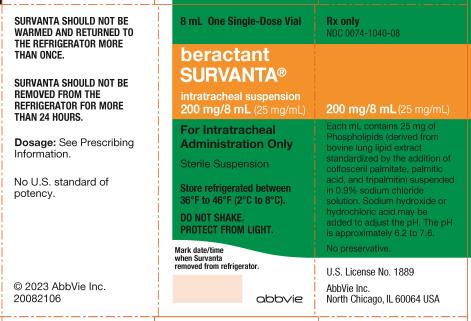
Label: SURVANTA- beractant suspension
- NDC Code(s): 0074-1040-04, 0074-1040-08
- Packager: AbbVie Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated October 15, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
SURVANTA® contains beractant, a pulmonary surfactant, which is a natural bovine lung extract containing phospholipids, neutral lipids, fatty acids, and surfactant-associated proteins (SP) to which colfosceril palmitate (dipalmitoylphosphatidylcholine), palmitic acid, and tripalmitin are added to standardize the composition and to mimic surface-tension lowering properties of natural lung surfactant. The resulting composition provides 25 mg/mL of phospholipids (including 11.0-15.5 mg/mL of disaturated phosphatidylcholine), 0.5-1.75 mg/mL of triglycerides, 1.4-3.5 mg/mL of free fatty acids, and less than 1 mg/mL of SP (including SP-B, a 79-amino acid protein, and SP-C, a 35-amino acid peptide).
SURVANTA (beractant) intratracheal suspension is a sterile, preservative-free, non-pyrogenic off-white to light brown liquid supplied in single-dose glass vials for intratracheal use only. Each vial contains 4 mL (100 mg phospholipids) or 8 mL (200 mg phospholipids). Each mL of SURVANTA contains 25 mg of phospholipids. It is suspended in 0.9% sodium chloride solution and heat-sterilized. SURVANTA contains no preservatives. Sodium hydroxide or hydrochloric acid may be added to adjust the pH. The pH is approximately 6.2 to 7.6.
-
CLINICAL PHARMACOLOGY
Endogenous pulmonary surfactant lowers surface tension on alveolar surfaces during respiration and stabilizes the alveoli against collapse at resting transpulmonary pressures. Deficiency of pulmonary surfactant causes Respiratory Distress Syndrome (RDS) in premature infants. SURVANTA replenishes surfactant and restores surface activity to the lungs of these infants.
Activity
In vitro, SURVANTA reproducibly lowers minimum surface tension to less than 8 dynes/cm as measured by the pulsating bubble surfactometer and Wilhelmy Surface Balance. In situ, SURVANTA restores pulmonary compliance to excised rat lungs artificially made surfactant-deficient. In vivo, single SURVANTA doses improve lung pressure-volume measurements, lung compliance, and oxygenation in premature rabbits and sheep.
Animal Metabolism
SURVANTA is administered directly to the target organ, the lungs, where biophysical effects occur at the alveolar surface. In surfactant-deficient premature rabbits and lambs, alveolar clearance of radio-labelled lipid components of SURVANTA is rapid. Most of the dose becomes lung-associated within hours of administration, and the lipids enter endogenous surfactant pathways of reutilization and recycling. In surfactant-sufficient adult animals, SURVANTA clearance is more rapid than in premature and young animals. There is less reutilization and recycling of surfactant in adult animals.
Limited animal experiments have not found effects of SURVANTA on endogenous surfactant metabolism. Precursor incorporation and subsequent secretion of saturated phosphatidylcholine in premature sheep are not changed by SURVANTA treatments.
No information is available about the metabolic fate of the surfactant-associated proteins in SURVANTA. The metabolic disposition in humans has not been studied.
-
CLINICAL STUDIES
Clinical effects of SURVANTA were demonstrated in six single-dose and four multiple-dose randomized, multi-center, controlled clinical trials involving approximately 1700 infants. Three open trials, including a Treatment IND, involved more than 8500 infants. Each dose of SURVANTA in all studies was 100 mg phospholipids/kg birth weight and was based on published experience with Surfactant TA, a lyophilized powder dosage form of SURVANTA having the same composition. SURVANTA significantly reduces the incidence of RDS, mortality due to RDS and air leak complications.
Prevention Studies
Infants of 600-1250 g birth weight and 23 to 29 weeks estimated gestational age were enrolled in two multiple-dose studies. A dose of SURVANTA was given within 15 minutes of birth to prevent the development of RDS. Up to three additional doses in the first 48 hours, as often as every 6 hours, were given if RDS subsequently developed and infants required mechanical ventilation with an FiO2 ≥ 0.30. Results of the studies at 28 days of age are shown in Table 1.
Table 1. Study 1 SURVANTA Control P-Value Number infants studied 119 124 Incidence of RDS (%) 27.6 63.5 < 0.001 Death due to RDS (%) 2.5 19.5 < 0.001 Death or BPD due to RDS (%) 48.7 52.8 0.536 Death due to any cause (%) 7.6 22.8 0.001 Air Leaksa (%) 5.9 21.7 0.001 Pulmonary interstitial emphysema (%) 20.8 40.0 0.001 Study 2b SURVANTA Control P-Value Number infants studied 91 96 Incidence of RDS (%) 28.6 48.3 0.007 Death due to RDS (%) 1.1 10.5 0.006 Death or BPD due to RDS (%) 27.5 44.2 0.018 Death due to any cause C(%) 16.5 13.7 0.633 Air Leaks a(%) 14.5 19.6 0.374 Pulmonary interstitial emphysema (%) 26.5 33.2 0.298 aPneumothorax or pneumopericardium
bStudy discontinued when Treatment IND initiated
cNo cause of death in the SURVANTA group was significantly increased; the higher number of deaths in this group was due to the sum of all causes.Rescue Studies
Infants of 600-1750 g birth weight with RDS requiring mechanical ventilation and an FiO2 ≥ 0.40 were enrolled in two multiple-dose rescue studies. The initial dose of SURVANTA was given after RDS developed and before 8 hours of age. Infants could receive up to three additional doses in the first 48 hours, as often as every 6 hours, if they required mechanical ventilation and an FiO2 ≥ 0.30. Results of the studies at 28 days of age are shown in Table 2.
Table 2. Study 3a SURVANTA Control P-Value Number infants studied 198 193 Death due to RDS (%) 11.6 18.1 0.071 Death or BPD due to RDS (%) 59.1 66.8 0.102 Death due to any cause (%) 21.7 26.4 0.285 Air Leaksb (%) 11.8 29.5 <0.001 Pulmonary interstitial emphysema (%) 16.3 34.0 <0.001 Study 4 SURVANTA Control P-Value Number infants studied 204 203 Death due to RDS (%) 6.4 22.3 < 0.001 Death or BPD due to RDS (%) 43.6 63.4 < 0.001 Death due to any cause (%) 15.2 28.2 0.001 Air Leaksb (%) 11.2 22.2 0.005 Pulmonary interstitial emphysema (%) 20.8 44.4 < 0.001 aStudy discontinued when Treatment IND initiated
bPneumothorax or pneumopericardiumAcute Clinical Effects
Marked improvements in oxygenation may occur within minutes of administration of SURVANTA.
All controlled clinical studies with SURVANTA provided information regarding the acute effects of SURVANTA on the arterial-alveolar oxygen ratio (a/APO2), FiO2, and mean airway pressure (MAP) during the first 48 to 72 hours of life. Significant improvements in these variables were sustained for 48-72 hours in SURVANTA-treated infants in four single-dose and two multiple-dose rescue studies and in two multiple-dose prevention studies. In the single-dose prevention studies, the FiO2 improved significantly.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
SURVANTA can rapidly affect oxygenation and lung compliance within minutes of administration of SURVANTA. Therefore, its use should be restricted to a highly supervised clinical setting with immediate availability of clinicians experienced with intubation, ventilator management, and general care of premature infants. Infants receiving SURVANTA should be frequently monitored with arterial or transcutaneous measurement of systemic oxygen and carbon dioxide.
During the dosing procedure, transient episodes of bradycardia and decreased oxygen saturation have been reported. If these occur, stop the dosing procedure and initiate appropriate measures to alleviate the condition. After stabilization, resume the dosing procedure.
-
PRECAUTIONS
General
Rales and moist breath sounds can occur transiently after administration. Endotracheal suctioning or other remedial action is not necessary unless clear-cut signs of airway obstruction are present.
Increased probability of post-treatment nosocomial sepsis in SURVANTA-treated infants was observed in the controlled clinical trials (Table 3). The increased risk for sepsis among SURVANTA-treated infants was not associated with increased mortality among these infants. The causative organisms were similar in treated and control infants. There was no significant difference between groups in the rate of post-treatment infections other than sepsis.
Use of SURVANTA in infants less than 600 g birth weight or greater than 1750 g birth weight has not been evaluated in controlled trials. There is no controlled experience with use of SURVANTA in conjunction with experimental therapies for RDS (eg, high-frequency ventilation or extracorporeal membrane oxygenation).
No information is available on the effects of doses other than 100 mg phospholipids/kg, more than four doses, dosing more frequently than every 6 hours, or administration after 48 hours of age.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with SURVANTA. SURVANTA was negative when tested in the Ames test for mutagenicity. Using the maximum feasible dose volume, SURVANTA up to 500 mg phospholipids/kg/day (approximately one-third the premature infant dose based on mg/m2/day) was administered subcutaneously to newborn rats for 5 days. The rats reproduced normally and there were no observable adverse effects in their offspring.
-
ADVERSE REACTIONS
The most commonly reported adverse experiences were associated with the dosing procedure. In the multiple-dose controlled clinical trials, each dose of SURVANTA was divided into four quarter-doses which were instilled through a catheter inserted into the endotracheal tube by briefly disconnecting the endotracheal tube from the ventilator. Transient bradycardia occurred with 11.9% of doses. Oxygen desaturation occurred with 9.8% of doses.
Other reactions during the dosing procedure occurred with fewer than 1% of doses and included endotracheal tube reflux, pallor, vasoconstriction, hypotension, endotracheal tube blockage, hypertension, hypocarbia, hypercarbia, and apnea. No deaths occurred during the dosing procedure, and all reactions resolved with symptomatic treatment.
The occurrence of concurrent illnesses common in premature infants was evaluated in the controlled trials. The rates in all controlled studies are in Table 3.
Table 3. All Controlled Studies Concurrent Event SURVANTA (%) Control (%) P-Valuea Patent ductus arteriosus 46.9 47.1 0.814 Intracranial hemorrhage 48.1 45.2 0.241 Severe intracranial hemorrhage 24.1 23.3 0.693 Pulmonary air leaks 10.9 24.7 < 0.001 Pulmonary interstitial emphysema 20.2 38.4 < 0.001 Necrotizing enterocolitis 6.1 5.3 0.427 Apnea 65.4 59.6 0.283 Severe apnea 46.1 42.5 0.114 Post-treatment sepsis 20.7 16.1 0.019 Post-treatment infection 10.2 9.1 0.345 Pulmonary hemorrhage 7.2 5.3 0.166 aP-value comparing groups in controlled studies When all controlled studies were pooled, there was no difference in intracranial hemorrhage. However, in one of the single-dose rescue studies and one of the multiple-dose prevention studies, the rate of intracranial hemorrhage was significantly higher in SURVANTA patients than control patients (63.3% v 30.8%, P = 0.001; and 48.8% v 34.2%, P = 0.047, respectively). The rate in a Treatment IND involving approximately 8100 infants was lower than in the controlled trials.
In the controlled clinical trials, there was no effect of SURVANTA on results of common laboratory tests: white blood cell count and serum sodium, potassium, bilirubin, and creatinine.
More than 4300 pretreatment and post-treatment serum samples from approximately 1500 patients were tested by Western Blot Immunoassay for antibodies to surfactant-associated proteins SP-B and SP-C. No IgG or IgM antibodies were detected.
Several other complications are known to occur in premature infants. The following conditions were reported in the controlled clinical studies. The rates of the complications were not different in treated and control infants, and none of the complications were attributed to SURVANTA.
Respiratory
lung consolidation, blood from the endotracheal tube, deterioration after weaning, respiratory decompensation, subglottic stenosis, paralyzed diaphragm, respiratory failure.
Cardiovascular
hypotension, hypertension, tachycardia, ventricular tachycardia, aortic thrombosis, cardiac failure, cardio-respiratory arrest, increased apical pulse, persistent fetal circulation, air embolism, total anomalous pulmonary venous return.
Gastrointestinal
abdominal distention, hemorrhage, intestinal perforations, volvulus, bowel infarct, feeding intolerance, hepatic failure, stress ulcer.
Follow-Up Evaluations
To date, no long-term complications or sequelae of SURVANTA therapy have been found.
Single-Dose Studies
Six-month adjusted-age follow-up evaluations of 232 infants (115 treated) demonstrated no clinically important differences between treatment groups in pulmonary and neurologic sequelae, incidence or severity of retinopathy of prematurity, rehospitalizations, growth, or allergic manifestations.
Multiple-Dose Studies
Six-month adjusted age follow-up evaluations have been completed in 631 (345 treated) of 916 surviving infants. There were significantly less cerebral palsy and need for supplemental oxygen in SURVANTA infants than controls. Wheezing at the time of examination was significantly more frequent among SURVANTA infants, although there was no difference in bronchodilator therapy.
Final twelve-month follow-up data from the multiple-dose studies are available from 521 (272 treated) of 909 surviving infants. There was significantly less wheezing in SURVANTA infants than controls, in contrast to the six-month results. There was no difference in the incidence of cerebral palsy at twelve months.
Twenty-four month adjusted age evaluations were completed in 429 (226 treated) of 906 surviving infants. There were significantly fewer SURVANTA infants with rhonchi, wheezing, and tachypnea at the time of examination. No other differences were found.
-
OVERDOSAGE
Overdosage with SURVANTA has not been reported. Based on animal data, overdosage might result in acute airway obstruction. Treatment should be symptomatic and supportive.
Rales and moist breath sounds can transiently occur after SURVANTA is given, and do not indicate overdosage. Endotracheal suctioning or other remedial action is not required unless clear-cut signs of airway obstruction are present.
-
DOSAGE AND ADMINISTRATION
Important Administration Instructions
For intratracheal administration only.
SURVANTA should be administered by or under the supervision of clinicians experienced in intubation, ventilator management, and general care of premature infants. The administration of SURVANTA is facilitated if one person administers the dose while another person positions and monitors the infant.
Before administering SURVANTA, assure proper placement and patency of the endotracheal tube. At the discretion of the clinician, the endotracheal tube may be suctioned before administering SURVANTA. The infant should be allowed to stabilize before proceeding with dosing.
Administer SURVANTA intratracheally by instillation through a 5 French end-hole catheter.
Recommended Dosage
Each dose of SURVANTA is 100 mg of phospholipids/kg birth weight (4 mL/kg).
In the prevention strategy, in premature infants with evidence of surfactant deficiency, give the first dose of SURVANTA as soon as possible, preferably within 15 minutes of birth.
To treat infants with RDS confirmed by radiographic and clinical findings, give the first dose of SURVANTA as soon as possible, preferably by 8 hours of age.
Four doses of SURVANTA can be administered in the first 48 hours of life. Doses should be given no more frequently than every 6 hours.
The need for additional doses of SURVANTA is determined by evidence of continuing respiratory distress. Radiographic confirmation of RDS should be obtained before administering additional doses to those who received a prevention dose.
Preparation of the SURVANTA Suspension
SURVANTA should be inspected visually for discoloration prior to administration. The color of SURVANTA is off-white to light brown. If settling occurs during storage, swirl the vial gently (DO NOT SHAKE) to redisperse. Do not filter SURVANTA. Some foaming at the surface may occur during handling and is inherent in the nature of the product.
SURVANTA is stored refrigerated (36°F to 46°F [2°C to 8°C]). Date and time need to be recorded in the box on front of the carton or vial, whenever SURVANTA is removed from the refrigerator. Before administration, SURVANTA should be warmed by standing at room temperature for at least 20 minutes or warmed in the hand for at least 8 minutes. Artificial warming methods should not be used. If a prevention dose is to be given, preparation of SURVANTA should begin before the infant’s birth.
Unopened, unused vials of SURVANTA that have been warmed to room temperature may be returned to the refrigerator within 24 hours of warming, and stored for future use. SURVANTA SHOULD NOT BE REMOVED FROM THE REFRIGERATOR FOR MORE THAN 24 HOURS. SURVANTA SHOULD NOT BE WARMED AND RETURNED TO THE REFRIGERATOR MORE THAN ONCE. Each single-dose vial of SURVANTA should be entered only once. Used vials with residual drug should be discarded.
SURVANTA does not require reconstitution or sonication before use.
Administration
For endotracheal administration using a 5 French end-hole catheter:
1. Slowly withdraw the entire contents of the vial into a plastic syringe through a large-gauge needle (e.g., at least 20 gauge).
2. Attach the premeasured 5 French end-hole catheter to the syringe. Fill the catheter with SURVANTA. Discard excess SURVANTA through the catheter so that only the total dose to be given remains in the syringe.
3. When administering SURVANTA using a 5 French end-hole catheter, administer in four quarter-dose aliquots. Each quarter-dose is administered with the infant in a different position:
- Head and body inclined 5-10° down, head turned to the right
- Head and body inclined 5-10° down, head turned to the left
- Head and body inclined 5-10° up, head turned to the right
- Head and body inclined 5-10° up, head turned to the left
4. First quarter-dose aliquot of SURVANTA suspension:
a. Position the infant appropriately in one of the four recommended positions.
b. Insert the 5-French end-hole catheter into the endotracheal tube. The tip of the catheter should protrude just beyond the end of the endotracheal tube above the infant’s carina. SURVANTA should not be instilled into a mainstem bronchus.
c. Gently inject the first quarter-dose aliquot through the catheter over 2-3 seconds.
d. After the first aliquot is instilled, remove the catheter from the endotracheal tube and manually ventilate the infant for at least 30 seconds or until clinically stable. Ventilate with sufficient oxygen to prevent cyanosis and sufficient positive pressure to provide adequate air exchange and chest wall excursion.
5. When the infant is stable, reposition the infant for instillation of the next quarter-dose.
6. Instill each remaining quarter-dose using the same procedures.
7. After instillation of the final quarter-dose, remove the catheter without flushing it. Do not suction the infant for 1 hour after dosing unless signs of significant airway obstruction occur.
- Head and body inclined 5-10° down, head turned to the right
-
HOW SUPPLIED
SURVANTA (beractant) intratracheal suspension is supplied in 100 mg/4 mL single-dose glass vials (NDC 0074-1040-04) or 200 mg/8 mL single-dose glass vials (NDC 0074-1040-08). Each mL contains 25 mg of phospholipids suspended in 0.9% sodium chloride solution. The color is off-white to light brown.
Store unopened vials refrigerated at 36°F to 46°F (2°C to 8°C). Do not shake. Protect from light. Store vials in carton until ready for use. Vials are for one-time use and for only one patient. Upon opening, discard unused drug.
LITHO IN USA
AbbVie Inc.
North Chicago, IL 60064, U.S.A.
US License Number 1889
20065798 October, 2020
- Principal Display Panel
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SURVANTA
beractant suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0074-1040 Route of Administration ENDOTRACHEAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BERACTANT (UNII: S866O45PIG) (BERACTANT PHOSPHOLIPIDS - UNII:LJA7V5FCW8) BERACTANT PHOSPHOLIPIDS 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength COLFOSCERIL PALMITATE (UNII: 319X2NFW0A) TRIPALMITIN (UNII: D133ZRF50U) PALMITIC ACID (UNII: 2V16EO95H1) SODIUM CHLORIDE (UNII: 451W47IQ8X) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Product Characteristics Color white (off-white to light brown) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0074-1040-04 1 in 1 CARTON 07/01/1991 1 4 mL in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product 2 NDC:0074-1040-08 1 in 1 CARTON 07/01/1991 2 8 mL in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA020032 07/01/1991 Labeler - AbbVie Inc. (078458370)