Label: SENSODYNE PRONAMEL INTENSIVE ENAMEL REPAIR EXTRA FRESH- sodium fluoride liquid
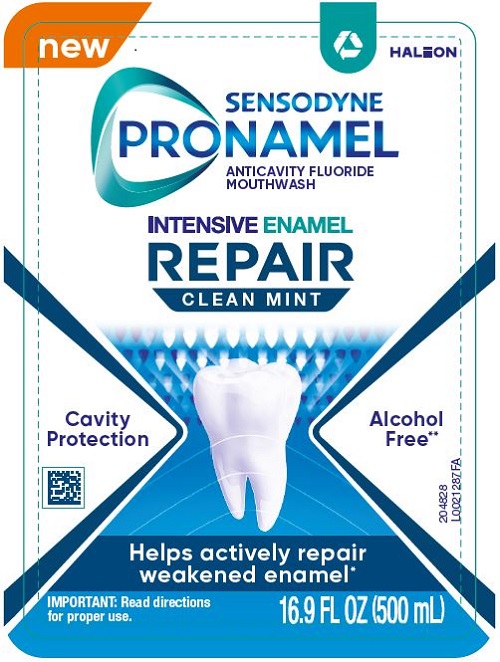
SENSODYNE PRONAMEL INTENSIVE ENAMEL REPAIR CLEAN MINT- sodium fluoride liquid
- NDC Code(s): 0135-0449-01, 0135-0451-01
- Packager: Haleon US Holdings LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warning
-
Directions
• adults and children 6 years of age and older:
- • use once a day after brushing your teeth with a toothpaste.
- • pour 10 milliliters into cap (10 mL mark on inside of cap); do not fill above the 10 mL mark.
- • vigorously swish 10 milliliters of rinse between your teeth for 1 minute and then spit out.
- • do not swallow the rinse.
- • do not drink from the bottle.
- • do not rinse mouth with water.
- • do not eat or drink for 30 minutes after rinsing.
- • instruct children under 12 years of age in good rinsing habits (to minimize swallowing).
- • supervise children as necessary until capable of using without supervision.
• children under 6 years of age:Consult a dentist or doctor.
- Other information
- Inactive ingredients (Clean Mint)
- Inactive ingredients (Extra Fresh)
- Questions or comments?
-
Other Information
Re-mineralizing action for strong and protected teeth*
- Alcohol Free**
- SLS Free
- Paraben Free
- Dye Free
ALWAYS FOLLOW THE LABEL
Do not use if printed safety seal around cap is broken or missing.
*when used as directed with a fluoride toothpaste
**contains no ethanolDistributed by: Haleon, Warren, NJ 07059
Trademarks are owned or licensed to the Haleon group of companies. ©2023 Haleon group of companies or its licensor.Made in Taiwan
- Principal Display Panel (Clean Mint)
- Principal Display Panel (Extra Fresh)
-
INGREDIENTS AND APPEARANCE
SENSODYNE PRONAMEL INTENSIVE ENAMEL REPAIR EXTRA FRESH
sodium fluoride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0135-0451 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) SODIUM FLUORIDE 0.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SODIUM BENZOATE (UNII: OJ245FE5EU) POLOXAMER 407 (UNII: TUF2IVW3M2) BENZYL ALCOHOL (UNII: LKG8494WBH) POLYSORBATE 20 (UNII: 7T1F30V5YH) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SACCHARIN SODIUM (UNII: SB8ZUX40TY) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) SODIUM PHOSPHATE, MONOBASIC, UNSPECIFIED FORM (UNII: 3980JIH2SW) EDETATE CALCIUM DISODIUM (UNII: 25IH6R4SGF) CETYLPYRIDINIUM CHLORIDE (UNII: D9OM4SK49P) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Product Characteristics Color blue Score Shape Size Flavor MINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0135-0451-01 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 01/02/2024 SENSODYNE PRONAMEL INTENSIVE ENAMEL REPAIR CLEAN MINT
sodium fluoride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0135-0449 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) SODIUM FLUORIDE 0.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SODIUM BENZOATE (UNII: OJ245FE5EU) POLOXAMER 407 (UNII: TUF2IVW3M2) BENZYL ALCOHOL (UNII: LKG8494WBH) POLYSORBATE 20 (UNII: 7T1F30V5YH) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SACCHARIN SODIUM (UNII: SB8ZUX40TY) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) SODIUM PHOSPHATE, MONOBASIC, UNSPECIFIED FORM (UNII: 3980JIH2SW) EDETATE CALCIUM DISODIUM (UNII: 25IH6R4SGF) CETYLPYRIDINIUM CHLORIDE (UNII: D9OM4SK49P) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) Product Characteristics Color Score Shape Size Flavor MINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0135-0449-01 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 01/02/2024 Labeler - Haleon US Holdings LLC (079944263)