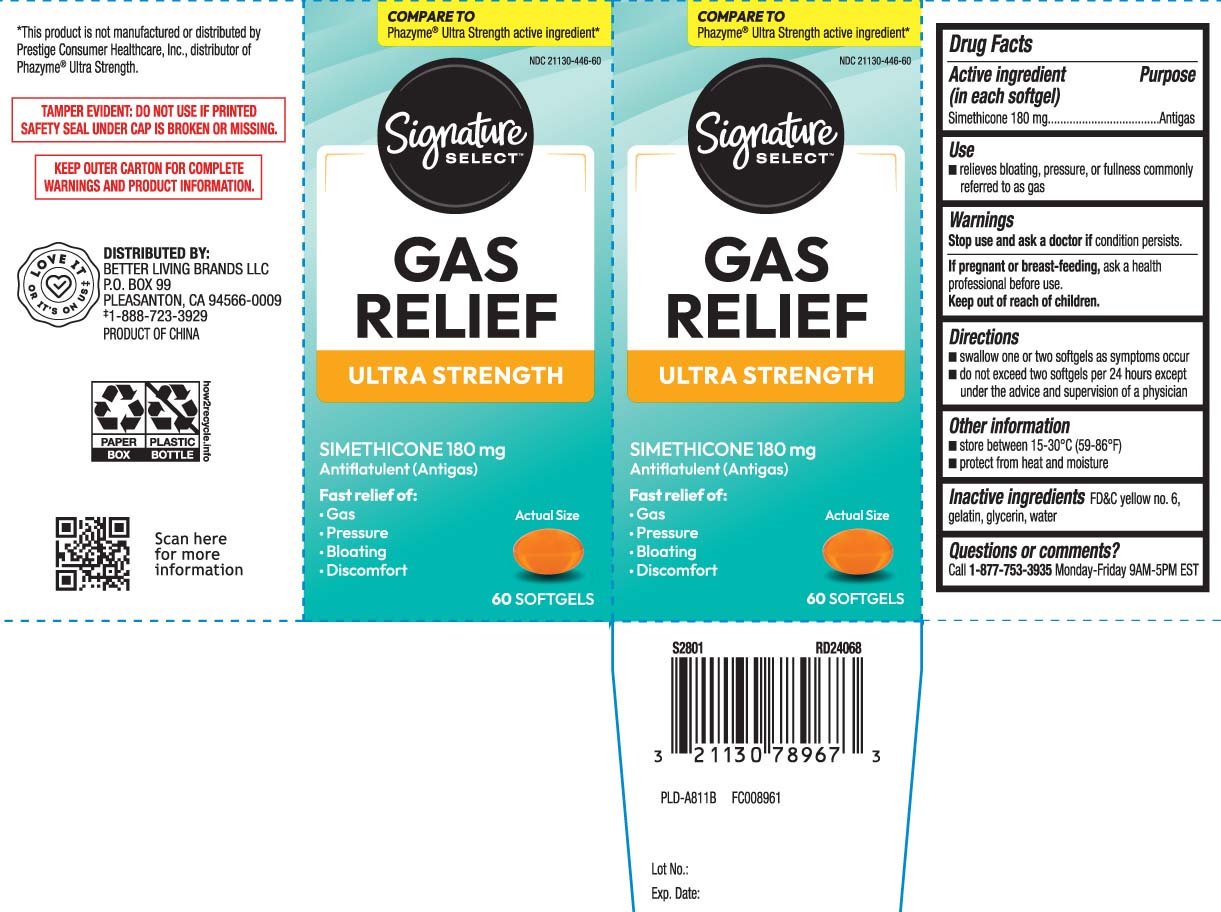
Label: GAS RELIEF- simethicone capsule, liquid filled
- NDC Code(s): 21130-446-60
- Packager: Safeway, Inc.
- This is a repackaged label.
- Source NDC Code(s): 51013-115
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 20, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each softgel)
- Purpose
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
COMPARE TO Phazyme® Ultra Strength active ingredient*
GAS RELIEF
ULTRA STRENGTH
SIMETHICONE 180 mg
Antiflatulent (Antigas)
Fast relief of:
- Gas
- Pressure
- Bloating
- Discomfort
Softgels
*This product is not manufactured or distributed by Prestige Consumer Healthcare, Inc., distributor of Phazyme® Ultra Strength.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
DISTRIBUTED BY
BETTER LIVING BRANDS LLC
P.O. BOX 99, PLEASANTON, CA 94566-0009
- Package Label
-
INGREDIENTS AND APPEARANCE
GAS RELIEF
simethicone capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-446(NDC:51013-115) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 180 mg Inactive Ingredients Ingredient Name Strength FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Product Characteristics Color orange (clear) Score no score Shape CAPSULE (oval) Size 10mm Flavor Imprint Code PC3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-446-60 1 in 1 BOX 12/31/2023 1 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M002 12/31/2023 Labeler - Safeway, Inc. (009137209)