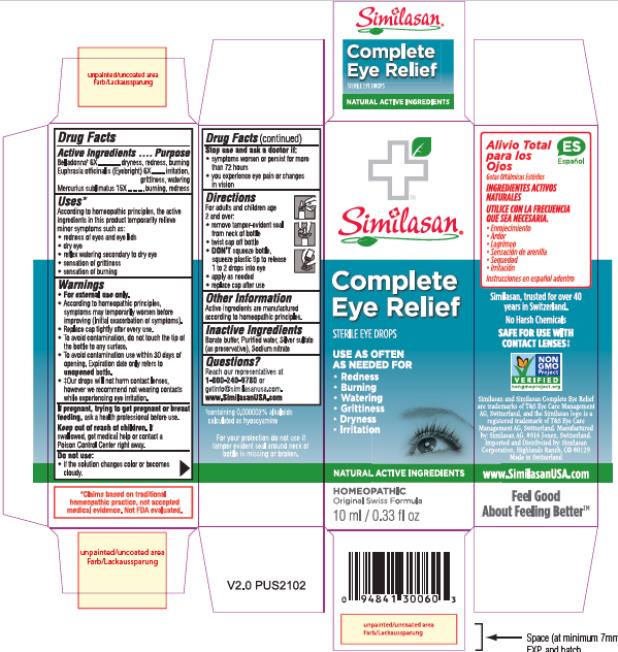
Label: COMPLETE EYE RELIEF- atropa belladonna, euphrasia stricta and mercuric chloride solution/ drops
- NDC Code(s): 53799-373-11
- Packager: Similasan AG
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated May 3, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
- Uses*
-
Warnings
• For external use only.
• According to homeopathic principles, symptoms may temporarily worsen before improving (Initial exacerbation of symptoms).
• Replace cap tightly after every use.
• To avoid contamination, do not touch the tip of the bottle to any surface.
• To avoid contamination use within 30 days of opening. Expiration date only refers to unopened bottle.
• ‡Our drops will not harm contact lenses, however we recommend not wearing contacts while experiencing eye irritation.
- Directions
- Other Information
- Inactive Ingredients
- Questions?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
COMPLETE EYE RELIEF
atropa belladonna, euphrasia stricta and mercuric chloride solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53799-373 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ATROPA BELLADONNA (UNII: WQZ3G9PF0H) (ATROPA BELLADONNA - UNII:WQZ3G9PF0H) ATROPA BELLADONNA 6 [hp_X] in 10 mL EUPHRASIA STRICTA (UNII: C9642I91WL) (EUPHRASIA STRICTA - UNII:C9642I91WL) EUPHRASIA STRICTA 6 [hp_X] in 10 mL MERCURIC CHLORIDE (UNII: 53GH7MZT1R) (MERCURIC CATION - UNII:ED30FJ8Y42) MERCURIC CHLORIDE 15 [hp_X] in 10 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SILVER SULFATE (UNII: 8QG6HV4ZPO) SODIUM NITRATE (UNII: 8M4L3H2ZVZ) SODIUM BORATE (UNII: 91MBZ8H3QO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53799-373-11 1 in 1 CARTON 02/15/2019 1 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 02/15/2019 Labeler - Similasan AG (481545754)