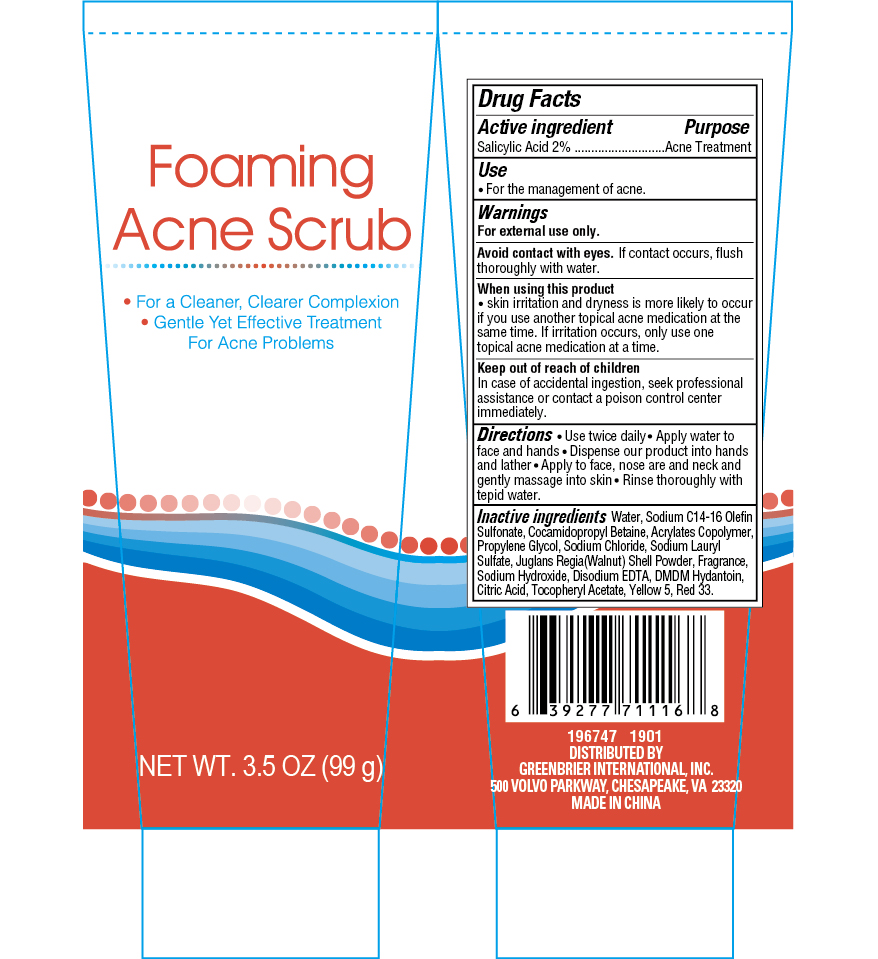
Label: FOAMING ACNE SCRUB FOR A CLEANER, CLEARER COMPLEXION- salicylic acid gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 33992-3038-1 - Packager: Greenbrier International Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 18, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PURPOSE
- ACTIVE INGREDIENT
- DOSAGE & ADMINISTRATION
- KEEP OUT OF REACH OF CHILDREN
- INACTIVE INGREDIENT
-
WARNINGS
Warnings
For external use only
When using this product
- Skin irritation and dryness are more likely to occur if you use another topical acne medication at the same time. If irritation occurs, oinly use one topical acne medication at a time.
- Avoid contact with eyes. If contact occurs, rinse thoroughly with water
- INDICATIONS & USAGE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FOAMING ACNE SCRUB FOR A CLEANER, CLEARER COMPLEXION
salicylic acid gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:33992-3038 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 2 g in 100 g Inactive Ingredients Ingredient Name Strength DMDM HYDANTOIN (UNII: BYR0546TOW) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 33 (UNII: 9DBA0SBB0L) BUTYL ACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID COPOLYMER (18000 MW) (UNII: JZ1374NL9E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) JUGLANS REGIA SHELL (UNII: PJ10MT7VKA) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM C14-16 OLEFIN SULFONATE (UNII: O9W3D3YF5U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) .ALPHA.-TOCOPHEROL ACETATE, D- (UNII: A7E6112E4N) WATER (UNII: 059QF0KO0R) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) ALOE VERA LEAF (UNII: ZY81Z83H0X) DISODIUM EDTA-COPPER (UNII: 6V475AX06U) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:33992-3038-1 99 g in 1 BOTTLE; Type 0: Not a Combination Product 12/16/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 12/16/2018 Labeler - Greenbrier International Inc. (610322518) Establishment Name Address ID/FEI Business Operations China Ningbo Shangge Cosmetic Technology Corp. 529287434 manufacture(33992-3038)