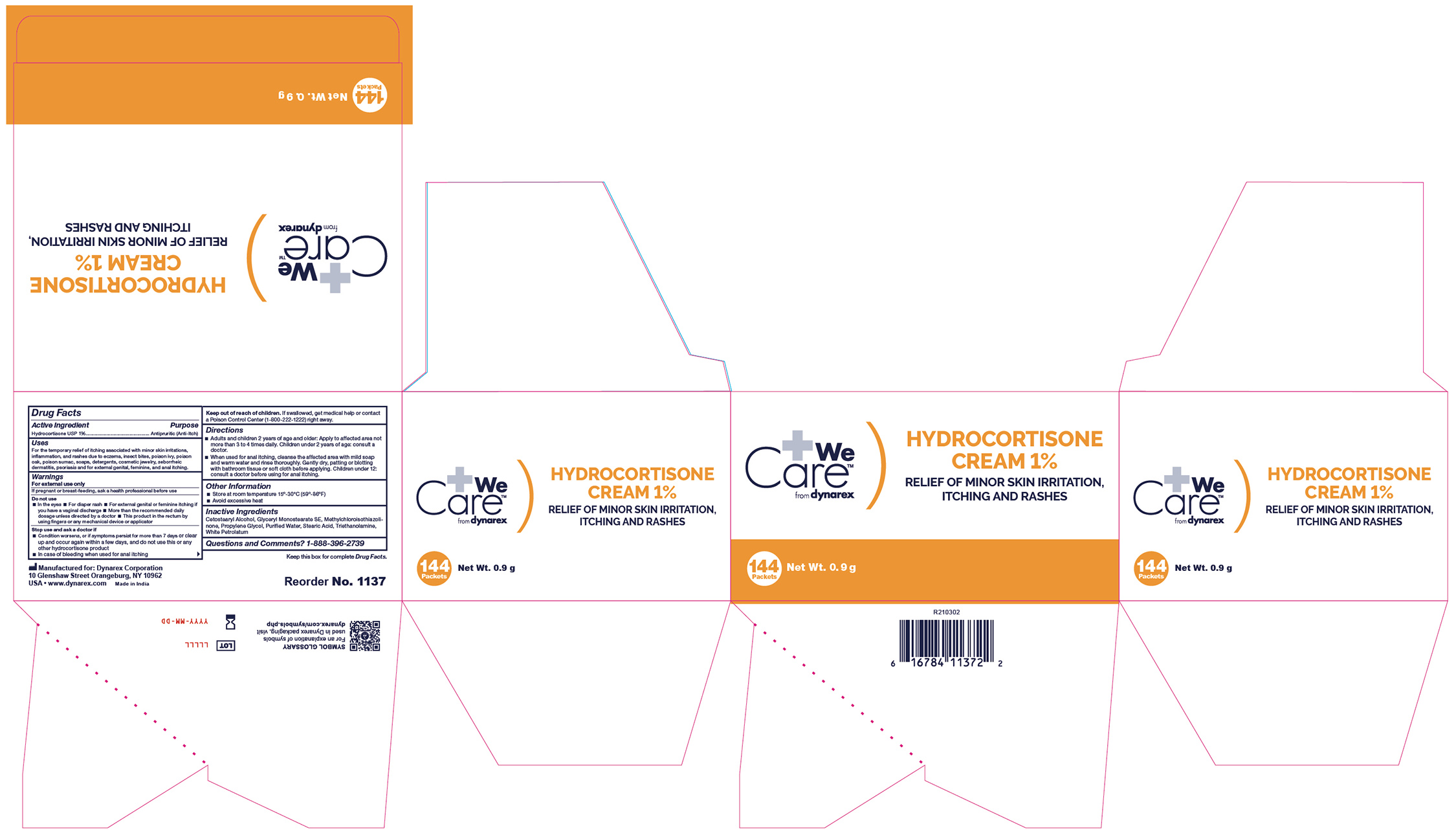
- Label
- Label
- Label 1137UB-6
- Label 1137-25
-
INGREDIENTS AND APPEARANCE
HYDROCORTISONE
antipruritic (anti-itch) creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67777-004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE (UNII: WI4X0X7BPJ) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 1 g in 100 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) WHITE PETROLATUM (UNII: B6E5W8RQJ4) STEARIC ACID (UNII: 4ELV7Z65AP) TROLAMINE (UNII: 9O3K93S3TK) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) WATER (UNII: 059QF0KO0R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) Product Characteristics Color Score Shape FREEFORM Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67777-004-04 12 in 1 CASE 03/25/2021 1 NDC:67777-004-03 144 in 1 BOX 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product 2 NDC:67777-004-01 72 in 1 CASE 03/25/2021 2 NDC:67777-004-02 1 in 1 BOX 2 28.4 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:67777-004-05 600 in 1 CASE 03/25/2021 3 NDC:67777-004-06 6 in 1 BOX 3 0.9 g in 1 PACKET; Type 0: Not a Combination Product 4 NDC:67777-004-07 1800 in 1 CASE 03/25/2021 4 NDC:67777-004-08 25 in 1 BOX 4 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 03/25/2021 Labeler - Dynarex Corporation (008124539)





 1137UB-6
1137UB-6
 1137-25
1137-25