Label: PRISMASOL BGK0/2.5- calcium chloride, magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride and sodium bicarbonate injection
PRISMASOL BGK4/2.5- calcium chloride, magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injection
PRISMASOL BGK2/3.5- calcium chloride, magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injection
PRISMASOL BGK2/0- magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injection
PRISMASOL B22GK4/0- magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injection
PRISMASOL BK0/0/1.2- magnesium chloride, lactic acid, sodium chloride and sodium bicarbonate injection
PRISMASOL BGK4/0/1.2- magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injection
PHOXILLUM BK4/2.5- calcium chloride, magnesium chloride, sodium chloride, sodium bicarbonate, potassium chloride and sodium phosphate dibasic dihydrate injection
PHOXILLUM B22K4/0- magnesium chloride, sodium chloride, sodium bicarbonate, potassium chloride and sodium phosphate dibasic dihydrate injection
-
NDC Code(s):
24571-102-06,
24571-103-06,
24571-105-06,
24571-108-06, view more24571-111-06, 24571-113-06, 24571-114-06, 24571-116-06, 24571-117-06
- Packager: Baxter Healthcare Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated May 2, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use PRISMASOL and PHOXILLUM safely and effectively. See full prescribing information for PRISMASOL and PHOXILLUM.
PRISMASOL renal replacement solution
PRISMASOL Initial U.S. Approval: 2006
PHOXILLUM renal replacement solution
PHOXILLUM Initial U.S. Approval: 2015INDICATIONS AND USAGE
PRISMASOL and PHOXILLUM solutions are indicated:
- •
- As a replacement solution in Continuous Renal Replacement Therapy (CRRT) and in case of drug poisoning when CRRT is used to remove dialyzable substances (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
Known hypersensitivities to PRISMASOL and PHOXILLUM solutions (4)
WARNINGS AND PRECAUTIONS
- •
- Monitor hemodynamic status and fluid inputs and outputs, potassium, phosphorus, other electrolytes and acid-base balance. Abnormalities may be corrected by the use of appropriate formulations and dosage of PRISMASOL and PHOXILLUM solutions (5.1)
- •
- Treatment may affect glucose levels. Monitor blood glucose levels.
- •
- Antidiabetic therapy adjustment or other corrective measures may be required during treatment (5.2)
To report SUSPECTED ADVERSE REACTIONS, contact Baxter Healthcare Corporation at 1-866-888-2472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Revised: 5/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Administration Instructions
2.2 Dosing Considerations
2.3 Preparing the Solution
2.4 Adding Drugs to the Solutions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Electrolyte and Volume Abnormalities
5.2 Blood Glucose Abnormalities
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Citrate
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
16 HOW SUPPLIED/STORAGE AND HANDLING
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
PRISMASOL and PHOXILLUM solutions are indicated in pediatric and adult patients for use as a replacement solution in Continuous Renal Replacement Therapy (CRRT) to replace plasma volume removed by ultrafiltration and to correct electrolyte and acid-base imbalances. They may also be used in case of drug poisoning when CRRT is used to remove dialyzable substances.
-
2 DOSAGE AND ADMINISTRATION
2.1 Administration Instructions
Visually inspect PRISMASOL and PHOXILLUM for particulate matter and discoloration prior to administration.
Administration should only be under the direction of a physician competent in intensive care treatment including CRRT. Use only with extracorporeal dialysis equipment appropriate for CRRT.
The prepared solution is for single patient use only.
Aseptic technique should be used throughout administration to the patient.
Discard any unused solution.
2.2 Dosing Considerations
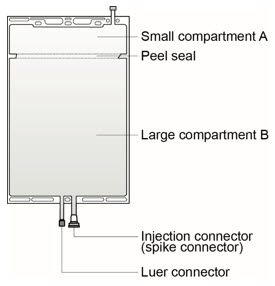
PRISMASOL replacement solutions contain 4 different combinations of active ingredients (7 different products with varying ingredient amounts). PHOXILLUM replacement solutions contain 2 different combinations of active ingredients (2 different products with varying ingredient amounts). PRISMASOL and PHOXILLUM are supplied in a two-compartment bag that must be mixed immediately prior to use [see Dosage and Administration (2.3)]:
- •
- Small compartment A (250 mL) containing an electrolyte solution, and
- •
- Large compartment B (4750 mL) containing the buffer solution.
See Table 1 for the concentrations of the active ingredients (after mixing) in these 9 different replacement solutions (total volume is 5 Liters).
Table 1: Concentrations of Active Ingredients in the 7 PRISMASOL and 2 PHOXILLUM Replacement Solutions after Mixing Ca2+
mEq/LHCO3-
mEq/LK+
mEq/LMg2+
mEq/LNa+
mEq/LHPO42-
mmol/LCl-
mEq/LLactate
mEq/LDextrose
mg/dLOsmolarity
mOsm/LCa2+ = calcium, HCO3- = bicarbonate, K+ = potassium, Mg2+ = magnesium, Na+ = sodium, HPO42- = phosphate, Cl- = chloride; osmolarity is estimated PRISMASOL Replacement Solutions
BGK0/2.5
2.5
32
0
1.5
140
0
109
3
100
292
BGK4/2.5
2.5
32
4
1.5
140
0
113
3
100
300
BGK2/3.5
3.5
32
2
1
140
0
111.5
3
100
296
BGK2/0
0
32
2
1
140
0
108
3
100
291
B22GK4/0
0
22
4
1.5
140
0
120.5
3
100
296
BGK4/0/1.2
0
32
4
1.2
140
0
110.2
3
100
295
BK0/0/1.2
0
32
0
1.2
140
0
106.2
3
0
282
PHOXILLUM Replacement Solutions
BK4/2.5
2.5
32
4
1.5
140
1
114.5
0
0
294
B22K4/0
0
22
4
1.5
140
1
122
0
0
290
The mode of therapy, solute formulation, flow rates, and length of PRISMASOL and PHOXILLUM replacement therapy in CRRT should be established by a physician based on the patient’s clinical condition, blood concentration of phosphate and other electrolytes, acid-base and glucose balance. Administer either PRISMASOL or PHOXILLUM into the extracorporeal circuit:
- •
- Before (pre-dilution) the hemofilter or hemodiafilter,
- •
- After (post-dilution) the hemofilter or hemodiafilter, or
- •
- Before and after the hemofilter or hemodiafilter.
2.3 Preparing the Solution
Use only if the overwrap is not damaged, all seals are intact, peel seal is not broken, and the solution is clear.
The solution may be warmed to 37°C/98.6°F prior to removing the overwrap to enhance patient comfort. However, only dry heat should be used. Solutions should not be heated in water or in a microwave oven. After heating, verify that the solution remains clear and contains no particulate matter.
The solutions are supplied in two different two-compartment bags made of polyolefin with a peel seal separating compartment A and B (see Figure 1).
Follow the instructions below when connecting the solution bags for correct use of the access ports.
Instructions for preparing solutions supplied in a two-compartment, polyolefin bag with a peel seal:
Step 1 Immediately before use, remove the overwrap from the bag and mix the solutions in the two different compartments. After removing the overwrap, inspect the bag for leakage by pressing firmly on the bag. Discard the bag if any leakage is detected since sterility cannot be assured. As soon as the overwrap is removed, the reconstitution of compartments A and B should be done and the mixed solution should be used immediately.
After removal of the overwrap, the solution is stable for 24 hours including the duration of the treatment.
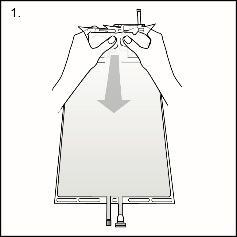
Hold the small compartment with both hands and squeeze it until an opening is created in the peel seal. (See Figure 2 beside)
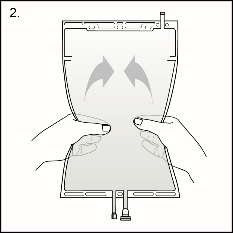
Step 2 Squeeze with both hands on the large compartment until the peel seal between the two compartments is entirely open. Shake gently to mix. (See Figure 3 beside)
The solution is now ready to use and the bag can be hung on the equipment.
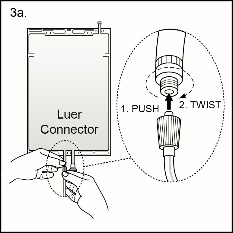
Step 3 The replacement line may be connected to the bag through either of the luer connector or the injection connector (spike connector).
Step 3a The luer connector is a needle-less and swabbable connector. Remove the cap with a twist and pull motion, and connect the male luer lock on the replacement line to the female luer receptor on the bag. (See Figure 4a beside)
Ensure that the connection is fully seated and tighten. The connector is now open. Verify that the fluid is flowing freely during use.
When the replacement line is disconnected from the luer connector, the connector will close and the flow of the solution will stop.
Figure 4b
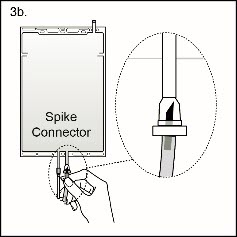
Step 3b If the injection connector (spike connector) is used, first remove the snap-off cap. Then introduce the replacement line spike through the swabbable rubber septum of the bag connector. (See Figure 4b beside)
Ensure that the spike is fully inserted and verify that the fluid is flowing freely during use.
2.4 Adding Drugs to the Solutions
After mixing, additional drugs may be added to the bag via injection connector (spike connector) in large compartment B. In general, administer drugs other than phosphate through a different access line.
When introducing drugs, use aseptic techniques and mix thoroughly prior to connecting the solution bag to the extracorporeal circuit.
Do not use if there is a color change and/or the appearance of precipitates, insoluble complexes or crystals after addition of medication.
Phosphate: Up to 1.2 mmol/L of phosphate can be added to the bag as potassium phosphate or sodium phosphate. The total potassium concentration of PRISMASOL solution should not exceed 4 mEq/L. Use sodium phosphate to add phosphate if the total potassium concentration in PRISMASOL solution is 4 mEq/L.
-
3 DOSAGE FORMS AND STRENGTHS
See Table 1 for the concentrations of the active ingredients (after mixing) in these 9 different replacement solutions [see Dosage and Administration (2.2)].
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Electrolyte and Volume Abnormalities
PHOXILLUM and PRISMASOL solutions can affect electrolytes and volume and may result in hyperkalemia or hyperphosphatemia. Monitor hemodynamic status and fluid inputs and outputs, potassium, phosphorous, calcium, other electrolytes and acid-base balance throughout the procedure. Abnormalities may be corrected by changing the formulation of replacement solution and/or dialysate, supplementation, or adjusting flow rates appropriately [see Dosage and Administration (2)].
PHOXILLUM replacement solutions contain hydrogen phosphate, a weak acid that may increase the risk of metabolic acidosis.
5.2 Blood Glucose Abnormalities
The use of PRISMASOL and PHOXILLUM replacement solutions can affect blood glucose levels resulting in hypo- or hyper-glycemia depending upon the dextrose content of the replacement solution. Monitor blood glucose levels regularly. Patients may require initiation of or modification of antidiabetic therapy or other corrective measures during treatment.
-
6 ADVERSE REACTIONS
The following adverse reactions have been identified during postapproval use with these or other similar products and therefore may occur with use of PHOXILLUM or PRISMASOL. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- •
- Metabolic acidosis
- •
- Hypotension
- •
- Acid-base disorders
- •
- Electrolyte imbalance including calcium ionized increased (reported in PRISMASOL solutions containing calcium), hyperphosphatemia, and hypophosphatemia
- •
- Fluid imbalance
-
7 DRUG INTERACTIONS
As with the use of other replacement solutions, blood concentrations of dialyzable drugs may be reduced by CRRT due to their removal by the hemofilter or hemodiafilter. The blood concentrations of certain drugs may need to be monitored and appropriate therapy implemented to correct for removal during treatment.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
PRISMASOL and PHOXILLUM are pharmacologically inactive solutions. While there are no adequate and well controlled studies in pregnant women, appropriate administration of PRISMASOL and PHOXILLUM solutions with monitoring of fluid, electrolyte, acid-base and glucose balance, is not expected to cause fetal harm. Animal reproduction studies have not been conducted with PRISMASOL and PHOXILLUM solutions.
The estimated background risk of major birth defects and miscarriage for the indicated population are unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
-
11 DESCRIPTION
PRISMASOL and PHOXILLUM solutions are clear, sterile, free of bacterial endotoxins and contain no bacteriostatic or antimicrobial agents. These solutions are used in Continuous Renal Replacement Therapies (CRRT) as a replacement solution in hemofiltration and hemodiafiltration. Depending on the product (see Table 2), the two compartments contain:
Calcium chloride, USP, is chemically designated calcium chloride dihydrate (CaCl2 • 2H2O).
Magnesium chloride, USP, is chemically designated magnesium chloride hexahydrate (MgCl2 • 6H2O).
Sodium chloride, USP, is chemically designated NaCl.
Potassium chloride, USP, is chemically designated KCl.
Sodium bicarbonate, USP, is chemically designated NaHCO3.
Dextrose, USP, is chemically designated D-Glucose anhydrous (C6H12O6) or D-Glucose monohydrate (C6H12O6 • H2O).
Lactic acid, USP, is chemically designated CH3CH(OH)COOH.
Dibasic sodium phosphate, USP, is chemically designated as disodium hydrogen phosphate, dihydrate (Na2HPO4 • 2H2O)
Table 2 - Compartment Composition (Before Mixing) Compartment A (g/L) Compartment B (g/L) Calcium Chloride ∙ 2H2O Magnesium Chloride ∙ 6H2O Dextrose anhydrous (as monohydrate) Lactic Acid Sodium Chloride Sodium bicarbonate Potassium Chloride Sodium Phosphate ∙ 2H2O PRISMASOL SOLUTIONS
BGK 0/2.5
3.68
3.05
20 (22)
5.40
6.46
3.09
0
0
BGK 4/2.5
3.68
3.05
20 (22)
5.40
6.46
3.09
0.314
0
BGK 2/3.5
5.15
2.03
20 (22)
5.40
6.46
3.09
0.157
0
BGK 2/0
0
2.03
20 (22)
5.40
6.46
3.09
0.157
0
B22GK 4/0
0
3.05
20 (22)
5.40
7.07
2.21
0.314
0
BK 0/0/1.2
0
2.44
0 (0)
5.40
6.46
3.09
0
0
BGK 4/0/1.2
0
2.44
20 (22)
5.40
6.46
3.09
0.314
0
PHOXILLUM SOLUTIONS
BK 4/2.5
3.68
3.05
0 (0)
0
6.34
3.09
0.314
0.187
B22K 4/0
0
3.05
0 (0)
0
6.95
2.21
0.314
0.187
The pH of the final solution is in the range of 7.0 to 8.5.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
PRISMASOL and PHOXILLUM solutions are pharmacologically inactive. The electrolyte concentrations in the solutions are chosen to restore plasma levels to clinically desired concentrations or maintain plasma levels at the desired concentrations.
PRISMASOL and PHOXILLUM solutions are used as replacement solution to replace water and electrolytes removed during hemofiltration and hemodiafiltration. Bicarbonate (or precursor lactate) in the solution is used as an alkalinizing buffer to restore acid-base balance to a clinically desirable level.
12.3 Pharmacokinetics
The distribution of electrolytes, bicarbonate, and dextrose is determined by the patient's clinical condition, metabolic status, and residual renal function.
The elimination and replacement of water, electrolytes and buffer depend on the patient's electrolyte and acid-base balance, metabolic status, residual renal function and ongoing physiologic losses through intestinal, respiratory and cutaneous routes.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
PRISMASOL and PHOXILLUM solutions are supplied in a two-compartment bag made of polyolefin. The 5000 mL bag is composed of a small compartment (250 mL) and a large compartment (4750 mL). The two compartments are separated by a peel seal.
The bag is overwrapped with a transparent overwrap. See Table 2 for the concentrations of the active ingredients in each compartment for each product [see Description (11)].
Container
Fill Volume
NDC
PRISMASOL Solutions
PRISMASOL BGK0/2.5
5000 mL
24571-108-06
PRISMASOL BGK4/2.5
5000 mL
24571-105-06
PRISMASOL BGK2/3.5
5000 mL
24571-103-06
PRISMASOL BGK2/0
5000 mL
24571-102-06
PRISMASOL B22GK4/0
5000 mL
24571-111-06
PRISMASOL BK0/0/1.2
5000 mL
24571-113-06
PRISMASOL BGK4/0/1.2
5000 mL
24571-114-06
PHOXILLUM Solutions
PHOXILLUM BK4/2.5
5000 mL
24571-116-06
PHOXILLUM B22K4/0
5000 mL
24571-117-06
Not all formulations may be marketed.
Storage conditions
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF). [See USP Controlled Room Temperature]
Do not freeze or expose to excessive heat. Do not use if precipitate has formed or if container seals have been damaged.
Manufactured for:
Baxter Healthcare Corporation
One Baxter Parkway
Deerfield, Illinois 6001507-19-00-6103
Baxter, Gambro, Phoxillum and PrismaSol are trademarks of Baxter International Inc., or its subsidiaries
-
Package/Label Display Panel
Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Barcode
NDC# 24571-108-06OK+
mEq/L2.5 Ca2+
mEq/LPrismaSolBGK0/2.5
Rx only
Barcode
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution each 1000 mL contains (g)
A
B
Calcium chloride • 2H2O
3.68
Magnesium chloride • 6H2O
3.05
Dextrose anhydrous
20.0
(as dextrose monohydrate)
22.0
Sodium chloride
6.46
Lactic acid
5.40
Sodium bicarbonate
3.09
Water for injections q.s, Carbon dioxide for pH adjustment
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Lactate
C3H5O3-Bicarbonate
HCO3-Potassium
K+Dextrose
mmol/L
1.25
0.75
140
109.0
3.0
32
0
5.5
mEq/L
2.5
1.5
140
109.0
3.0
32
0
(100 mg/dL)
- Theoretical osmolarity: 292 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information andfurther instructions.
Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if
solution is clear. For single use only.
DISCARD ANY UNUSED SOLUTION.
Store at +20°C to +25°C (+68°F to +77°F); excursions
permitted to +15°C to +30°C (+59°F to
+86°F). [See USP Controlled room Temperature.]
Do not freeze or expose to excessive heat.
As soon as the overwrap is removed, the reconstitution
of compartments A and B should be done
and the reconstituted solution should be used immediately.
After removal of the overwrap, the solution
is stable for 24 hours including the duration of
the treatment. Mix additives BEFORE connecting
this bag to the extracorporeal circuit. (See insert
for further information.) This product is not made
with natural rubber latex.
5000 mL
EAN-14: 07332414091613
Product # 110240Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in ItalyGAMBRO Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy07-25-00-0110
Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Barcode
NDC# 24571-105-064K+
mEq/L2.5 Ca2+
mEq/LPrismaSol BGK4/2.5
Rx only
Barcode
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution each 1000 mL contains (g)
A
B
Calcium chloride • 2H2O
3.68
Magnesium chloride • 6H2O
3.05
Dextrose anhydrous
20.0
(as dextrose monohydrate)
22.0
Sodium chloride
6.46
Potassium chloride
0.314
Lactic acid
5.40
Sodium bicarbonate
3.09
Water for injections q.s, Carbon dioxide for pH adjustment
A
250 mLB
4750 mLAfter reconstitution, A + B
After reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Lactate
C3H5O3-Bicarbonate
HCO3-Potassium
K+Dextrose
mmol/L
1.25
0.75
140
113.0
3.0
32
4.0
5.5
mEq/L
2.5
1.5
140
113.0
3.0
32
4.0
(100 mg/dL)
- Theoretical osmolarity: 300 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information andfurther instructions.
Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if
solution is clear. For single use only.
DISCARD ANY UNUSED SOLUTION.
Store at +20°C to +25°C (+68°F to +77°F); excursions
permitted to +15°C to +30°C (+59°F to
+86°F). [See USP Controlled room Temperature.]
Do not freeze or expose to excessive heat.
As soon as the overwrap is removed, the reconstitution
of compartments A and B should be done
and the reconstituted solution should be used immediately.
After removal of the overwrap, the solution
is stable for 24 hours including the duration of
the treatment. Mix additives BEFORE connecting
this bag to the extracorporeal circuit. (See insert
for further information.) This product is not made
with natural rubber latex.
5000 mL
EAN-14: 07332414091637
Product # 110242Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in MexicoBaxter Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy
07-25-00-3317Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Barcode
NDC# 24571-103-062K+
mEq/L3.5 Ca2+
mEq/LPrismaSolBGK2/3.5
Rx only
Barcode
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution each 1000 mL contains (g)
A
B
Calcium chloride • 2H2O
5.15
Magnesium chloride • 6H2O
2.03
Dextrose anhydrous
20.0
(as dextrose monohydrate)
22.0
Sodium chloride
6.46
Potassium chloride
0.157
Lactic acid
5.40
Sodium bicarbonate
3.09
Water for injections q.s, Carbon dioxide for pH adjustment
Rx only
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Lactate
C3H5O3-Bicarbonate
HCO3-Potassium
K+Dextrose
mmol/L
1.75
0.5
140
111.5
3.0
32
2.0
5.5
mEq/L
3.5
1.0
140
111.5
3.0
32
2.0
(100 mg/dL)
- Theoretical osmolarity: 296 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information andfurther instructions.
Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if
solution is clear. For single use only.
DISCARD ANY UNUSED SOLUTION.
Store at +20°C to +25°C (+68°F to +77°F); excursions
permitted to +15°C to +30°C (+59°F to
+86°F). [See USP Controlled room Temperature.]
Do not freeze or expose to excessive heat.
As soon as the overwrap is removed, the reconstitution
of compartments A and B should be done
and the reconstituted solution should be used immediately.
After removal of the overwrap, the solution
is stable for 24 hours including the duration of
the treatment. Mix additives BEFORE connecting
this bag to the extracorporeal circuit. (See insert
for further information.) This product is not made
with natural rubber latex.
5000 mL
EAN-14: 07332414091644
Product # 110243Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in MexicoBaxter Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy
07-25-00-4423Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Barcode
NDC# 24571-102-062K+
mEq/L0Ca2+
mEq/LPrismaSol BGK2/0
Rx only
Barcode
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution each 1000 mL contains (g)
A
B
Magnesium chloride • 6H2O
2.03
Dextrose anhydrous
20.0
(as dextrose monohydrate)
22.0
Sodium chloride
6.46
Potassium chloride
0.157
Lactic acid
5.40
Sodium bicarbonate
3.09
Water for injections q.s, Carbon dioxide for pH adjustment
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Lactate
C3H5O3-Bicarbonate
HCO3-Potassium
K+Dextrose
mmol/L
0
0.5
140
108.0
3.0
32
2.0
5.5
mEq/L
0
1.0
140
108.0
3.0
32
2.0
(100 mg/dL)
- Theoretical osmolarity: 291 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information andfurther instructions.
Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if
solution is clear. For single use only.
DISCARD ANY UNUSED SOLUTION.
Store at +20°C to +25°C (+68°F to +77°F); excursions
permitted to +15°C to +30°C (+59°F to
+86°F). [See USP Controlled room Temperature.]
Do not freeze or expose to excessive heat.
As soon as the overwrap is removed, the reconstitution
of compartments A and B should be done
and the reconstituted solution should be used immediately.
After removal of the overwrap, the solution
is stable for 24 hours including the duration of
the treatment. Mix additives BEFORE connecting
this bag to the extracorporeal circuit. (See insert
for further information.) This product is not made
with natural rubber latex.
5000 mL
EAN-14: 07332414091651
Product # 110244Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in ItalyGAMBRO Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy07-25-00-0114
Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Barcode
NDC# 24571-111-064K+
mEq/LBicarbonate 22
0Ca2+
mEq/LPrismaSol B22GK4/0
Rx only
Barcode
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution each 1000 mL contains (g)
A
B
Magnesium chloride • 6H2O
3.05
Dextrose anhydrous
20.0
(as dextrose monohydrate)
22.0
Sodium chloride
7.07
Potassium chloride
0.314
Lactic acid
5.40
Sodium bicarbonate
2.21
Water for injections q.s, Carbon dioxide for pH adjustment
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Lactate
C3H5O3-Bicarbonate
HCO3-Potassium
K+Dextrose
mmol/L
0
0.75
140
120.5
3.0
22
4.0
5.5
mEq/L
0
1.5
140
120.5
3.0
22
4.0
(100 mg/dL)
- Theoretical osmolarity: 296 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information andfurther instructions.
Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if
solution is clear. For single use only.
DISCARD ANY UNUSED SOLUTION.
Store at +20°C to +25°C (+68°F to +77°F); excursions
permitted to +15°C to +30°C (+59°F to
+86°F). [See USP Controlled room Temperature.]
Do not freeze or expose to excessive heat.
As soon as the overwrap is removed, the reconstitution
of compartments A and B should be done
and the reconstituted solution should be used immediately.
After removal of the overwrap, the solution
is stable for 24 hours including the duration of
the treatment. Mix additives BEFORE connecting
this bag to the extracorporeal circuit. (See insert
for further information.) This product is not made
with natural rubber latex.
5000 mL
EAN-14: 07332414116781
Product # 115001Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in ItalyGAMBRO Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy07-25-00-0115
Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Barcode
NDC# 24571-113-060K+
mEq/L0Ca2+
mEq/LPrismaSol BK0/0/1.2
Rx only
Barcode
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution each 1000 mL contains (g)
A
B
Magnesium chloride • 6H2O
2.44
Sodium chloride
6.46
Lactic acid
5.40
Sodium bicarbonate
3.09
Water for injections q.s, Carbon dioxide for pH adjustment
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Lactate
C3H5O3-Bicarbonate
HCO3-Potassium
K+Dextrose
mmol/L
0
0.6
140
106.2
3.0
32
0
0
mEq/L
0
1.2
140
106.2
3.0
32
0
(0 mg/dL)
- Theoretical osmolarity: 282 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information andfurther instructions.
Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if
solution is clear. For single use only.
DISCARD ANY UNUSED SOLUTION.
Store at +20°C to +25°C (+68°F to +77°F); excursions
permitted to +15°C to +30°C (+59°F to
+86°F). [See USP Controlled room Temperature.]
Do not freeze or expose to excessive heat.
As soon as the overwrap is removed, the reconstitution
of compartments A and B should be done
and the reconstituted solution should be used immediately.
After removal of the overwrap, the solution
is stable for 24 hours including the duration of
the treatment. Mix additives BEFORE connecting
this bag to the extracorporeal circuit. (See insert
for further information.) This product is not made
with natural rubber latex.
5000 mL
EAN-14: 07332414091309
Product # 110239Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in ItalyGAMBRO Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy07-25-00-0109
Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Barcode
NDC# 24571-114-064K+
mEq/L0Ca2+
mEq/LPrismaSol BGK4/0/1.2
Rx only
Barcode
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution each 1000 mL contains (g)
A
B
Magnesium chloride • 6H2O
2.44
Dextrose anhydrous
20.0
(as dextrose monohydrate)
22.0
Sodium chloride
6.46
Potassium chloride
0.314
Lactic acid
5.40
Sodium bicarbonate
3.09
Water for injections q.s, Carbon dioxide for pH adjustment
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Lactate
C3H5O3-Bicarbonate
HCO3-Potassium
K+Dextrose
mmol/L
0
0.6
140
110.2
3.0
32
4.0
5.5
mEq/L
0
1.2
140
110.2
3.0
32
4.0
(100 mg/dL)
- Theoretical osmolarity: 295 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information andfurther instructions.
Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if
solution is clear. For single use only.
DISCARD ANY UNUSED SOLUTION.
Store at +20°C to +25°C (+68°F to +77°F); excursions
permitted to +15°C to +30°C (+59°F to
+86°F). [See USP Controlled room Temperature.]
Do not freeze or expose to excessive heat.
As soon as the overwrap is removed, the reconstitution
of compartments A and B should be done
and the reconstituted solution should be used immediately.
After removal of the overwrap, the solution
is stable for 24 hours including the duration of
the treatment. Mix additives BEFORE connecting
this bag to the extracorporeal circuit. (See insert
for further information.) This product is not made
with natural rubber latex.
5000 mL
EAN-14: 07332414091620
Product # 110241Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in ItalyGAMBRO Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy07-25-00-0111
Mixing steps
① SQUEEZE TOP corners to break seal② Squeeze BAG SIDES to fully open seal
Compartment B
Barcode
NDC# 24571-116-064K+
mEq/L1 Phosphate
mmol/L2.5 Ca2+
mEq/LPhoxillum BK4/2.5
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution, each 1000 mL contains (g):
A
B
Calcium chloride • 2H2O
3.68
Magnesium chloride • 6H2O
3.05
Sodium chloride
6.34
Potassium chloride
0.314
Sodium bicarbonate
3.09
Dibasic sodium phosphate • 2H2O
0.187
Water for injections q.s
Rx only
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Bicarbonate
HCO3-Potassium
K+Phosphate
HPO42-Dextrose
mmol/L
1.25
0.75
140
114.5
32
4.0
1
0
mEq/L
2.5
1.5
140
114.5
32
4.0
(1 mmol/L)
(0 mg/dL)
- Theoretical osmolarity: 294 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information and further instructions. Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if solution is clear. For single use only. DISCARD ANY UN-
USED SOLUTION. Store at +20ºC to +25ºC (+68ºF to +77ºF); excursions permitted to +15ºC - +30ºC (+59ºF
to +86ºF). [See USP Controlled Room Temperature]. Do not freeze or expose to excessive heat. As soon as the
overwrap is removed, the reconstitution of compartments A and B should be done and the reconstituted
solution should be used immediately. After removal of the overwrap, the solution is stable for 24 hours including the duration of the treatment. Mix additives BEFORE connecting this bag to the extracorporeal circuit. (See insert for
further information.) This product is not made with natural rubber latex. Carbon dioxide and diluted hydrochloricacid added for pH adjustment.
5000 mL
EAN-14: 07332414116040
Product # 114905Batch No. and expiry date are printed on the
back of the bag.Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in ItalyGAMBRO Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy07-25-00-0107
Mixing steps
① SQUEEZE TOP corners to break seal
② Squeeze BAG SIDES to fully open seal
Compartment B
Barcode
NDC# 24571-117-064K+
mEq/L0 Ca2+
mEq/L22 Bicarbonate
mEq/L1 Phosphate
mmol/LPhoxillum B22K4/0
Replacement Solution for Continuous Renal Replacement TherapyBefore reconstitution, each 1000 mL contains (g):
A
B
Magnesium chloride • 6H2O
3.05
Sodium chloride
6.95
Potassium chloride
0.314
Sodium bicarbonate
2.21
Dibasic sodium phosphate • 2H2O
0.187
Water for injections q.s
Rx only
A
250 mLB
4750 mLAfter reconstitution, A + B
Calcium
Ca2+Magnesium
Mg2+Sodium
Na+Chloride
Cl-Bicarbonate
HCO3-Potassium
K+Phosphate
HPO42-Dextrose
mmol/L
0
0.75
140
122.0
22
4.0
1
0
mEq/L
0
1.5
140
122.0
22
4.0
(1 mmol/L)
(0 mg/dL)
- Theoretical osmolarity: 290 mOsm/L pH: 7.0 – 8.5
Mix both compartments before use.
See package insert for dosage information and further instructions. Sterile and free from bacterial endotoxins.
Confirm the integrity of the packaging. Use only if solution is clear. For single use only. DISCARD ANY UN-
USED SOLUTION. Store at +20°C to +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F
to +86°F). [See USP Controlled Room Temperature]. Do not freeze or expose to excessive heat. As soon as the
overwrap is removed, the reconstitution of compartments A and B should be done and the reconstituted solution
should be used immediately. After removal of the overwrap, the solution is stable for 24 hours including the duration
of the treatment. Mix additives BEFORE connecting this bag to the extracorporeal circuit. (See insert for
further information.) This product is not made with natural rubber latex. Carbon dioxide and diluted hydrochloric
acid added for pH adjustment.5000 mL
EAN-14: 07332414116057
Product # 114906Batch No. and expiry date are printed on the
back of the bag.Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in ItalyGAMBRO Logo
REPLACEMENT
Solution for Continuous Renal Replacement Therapy07-25-00-0108
-
INGREDIENTS AND APPEARANCE
PRISMASOL BGK0/2.5
calcium chloride, magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride and sodium bicarbonate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-108 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB, CHLORIDE ION - UNII:Q32ZN48698) CALCIUM CHLORIDE 3.68 g in 1 L MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 3.05 g in 1 L DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 22 g in 1 L LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) (LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) LACTIC ACID, UNSPECIFIED FORM 5.4 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.46 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 3.09 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-108-06 2 in 1 CASE 10/25/2006 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021703 10/25/2006 PRISMASOL BGK4/2.5
calcium chloride, magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-105 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB, CHLORIDE ION - UNII:Q32ZN48698) CALCIUM CHLORIDE 3.68 g in 1 L MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 3.05 g in 1 L DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 22 g in 1 L LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) (LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) LACTIC ACID, UNSPECIFIED FORM 5.4 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.46 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 3.09 g in 1 L POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 0.314 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-105-06 2 in 1 CASE 10/25/2006 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021703 10/25/2006 PRISMASOL BGK2/3.5
calcium chloride, magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-103 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB, CHLORIDE ION - UNII:Q32ZN48698) CALCIUM CHLORIDE 5.15 g in 1 L MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 2.03 g in 1 L DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 22 g in 1 L LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) (LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) LACTIC ACID, UNSPECIFIED FORM 5.4 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.46 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 3.09 g in 1 L POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 0.157 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-103-06 2 in 1 CASE 10/25/2006 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021703 10/25/2006 PRISMASOL BGK2/0
magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-102 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 2.03 g in 1 L DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 22 g in 1 L LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) (LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) LACTIC ACID, UNSPECIFIED FORM 5.4 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.46 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 3.09 g in 1 L POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 0.157 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-102-06 2 in 1 CASE 10/25/2006 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021703 10/25/2006 PRISMASOL B22GK4/0
magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-111 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 3.05 g in 1 L DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 22 g in 1 L LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) (LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) LACTIC ACID, UNSPECIFIED FORM 5.4 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 7.07 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 2.21 g in 1 L POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 0.314 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-111-06 2 in 1 CASE 10/10/2008 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021703 10/25/2006 PRISMASOL BK0/0/1.2
magnesium chloride, lactic acid, sodium chloride and sodium bicarbonate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-113 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 2.44 g in 1 L LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) (LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) LACTIC ACID, UNSPECIFIED FORM 5.4 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.46 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 3.09 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-113-06 2 in 1 CASE 10/10/2008 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021703 10/25/2006 PRISMASOL BGK4/0/1.2
magnesium chloride, dextrose monohydrate, lactic acid, sodium chloride, sodium bicarbonate and potassium chloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-114 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 2.44 g in 1 L DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 22 g in 1 L LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) (LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) LACTIC ACID, UNSPECIFIED FORM 5.4 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.46 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 3.09 g in 1 L POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 0.314 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-114-06 2 in 1 CASE 10/10/2008 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021703 10/25/2006 PHOXILLUM BK4/2.5
calcium chloride, magnesium chloride, sodium chloride, sodium bicarbonate, potassium chloride and sodium phosphate dibasic dihydrate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-116 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB, CHLORIDE ION - UNII:Q32ZN48698) CALCIUM CHLORIDE 3.68 g in 1 L MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 3.05 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.34 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 3.09 g in 1 L POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 0.314 g in 1 L SODIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: 94255I6E2T) (SODIUM CATION - UNII:LYR4M0NH37, PHOSPHATE ION - UNII:NK08V8K8HR) SODIUM PHOSPHATE, DIBASIC, DIHYDRATE 0.187 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-116-06 2 in 1 CASE 01/13/2015 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA207026 01/13/2015 PHOXILLUM B22K4/0
magnesium chloride, sodium chloride, sodium bicarbonate, potassium chloride and sodium phosphate dibasic dihydrate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24571-117 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838, CHLORIDE ION - UNII:Q32ZN48698) MAGNESIUM CHLORIDE 3.05 g in 1 L SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 6.95 g in 1 L SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37, BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 2.21 g in 1 L POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 0.314 g in 1 L SODIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: 94255I6E2T) (SODIUM CATION - UNII:LYR4M0NH37, PHOSPHATE ION - UNII:NK08V8K8HR) SODIUM PHOSPHATE, DIBASIC, DIHYDRATE 0.187 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CARBON DIOXIDE (UNII: 142M471B3J) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24571-117-06 2 in 1 CASE 01/13/2015 1 5 L in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA207026 01/13/2015 Labeler - Baxter Healthcare Corporation (005083209) Registrant - Baxter Healthcare Corporation (005083209) Establishment Name Address ID/FEI Business Operations Baxter, S.A. de C.V. 810432484 ANALYSIS(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114) , LABEL(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114) , MANUFACTURE(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114) , PACK(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114) , STERILIZE(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114) Establishment Name Address ID/FEI Business Operations Bieffe Medital S.p.A. 437668413 ANALYSIS(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114, 24571-116, 24571-117) , LABEL(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114, 24571-116, 24571-117) , MANUFACTURE(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114, 24571-116, 24571-117) , PACK(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114, 24571-116, 24571-117) , STERILIZE(24571-108, 24571-105, 24571-103, 24571-102, 24571-111, 24571-113, 24571-114, 24571-116, 24571-117)