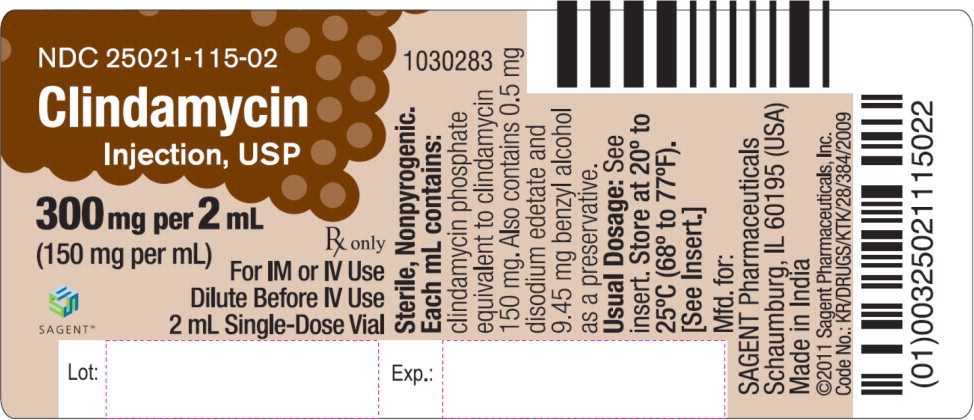
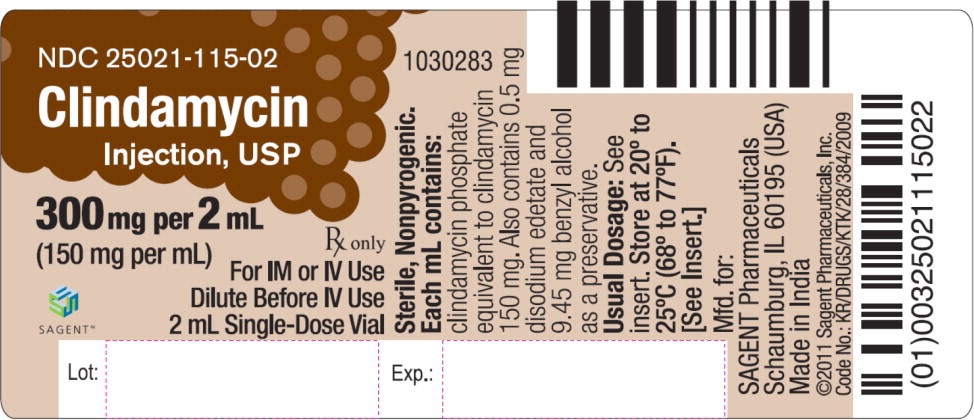
Label: CLINDAMYCIN- clindamycin phosphate injection, solution
- NDC Code(s): 25021-115-02, 25021-115-04, 25021-115-06
- Packager: Sagent Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 12, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
SAGENT®
Rx onlyTo reduce the development of drug-resistant bacteria and maintain the effectiveness of Clindamycin Injection, USP and other antibacterial drugs, Clindamycin Injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
Sterile Solution is for Intramuscular and Intravenous Use
-
BOXED WARNING
(What is this?)
WARNING
Clostridium difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including clindamycin and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
Because clindamycin therapy has been associated with severe colitis which may end fatally, it should be reserved for serious infections where less toxic antimicrobial agents are inappropriate, as described in the INDICATIONS AND USAGE section. It should not be used in patients with nonbacterial infections such as most upper respiratory tract infections. C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
-
DESCRIPTION
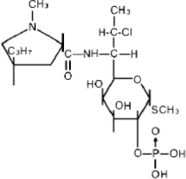
Clindamycin Injection, USP, a clear colorless to pale yellow sterile solution, contains clindamycin phosphate, a water soluble ester of clindamycin and phosphoric acid. Each mL contains the equivalent of 150 mg clindamycin, 0.5 mg disodium edetate and 9.45 mg benzyl alcohol added as preservative in each mL. Sodium hydroxide and/or hydrochloric acid may be added to adjust pH. Clindamycin is a semisynthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent compound lincomycin.
The chemical name of clindamycin phosphate is L-threo-α-D-galacto-Octopyranoside, methyl-7-chloro-6,7,8-trideoxy-6-[[(1-methyl-4-propyl-2-pyrrolidinyl)carbonyl]amino]-1-thio-,2-(dihydrogen phosphate), (2S-trans)-.
The molecular formula is C18H34ClN2O8PS and the molecular weight is 504.96.
The structural formula is represented below:
-
CLINICAL PHARMACOLOGY
Distribution
Biologically inactive clindamycin phosphate is converted to active clindamycin. By the end of short-term intravenous infusion, peak serum concentrations of active clindamycin are reached.
After intramuscular injection of clindamycin phosphate, peak concentrations of active clindamycin are reached within 3 hours in adults and 1 hour in pediatric patients. Serum concentration-time curves may be constructed from IV peak serum concentrations as given in Table 1 by application of elimination half-lives (see Excretion).
Serum concentrations of clindamycin can be maintained above the in vitro minimum inhibitory concentrations for most indicated organisms by administration of clindamycin phosphate every 8 to 12 hours in adults and every 6 to 8 hours in pediatric patients, or by continuous intravenous infusion. An equilibrium state is reached by the third dose.
No significant concentrations of clindamycin are attained in the cerebrospinal fluid even in the presence of inflamed meninges.
Metabolism
In vitro studies in human liver and intestinal microsomes indicated that clindamycin is predominantly metabolized by Cytochrome P450 3A4 (CYP3A4), with minor contribution from CYP3A5, to form clindamycin sulfoxide and a minor metabolite, N-desmethylclindamycin.
Excretion
Biologically inactive clindamycin phosphate disappears from the serum with 6 minutes of the average elimination half-life; however, the average serum elimination half-life of active clindamycin is about 3 hours in adults and 2½ hours in pediatric patients.
Specific Populations
Patients with Renal/Hepatic Impairment
The elimination half-life of clindamycin is increased slightly in patients with markedly reduced renal or hepatic function. Hemodialysis and peritoneal dialysis are not effective in removing clindamycin from the serum. Dosage schedules need not be modified in the presence of mild or moderate renal or hepatic disease.
Elderly Patients
Pharmacokinetic studies in elderly volunteers (61 to 79 years) and younger adults (18 to 39 years) indicate that age alone does not alter clindamycin pharmacokinetics (clearance, elimination half-life, volume of distribution, and area under the serum concentration-time curve) after IV administration of clindamycin phosphate. After oral administration of clindamycin hydrochloride, the average elimination half-life is increased to approximately 4 hours (range 3.4 to 5.1 h) in the elderly, compared to 3.2 hours (range 2.1 to 4.2 h) in younger adults. The extent of absorption, however, is not different between age groups and no dosage alteration is necessary for the elderly with normal hepatic function and normal (age-adjusted) renal function1.
Table 1. Average Peak and Trough Serum Concentrations of Active Clindamycin After Dosing with Clindamycin Phosphate *Data in this group from patients being treated for infection.
Dosage Regimen Peak
mcg/mLTrough
mcg/mLHealthy Adult Males (Post equilibrium) 600 mg IV in 30 min q6h 10.9 2.0 600 mg IV in 30 min q8h 10.8 1.1 900 mg IV in 30 min q8h 14.1 1.7 600 mg IM q12h* 9 Pediatric Patients (first dose)* 5 to 7 mg/kg IV in 1 hour 10 5 to 7 mg/kg IM 8 3 to 5 mg/kg IM 4 Obese Pediatric Patients Aged 2 to Less than 18 Years and Obese Adults Aged 18 to 20 Years
An analysis of pharmacokinetic data in obese pediatric patients aged 2 to less than 18 years and obese adults aged 18 to 20 years demonstrated that clindamycin clearance and volume of distribution, normalized by total body weight, are comparable regardless of obesity.
Microbiology
Mechanism of Action
Clindamycin inhibits bacterial protein synthesis by binding to the 23S RNA of the 50S subunit of the ribosome. Clindamycin is bacteriostatic.
Resistance
Resistance to clindamycin is most often caused by modification of specific bases of the 23S ribosomal RNA. Cross-resistance between clindamycin and lincomycin is complete. Because the binding sites for these antibacterial drugs overlap, cross-resistance is sometimes observed among lincosamides, macrolides and streptogramin B. Macrolide-inducible resistance to clindamycin occurs in some isolates of macrolide-resistant bacteria. Macrolide-resistant isolates of staphylococci and beta-hemolytic streptococci should be screened for induction of clindamycin resistance using the D-zone test.
Antimicrobial Activity
Clindamycin has been shown to be active against most of the isolates of the following microorganisms, both in vitro and in clinical infections (see INDICATIONS AND USAGE):
Gram-positive bacteria
- Staphylococcus aureus (methicillin-susceptible strains)
- Streptococcus pneumoniae (penicillin-susceptible strains)
- Streptococcus pyogenes
Anaerobic bacteria
- Clostridium perfringens
- Fusobacterium necrophorum
- Fusobacterium nucleatum
- Peptostreptococcus anaerobius
- Prevotella melaninogenica
The following in vitro data are available, but their clinical significance is unknown. At least 90 percent of the following bacteria exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for clindamycin against isolates of a similar genus or organism group. However, the efficacy of clindamycin in treating clinical infections due to these bacteria has not been established in adequate and well-controlled clinical trials.
Gram-positive bacteria
- Staphylococcus epidermidis (methicillin-susceptible strains)
- Streptococcus agalactiae
- Streptococcus anginosus
- Streptococcus mitis
- Streptococcus oralis
Anaerobic bacteria
- Actinomyces israelii
- Clostridium clostridioforme
- Eggerthella lenta
- Finegoldia (Peptostreptococcus) magna
- Micromonas (Peptostreptococcus) micros
- Prevotella bivia
- Prevotella intermedia
- Propionibacterium acnes
-
INDICATIONS AND USAGE
Clindamycin Injection, USP is indicated in the treatment of serious infections caused by susceptible anaerobic bacteria.
Clindamycin Injection, USP is also indicated in the treatment of serious infections due to susceptible strains of streptococci, pneumococci, and staphylococci. Its use should be reserved for penicillin-allergic patients or other patients for whom, in the judgment of the physician, a penicillin is inappropriate. Because of the risk of antibiotic-associated pseudomembranous colitis, as described in the BOXED WARNING, before selecting clindamycin the physician should consider the nature of the infection and the suitability of less toxic alternatives (e.g., erythromycin).
Bacteriologic studies should be performed to determine the causative organisms and their susceptibility to clindamycin.
Indicated surgical procedures should be performed in conjunction with antibiotic therapy.
Clindamycin Injection, USP is indicated in the treatment of serious infections caused by susceptible strains of the designated organisms in the conditions listed below:
Lower respiratory tract infections including pneumonia, empyema, and lung abscess caused by anaerobes, Streptococcus pneumoniae, other streptococci (except E. faecalis), and Staphylococcus aureus.
Skin and skin structure infections caused by Streptococcus pyogenes, Staphylococcus aureus, and anaerobes.
Gynecological infections including endometritis, nongonococcal tubo-ovarian abscess, pelvic cellulitis, and postsurgical vaginal cuff infection caused by susceptible anaerobes.
Intra-abdominal infections including peritonitis and intra-abdominal abscess caused by susceptible anaerobic organisms.
Septicemia caused by Staphylococcus aureus, streptococci (except Enterococcus faecalis), and susceptible anaerobes.
Bone and joint infections including acute hematogenous osteomyelitis caused by Staphylococcus aureus and as adjunctive therapy in the surgical treatment of chronic bone and joint infections due to susceptible organisms.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Clindamycin Injection, USP and other antibacterial drugs, Clindamycin Injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
- CONTRAINDICATIONS
-
WARNINGS
See BOXED WARNING.
Clostridium difficile Associated Diarrhea
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including clindamycin injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Anaphylactic and Severe Hypersensitivity Reactions
Anaphylactic shock and anaphylactic reactions have been reported (see ADVERSE REACTIONS).
Severe hypersensitivity reactions, including severe skin reactions such as toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS), some with fatal outcome, have been reported (see ADVERSE REACTIONS).
In case of such an anaphylactic or severe hypersensitivity reaction, discontinue treatment permanently and institute appropriate therapy.
A careful inquiry should be made concerning previous sensitivities to drugs and other allergens.
Benzyl Alcohol Toxicity in Pediatric Patients ("Gasping Syndrome")
This product contains benzyl alcohol as a preservative. The preservative benzyl alcohol has been associated with serious adverse events, including the "gasping syndrome", and death in pediatric patients. Although normal therapeutic doses of this product ordinarily deliver amounts of benzyl alcohol that are substantially lower than those reported in association with the "gasping syndrome", the minimum amount of benzyl alcohol at which toxicity may occur is not known.
The risk of benzyl alcohol toxicity depends on the quantity administered and the liver and kidneys' capacity to detoxify the chemical. Premature and low birth weight infants may be more likely to develop toxicity.
-
PRECAUTIONS
General
Review of experience to date suggests that a subgroup of older patients with associated severe illness may tolerate diarrhea less well. When clindamycin is indicated in these patients, they should be carefully monitored for change in bowel frequency.
Clindamycin injection products should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Clindamycin injection should be prescribed with caution in atopic individuals.
Certain infections may require incision and drainage or other indicated surgical procedures in addition to antibiotic therapy.
The use of clindamycin injection may result in overgrowth of nonsusceptible organisms-particularly yeasts. Should superinfections occur, appropriate measures should be taken as indicated by the clinical situation.
Clindamycin injection should not be injected intravenously undiluted as a bolus, but should be infused over at least 10 to 60 minutes as directed in the DOSAGE AND ADMINISTRATION section.
Clindamycin dosage modification may not be necessary in patients with renal disease. In patients with moderate to severe liver disease, prolongation of clindamycin half-life has been found. However, it was postulated from studies that when given every eight hours, accumulation should rarely occur. Therefore, dosage modification in patients with liver disease may not be necessary. However, periodic liver enzyme determinations should be made when treating patients with severe liver disease.
Prescribing clindamycin injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Information for Patients
Patients should be counseled that antibacterial drugs including clindamycin injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When clindamycin injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by clindamycin injection or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Laboratory Tests
During prolonged therapy periodic liver and kidney function tests and blood counts should be performed.
Drug Interactions
Clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, it should be used with caution in patients receiving such agents.
Clindamycin is metabolized predominantly by CYP3A4, and to a lesser extent by CYP3A5, to the major metabolite clindamycin sulfoxide and minor metabolite N-desmethylclindamycin. Therefore, inhibitors of CYP3A4 and CYP3A5 may increase plasma concentrations of clindamycin and inducers of these isoenzymes may reduce plasma concentrations of clindamycin. In the presence of strong CYP3A4 inhibitors, monitor for adverse reactions. In the presence of strong CYP3A4 inducers such as rifampicin, monitor for loss of effectiveness.
In vitro studies indicate that clindamycin does not inhibit CYP1A2, CYP2C9, CYP2C19, CYP2E1 or CYP2D6 and only moderately inhibits CYP3A4.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals have not been performed with clindamycin to evaluate carcinogenic potential. Genotoxicity tests performed included a rat micronucleus test and an Ames Salmonella reversion test. Both tests were negative.
Fertility studies in rats treated orally with up to 300 mg/kg/day (approximately 1.1 times the highest recommended adult human dose based on mg/m2) revealed no effects on fertility or mating ability.
Pregnancy: Teratogenic effects
In clinical trials with pregnant women, the systemic administration of clindamycin during the second and third trimesters, has not been associated with an increased frequency of congenital abnormalities.
Clindamycin should be used during the first trimester of pregnancy only if clearly needed. There are no adequate and well-controlled studies in pregnant women during the first trimester of pregnancy. Because animal reproduction studies are not always predictive of the human response, this drug should be used during pregnancy only if clearly needed.
Reproduction studies performed in rats and mice using oral doses of clindamycin up to 600 mg/kg/day (2.1 and 1.1 times the highest recommended adult human dose based on mg/m2, respectively) or subcutaneous doses of clindamycin up to 250 mg/kg/day (0.9 and 0.5 times the highest recommended adult human dose based on mg/m2, respectively) revealed no evidence of teratogenicity.
Clindamycin injection contains benzyl alcohol. Benzyl alcohol can cross the placenta. See WARNINGS.
-
Nursing Mothers
Limited published data based on breast milk sampling reports that clindamycin appears in human breast milk in the range of less than 0.5 to 3.8 mcg/mL at dosages of 150 mg orally to 600 mg intravenously. Clindamycin has the potential to cause adverse effects on the breastfed infant's gastrointestinal flora. If oral or intravenous clindamycin is required by a nursing mother, it is not a reason to discontinue breastfeeding, but an alternate drug may be preferred. Monitor the breastfed infant for possible adverse effects on the gastrointestinal flora, such as diarrhea, candidiasis (thrush, diaper rash) or rarely, blood in the stool indicating possible antibiotic-associated colitis.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for clindamycin and any potential adverse effects on the breastfed child from clindamycin or from the underlying maternal condition.
- Pediatric Use
-
Usage in Newborns and Infants
This product contains benzyl alcohol as a preservative. Benzyl alcohol has been associated with a fatal "Gasping Syndrome" in premature infants. See WARNINGS.
The potential for the toxic effect in the pediatric population from chemicals that may leach from the single dose premixed IV preparation in plastic has not been evaluated. See WARNINGS.
-
Geriatric Use
Clinical studies of clindamycin did not include sufficient numbers of patients age 65 and over to determine whether they respond differently from younger patients. However, other reported clinical experience indicates that antibiotic-associated colitis and diarrhea (due to Clostridium difficile) seen in association with most antibiotics occur more frequently in the elderly (>60 years) and may be more severe. These patients should be carefully monitored for the development of diarrhea.
Pharmacokinetic studies with clindamycin have shown no clinically important differences between young and elderly subjects with normal hepatic function and normal (age-adjusted) renal function after oral or intravenous administration.
-
ADVERSE REACTIONS
The following reactions have been reported with the use of clindamycin.
Infections and Infestations:Clostridium difficile colitis.
Gastrointestinal: Antibiotic-associated colitis (see WARNINGS), pseudomembranous colitis, abdominal pain, nausea, and vomiting. The onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment (see WARNINGS). An unpleasant or metallic taste has been reported after intravenous administration of the higher doses of clindamycin phosphate.
Hypersensitivity Reactions: Maculopapular rash and urticaria have been observed during drug therapy. Generalized mild to moderate morbilliform-like skin rashes are the most frequently reported of all adverse reactions.
Severe skin reactions such as Toxic Epidermal Necrolysis, some with fatal outcome, have been reported (see WARNINGS). Cases of Acute Generalized Exanthematous Pustulosis (AGEP), erythema multiforme, some resembling Stevens-Johnson syndrome, have been associated with clindamycin. Anaphylactic shock, anaphylactic reaction and hypersensitivity have also been reported (see WARNINGS).
Skin and Mucous Membranes: Pruritus, vaginitis, angioedema and rare instances of exfoliative dermatitis have been reported (see Hypersensitivity Reactions).
Liver: Jaundice and abnormalities in liver function tests have been observed during clindamycin therapy.
Renal: Although no direct relationship of clindamycin to renal damage has been established, renal dysfunction as evidenced by azotemia, oliguria, and/or proteinuria has been observed.
Hematopoietic: Transient neutropenia (leukopenia) and eosinophilia have been reported. Reports of agranulocytosis and thrombocytopenia have been made. No direct etiologic relationship to concurrent clindamycin therapy could be made in any of the foregoing.
Immune System: Drug reaction with eosinophilia and systemic symptoms (DRESS) cases have been reported.
Local Reactions: Injection site irritation, pain, induration and sterile abscess have been reported after intramuscular injection and thrombophlebitis after intravenous infusion. Reactions can be minimized or avoided by giving deep intramuscular injections and avoiding prolonged use of indwelling intravenous catheters.
Cardiovascular: Cardiopulmonary arrest and hypotension have been reported following too rapid intravenous administration (see DOSAGE AND ADMINISTRATION).
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals, Inc. at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
OVERDOSAGE
Significant mortality was observed in mice at an intravenous dose of 855 mg/kg and in rats at an oral or subcutaneous dose of approximately 2618 mg/kg. In the mice, convulsions and depression were observed.
Hemodialysis and peritoneal dialysis are not effective in removing clindamycin from the serum.
-
DOSAGE AND ADMINISTRATION
If diarrhea occurs during therapy, this antibiotic should be discontinued (see WARNING box).
Clindamycin injection IM administration should be used undiluted.
Clindamycin injection IV administration should be diluted (see Dilution for IV use and IV Infusion Rates below).
Adults: Parenteral (IM or IV Administration): Serious infections due to aerobic gram-positive cocci and the more susceptible anaerobes (NOT generally including Bacteroides fragilis, Peptococcus species and Clostridium species other than Clostridium perfringens):
- 600 mg to 1,200 mg per day in 2, 3 or 4 equal doses.
More severe infections, particularly those due to proven or suspected Bacteroides fragilis, Peptococcus species, or Clostridium species other than Clostridium perfringens:
- 1,200 mg to 2,700 mg per day in 2, 3 or 4 equal doses.
For more serious infections, these doses may have to be increased. In life-threatening situations due to either aerobes or anaerobes these doses may be increased. Doses of as much as 4,800 mg daily have been given intravenously to adults. See Dilution for IV use and IV Infusion Rates section below.
Single intramuscular injections of greater than 600 mg are not recommended.
Alternatively, drug may be administered in the form of a single rapid infusion of the first dose followed by continuous IV infusion as follows:
To maintain serum
clindamycin levelsRapid infusion rate Maintenance
infusion rateAbove 4 mcg per mL
Above 5 mcg per mL
Above 6 mcg per mL10 mg/min for 30 min
15 mg/min for 30 min
20 mg/min for 30 min0.75 mg/min
1 mg/min
1.25 mg/minNeonates (less than 1 month): 15 to 20 mg/kg/day in 3 to 4 equal doses. The lower dosage may be adequate for small prematures.
Pediatric patients 1 month of age to 16 years: Parenteral (IM or IV) Administration: 20 to 40 mg/kg/day in 3 or 4 equal doses. The higher doses would be used for more severe infections. Clindamycin should be dosed based on total body weight regardless of obesity. As an alternative to dosing on a body weight basis, pediatric patients may be dosed on the basis of square meters body surface: 350 mg/m2/day for serious infections and 450 mg/m2/day for more severe infections.
Parenteral therapy may be changed to oral clindamycin flavored granules (clindamycin palmitate hydrochloride) or clindamycin capsules (clindamycin hydrochloride) when the condition warrants and at the discretion of the physician.
In cases of β-hemolytic streptococcal infections, treatment should be continued for at least 10 days.
Dilution for IV use and IV Infusion Rates: The concentration of clindamycin in diluent for infusion should not exceed 18 mg per mL. Infusion rates should not exceed 30 mg per minute. The usual infusion dilutions and rates are as follows:
Dose Diluent Time 300 mg
600 mg
900 mg
1200 mg50 mL
50 mL
50 to 100 mL
100 mL10 min
20 min
30 min
40 minAdministration of more than 1200 mg in a single 1-hour infusion is not recommended.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Dilution and Compatibility: Physical and biological compatibility studies monitored for 24 hours at room temperature have demonstrated no inactivation or incompatibility with the use of clindamycin injection (clindamycin phosphate) in IV solutions containing sodium chloride, glucose, calcium or potassium, and solutions containing vitamin B complex in concentrations usually used clinically. No incompatibility has been demonstrated with the antibiotics cephalothin, kanamycin, gentamicin, penicillin or carbenicillin.
The following drugs are physically incompatible with clindamycin phosphate: ampicillin sodium, phenytoin sodium, barbiturates, aminophylline, calcium gluconate, and magnesium sulfate.
The compatibility and duration of stability of drug admixtures will vary depending on concentration and other conditions. For current information regarding compatibilities of clindamycin phosphate under specific conditions, please contact the Medical Affairs department toll-free at 1-866-625-1618.
Physico-Chemical Stability of Diluted Solutions of Clindamycin Injection
Room Temperature: 6, 9 and 12 mg per mL (equivalent to clindamycin base) in dextrose injection 5%, sodium chloride injection 0.9%, or Lactated Ringers Injection in glass bottles or Mini-Bag containers, demonstrated physical and chemical stability for at least 16 days at 25°C. Also, 18 mg per mL (equivalent to clindamycin base) in dextrose injection 5%, in Mini-Bag containers, demonstrated physical and chemical stability for at least 16 days at 25°C.
Refrigeration: 6, 9 and 12 mg per mL (equivalent to clindamycin base) in dextrose injection 5%, sodium chloride injection 0.9%, or Lactated Ringers Injection in glass bottles or Mini-Bag containers, demonstrated physical and chemical stability for at least 32 days at 4°C.
IMPORTANT: This chemical stability information in no way indicates that it would be acceptable practice to use this product well after the preparation time. Good professional practice suggests that compounded admixtures should be administered as soon after preparation as is feasible.
Frozen: 6, 9 and 12 mg per mL (equivalent to clindamycin base) in dextrose injection 5%, sodium chloride injection 0.9%, or Lactated Ringers Injection in Mini-Bag containers demonstrated physical and chemical stability for at least eight weeks at -10°C.
Frozen solutions should be thawed at room temperature and not refrozen.
-
HOW SUPPLIED
Each mL of Clindamycin Injection, USP contains clindamycin phosphate equivalent to 150 mg of clindamycin. Also contains 0.5 mg disodium edetate and 9.45 mg benzyl alcohol as a preservative. Sodium hydroxide and/or hydrochloric acid may be added to adjust pH.
Clindamycin Injection, USP is supplied as follows: NDC Clindamycin Injection, USP (150 mg per mL) Package Factor 25021-115-02 300 mg per 2 mL Single-Dose Vial 25 vials per carton 25021-115-04 600 mg per 4 mL Single-Dose Vial 25 vials per carton 25021-115-06 900 mg per 6 mL Single-Dose Vial 25 vials per carton Clindamycin Injection, USP Pharmacy Bulk Package is also available as follows: NDC Clindamycin Injection, USP (150 mg per mL) Package Factor 25021-115-51 9,000 mg per 60 mL Pharmacy Bulk Package Bottle 1 bottle per carton -
REFERENCES
- Smith RB, Phillips JP: Evaluation of CLEOCIN HCl and CLEOCIN Phosphate in an Aged Population. Upjohn TR 8147-82-9122-021, December 1982.
Brands listed are the trademarks of their respective owners.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2020 Sagent Pharmaceuticals, Inc.Revised: March 2020
SAGENT Pharmaceuticals®
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CLINDAMYCIN
clindamycin phosphate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:25021-115 Route of Administration INTRAVENOUS, INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength clindamycin phosphate (UNII: EH6D7113I8) (clindamycin - UNII:3U02EL437C) clindamycin 150 mg in 1 mL Inactive Ingredients Ingredient Name Strength edetate disodium (UNII: 7FLD91C86K) benzyl alcohol (UNII: LKG8494WBH) sodium hydroxide (UNII: 55X04QC32I) hydrochloric acid (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:25021-115-02 25 in 1 CARTON 05/15/2012 1 2 mL in 1 VIAL; Type 0: Not a Combination Product 2 NDC:25021-115-04 25 in 1 CARTON 05/15/2012 2 4 mL in 1 VIAL; Type 0: Not a Combination Product 3 NDC:25021-115-06 25 in 1 CARTON 05/15/2012 3 6 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090108 05/15/2012 Labeler - Sagent Pharmaceuticals (080579617)