Label: POLY-VI-FLOR- multivitamin and fluoride supplement suspension/ drops
- NHRIC Code(s): 23594-605-50
- Packager: Aytu Therapeutics LLC
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated September 15, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Poly-Vi-Flor
-
Health Claim
Multivitamin and Fluoride Supplement
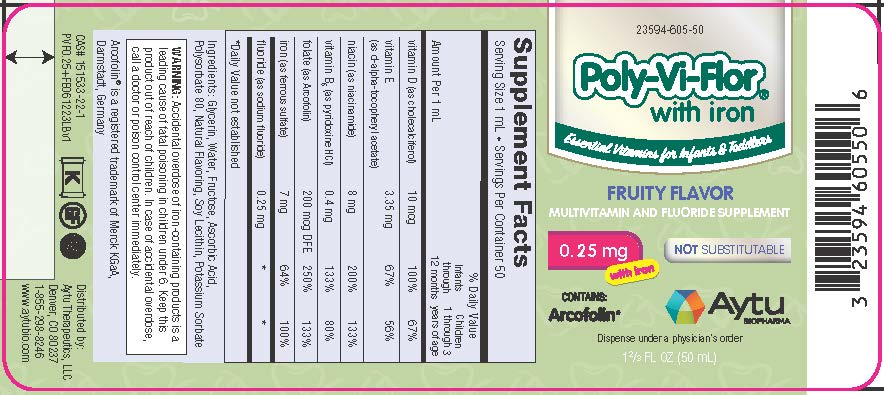
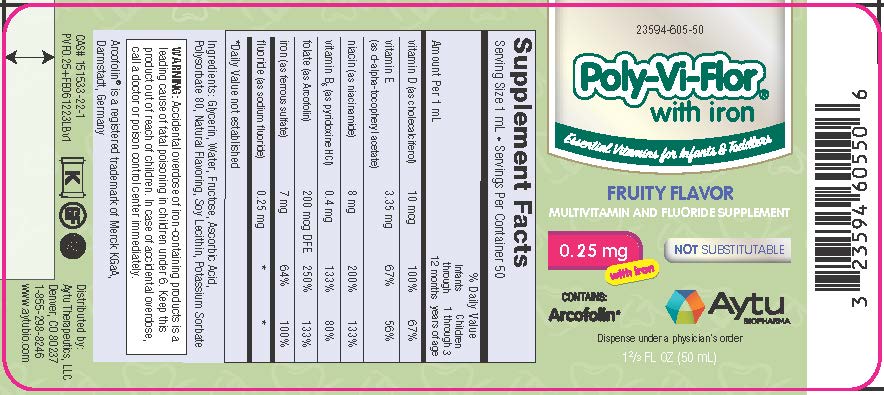
Supplement Facts
Serving Size 1 mL • Servings Per Container 50% Daily Value Amount Per 1 mL Infants
through
12 monthsChildren
1 through 3
years of agevitamin D (as cholecalciferol) 10 mcg 100% 67% vitamin E (as d-alpha-tocopheryl acetate) 3.35 mg 67% 56% niacin (as niacinamide) 8 mg 200% 133% vitamin B6 (as pyridoxine HCl) 0.4 mg 133% 80% folate (as Arcofolin) 200 mcg DFE 250% 133% iron (as ferrous sulfate) 7 mg 64% 100% fluoride (as sodium fluoride) 0.25 mg * * *Daily Value not established
Ingredients: Glycerin, Water, Fructose, Ascorbic Acid, Polysorbate 80, Natural Flavoring, Soy Lecithin, Potassium Sorbate
Poly-Vi-Flor drops are specially designed to help meet the nutritional needs of your growing infant or toddler.
- Poly-Vi-Flor Drops are the ideal vitamin supplement during your child's transition to solid foods, during growth spurts, or for the picky eater.
- One daily dose of Poly-Vi-Flor Drops provides 5 essential vitamins and is fruit flavored.
- Drops are dye-free and gluten-free.
-
Dosage and Administration
How to Use the Enclosed Dropper
Fill the dropper to the 1 mL line to provide the full daily dosage. Use full dosage, regardless of age or weight, unless your child's doctor recommends a different dosage.
Dispense with a single squeeze of the dropper bulb. It is normal for a small amount to remain in the tip.
Dosing Tips
Drops may be given by dispensing directly into the mouth. Drops may also be mixed with formula, fruit juice, cereal or other foods to increase your child's acceptance.
Shake well before use.
This product comes with a child-resistant cap. Refrigeration not required. After opening, store away from direct sunlight.
-
Warning
Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately.
Arcofolin® is a registered trademark of Merck KGaA, Darmstadt, Germany
Distributed by: Aytu Therapeutics, LLC
Denver, CO 80237
1-855-298-8246
www.aytubio.com - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
POLY-VI-FLOR
multivitamin and fluoride supplement suspension/ dropsProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:23594-605 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHOLECALCIFEROL (UNII: 1C6V77QF41) (CHOLECALCIFEROL - UNII:1C6V77QF41) CHOLECALCIFEROL 10 ug in 1 mL .ALPHA.-TOCOPHEROL ACETATE, D- (UNII: A7E6112E4N) (.ALPHA.-TOCOPHEROL, D- - UNII:N9PR3490H9) .ALPHA.-TOCOPHEROL, D- 3.35 mg in 1 mL NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 8 mg in 1 mL PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 0.4 mg in 1 mL LEVOMEFOLATE CALCIUM (UNII: A9R10K3F2F) (LEVOMEFOLIC ACID - UNII:8S95DH25XC) LEVOMEFOLIC ACID 200 ug in 1 mL SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.25 mg in 1 mL FERROUS SULFATE (UNII: 39R4TAN1VT) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 7 mg in 1 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) FRUCTOSE (UNII: 6YSS42VSEV) ASCORBIC ACID (UNII: PQ6CK8PD0R) POLYSORBATE 80 (UNII: 6OZP39ZG8H) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:23594-605-50 1 in 1 PACKAGE 1 50 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 09/15/2023 Labeler - Aytu Therapeutics LLC (117244733)