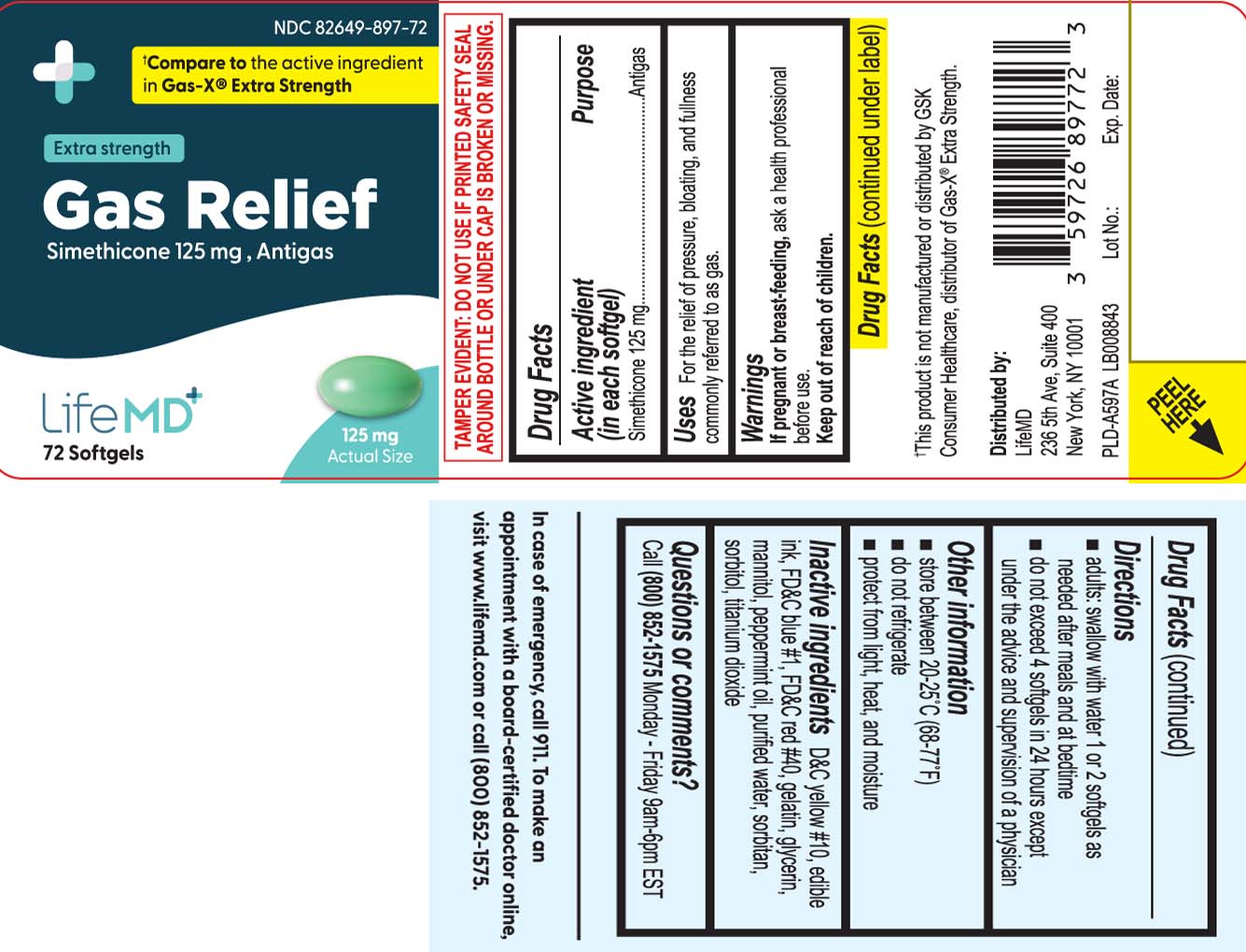
Label: GAS RELIEF EXTRA STRENGTH- simethicone capsule, liquid filled
- NDC Code(s): 82649-897-72
- Packager: LifeMD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each softgel)
- Purpose
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
†Compare to the active ingredient in Gas-X® Extra Strength
Extra strength
Gas Relief
Simethicone 125 mg, anti-gas
Softgels
†This product is not manufactured or distributed by GSK Consumer Healthcare, distributor of Gas-X® Extra Strength.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL AROUND BOTTLE OR UNDER CAP IS BROKEN OR MISSING.
Distributed by:
LifeMD
236 5th Ave, Suite 400
New York, NY 10001
- Product Label
-
INGREDIENTS AND APPEARANCE
GAS RELIEF EXTRA STRENGTH
simethicone capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82649-897 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 125 mg Inactive Ingredients Ingredient Name Strength D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) PEPPERMINT OIL (UNII: AV092KU4JH) WATER (UNII: 059QF0KO0R) SORBITAN (UNII: 6O92ICV9RU) SORBITOL (UNII: 506T60A25R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) MANNITOL (UNII: 3OWL53L36A) Product Characteristics Color green Score no score Shape OVAL Size 9mm Flavor Imprint Code PO Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82649-897-72 72 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 08/26/2022 08/26/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 08/26/2022 08/26/2025 Labeler - LifeMD (015464409)