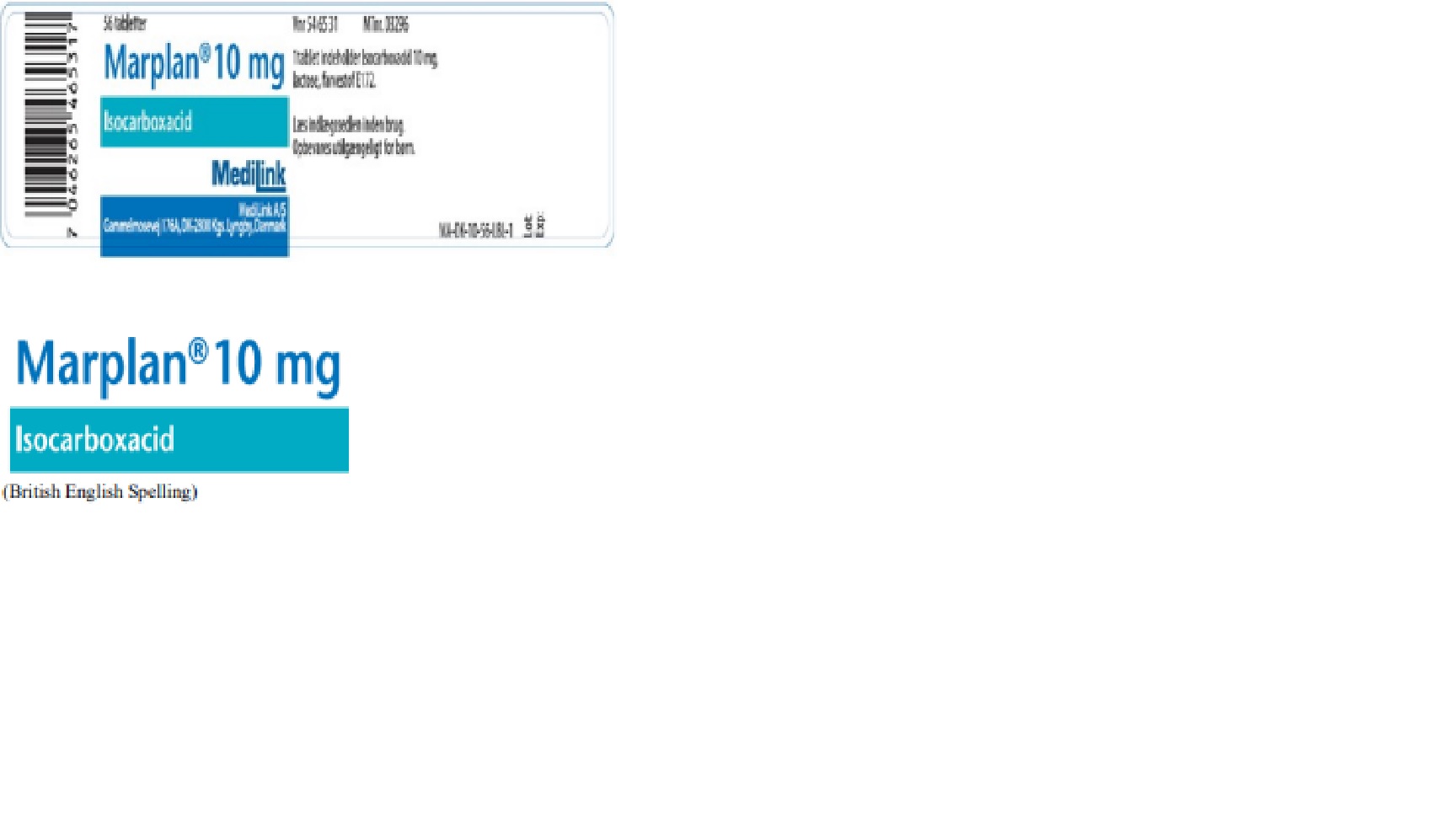
Label: MARPLAN- isocarboxacid tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 72604-032-01 - Packager: MediLink A/S
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Unapproved drug for use in drug shortage
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 20, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Temporary Importation of Marplan (iscarboxacid) Tablets to Address Critical Drug Shortage
IMPORTANT PRESCRIBING INFORMATION
December 20, 2018
Subject: Temporary Importation of Marplan® (isocarboxacid) Tablets to Address Critical Drug Shortage
Dear Healthcare Professional,
In order to address the critical shortage of Marplan® (isocarboxazid) 10 mg Tablets in the United States (US) market, Validus Pharmaceuticals LLC is coordinating with the U.S. Food and Drug Administration (FDA) to make Marplan available to our patients. Validus has initiated temporary importation of another manufacturer’s Marplan (isocarboxacid) into the U.S. market. This product is manufactured and marketed in Denmark by MediLink.
At this time, no other entity except Validus Pharmaceuticals LLC is authorized by the FDA to import and distribute MediLink’s Marplan (isocarboxacid) 10 mg Tablets in the United States. FDA has not approved MediLink’s Marplan (isocarboxacid) 10 mg Tablets but does not object to its importation into the United States. Effective immediately, and during this temporary period, Validus will offer the following product:
Product Name and Description Bottle Count NDC Code Manufacturer Marplan (isocarboxacid) 10 mg Tablets 56 tablets 72604-032-01 MediLink
It is important to note the following:
• The labeling is in Danish. MediLink’s Marplan (isocarboxacid) 10 mg Tablets’ labeling, with the exception of the product name “Marplan”, will appear in Danish. There are key differences in the labeling between Validus’ Marplan and Medilink’s Marplan. A side by side comparison of the FDA approved Marplan (isocarboxazid) Prescribing Information and the imported Marplan (isocarboxacid) product information is included at the end of this letter.
• The tablets have different appearance. MediLink’s Marplan (isocarboxacid) 10 mg tablets are “dull pink” in color whereas Validus’ Marplan (isocarboxazid) 10 mg tablets are “peach-colored.” MediLink’s Marplan 10 mg tablets are imprinted with “CL 3M3” whereas Validus’ Marplan 10 mg tablets are imprinted with “Marplan” and “10” on one side. Please refer to the “Product Comparison” table at the end of this letter for details and images.
• The barcode may not register accurately in the U.S. scanning systems. Institutions should manually input the product into their systems and confirm that the barcode Marplan DHCP Letter Page 1 of 22 systems do not provide incorrect information when the product is scanned. Alternative procedures should be followed to assure that the correct drug product is being used and administered to individual patients.
To order, or if you have any questions about MediLink’s Marplan, please contact Validus Customer Service by phone at 1-866-982-5438 or by e-mail at info@validuspharma.com.
To report Adverse Events or Quality Problems among patients who have received - MediLink’s Marplan, please contact Validus at 1-866-982-5438 or by e-mail at info@validuspharma.com. Adverse events or quality problems may also be reported to FDA’s MedWatch Adverse reporting Program either online, by regular mail, or by fax:
• Complete and submit the report Online: www.fda.gov/medwatch/report.htm
• Regular Mail or Fax: Download form www.fda.gov/Medwatch/getforms.htm or call 1- 800-332-1088 to request a reporting form. Complete and return the form to the address on the pre-addressed form, or submit it by fax to 1-800-FDA-0178 (1-800- 332-1078).
If you have any questions about the information contained in this letter or the safe and effective use of MediLink’s Marplan (isocarboxacid) 10mg Tablets, please contact Validus at 1-866-982- 5438 or by e-mail at info@validuspharma.com.
Sincerely,
Gina Walljasper
Sr. Director Regulatory and Quality Operations
Validus Pharmaceuticals LLC
- Principal Display Panel - Marplan (isocarboxacid) 10mg Tablets
-
INGREDIENTS AND APPEARANCE
MARPLAN
isocarboxacid tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:72604-032 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOCARBOXAZID (UNII: 34237V843T) (ISOCARBOXAZID - UNII:34237V843T) ISOCARBOXAZID 10 mg Product Characteristics Color pink (Dull Pink) Score no score Shape ROUND Size 8mm Flavor Imprint Code CL;3M3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72604-032-01 1 in 1 CARTON 12/20/2018 1 56 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug for use in drug shortage 12/20/2018 Labeler - MediLink A/S (587619958)