Label: EXTRA STRENGTH NASAL- phenylephrine hydrochloride spray
- NDC Code(s): 69842-443-30, 69842-443-40
- Packager: CVS PHARMACY
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
- Warnings
- Ask a doctor before use if you have
-
When using this product
- ▪
- do not use more than directed
- ▪
- do not use for more than 3 days. Use only as directed.
Frequent or prolonged use may cause nasal congestion to recur or worsen
- ▪
- temporary discomfort may occur such as burning, stinging, sneezing and increase in nasal discharge
- ▪
- use of this container by more than one person may spread infection
- Stop use and ask a doctor if
- If pregnant or breast-feeding
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
-
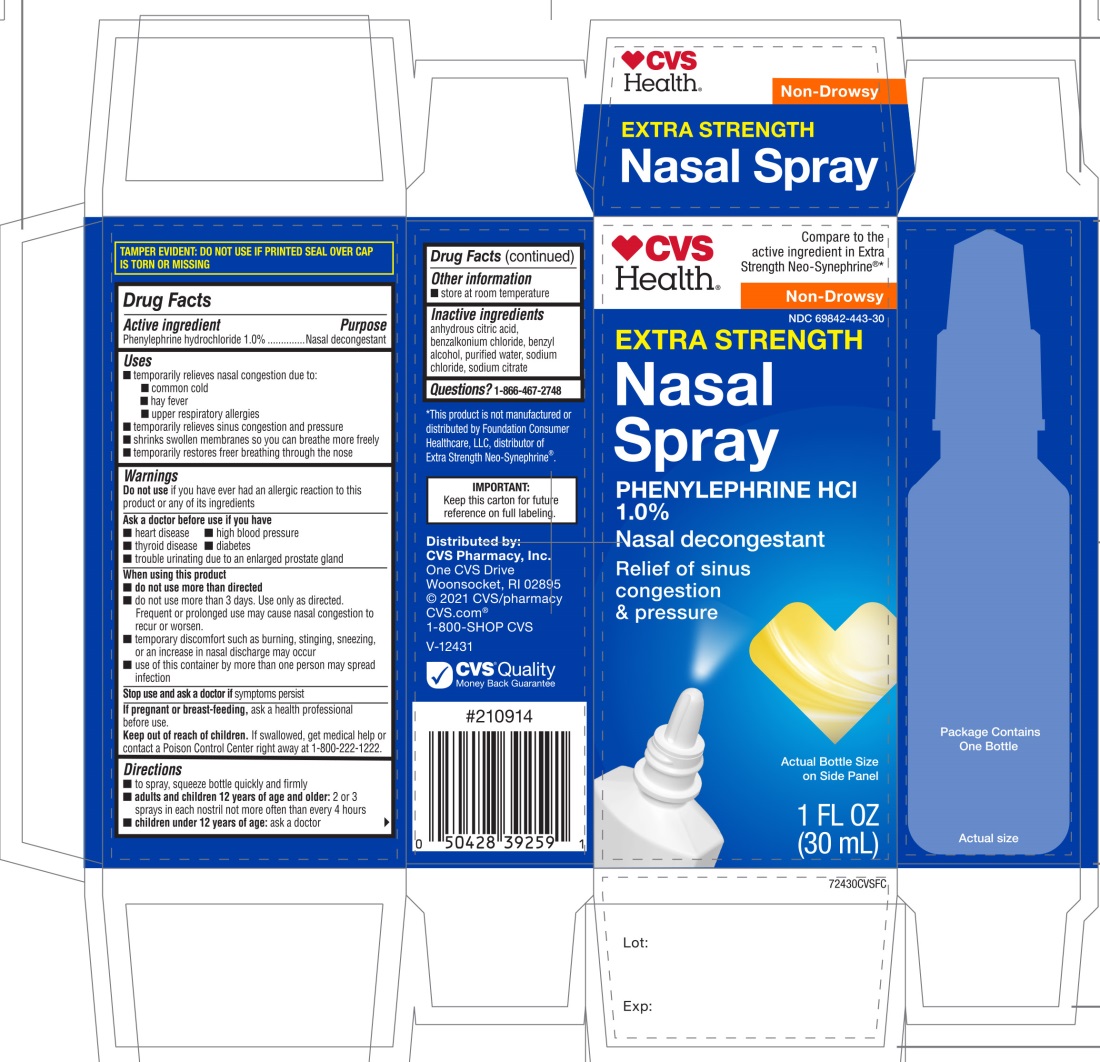
Principal Display
Compare to the active ingredient in Extra Strength Neo-Synephrine®*
Extra Strength
Nasal Spray
Phenylephrine hydrochloride 1.0 %
Nasal Decongestant
NON-DROWSY
COLD+ALLERGY
Nasal Congestion RELIEF
You can use every 4 hours
Fast Relief Of:
Sinus Congestion & Pressure
- Colds, Allergies
*This product is not manufactured or distributed by Foundation Consumer Healthcare LLC, distributor of Extra Strength Neo-Synephrine®.
IMPORTANT: Keep this carton for future reference on full labeling.
Do not use if printed seal over cap is torn or missing
Distributed by:
CVS Pharmacy, Inc.
One CVS Drive
Woonsocket, RI 02895
© 2021 CVS/pharmacy, CVS.com®
1-800-SHOP CVS V-12431
CVS Quality Money Back guarantee
* This product is not manufactured or distributed by Foundation Consumer Healthcare LLC owner of the registered trademark Neo-Synephrine® Extra Strength.
Package Label for Extra Strength Nasal Spray
-
INGREDIENTS AND APPEARANCE
EXTRA STRENGTH NASAL
phenylephrine hydrochloride sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-443 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 1 g in 100 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BENZYL ALCOHOL (UNII: LKG8494WBH) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-443-30 1 in 1 CARTON 09/21/2020 1 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 2 NDC:69842-443-40 1 in 1 CARTON 09/21/2020 2 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 09/21/2020 Labeler - CVS PHARMACY (062312574)