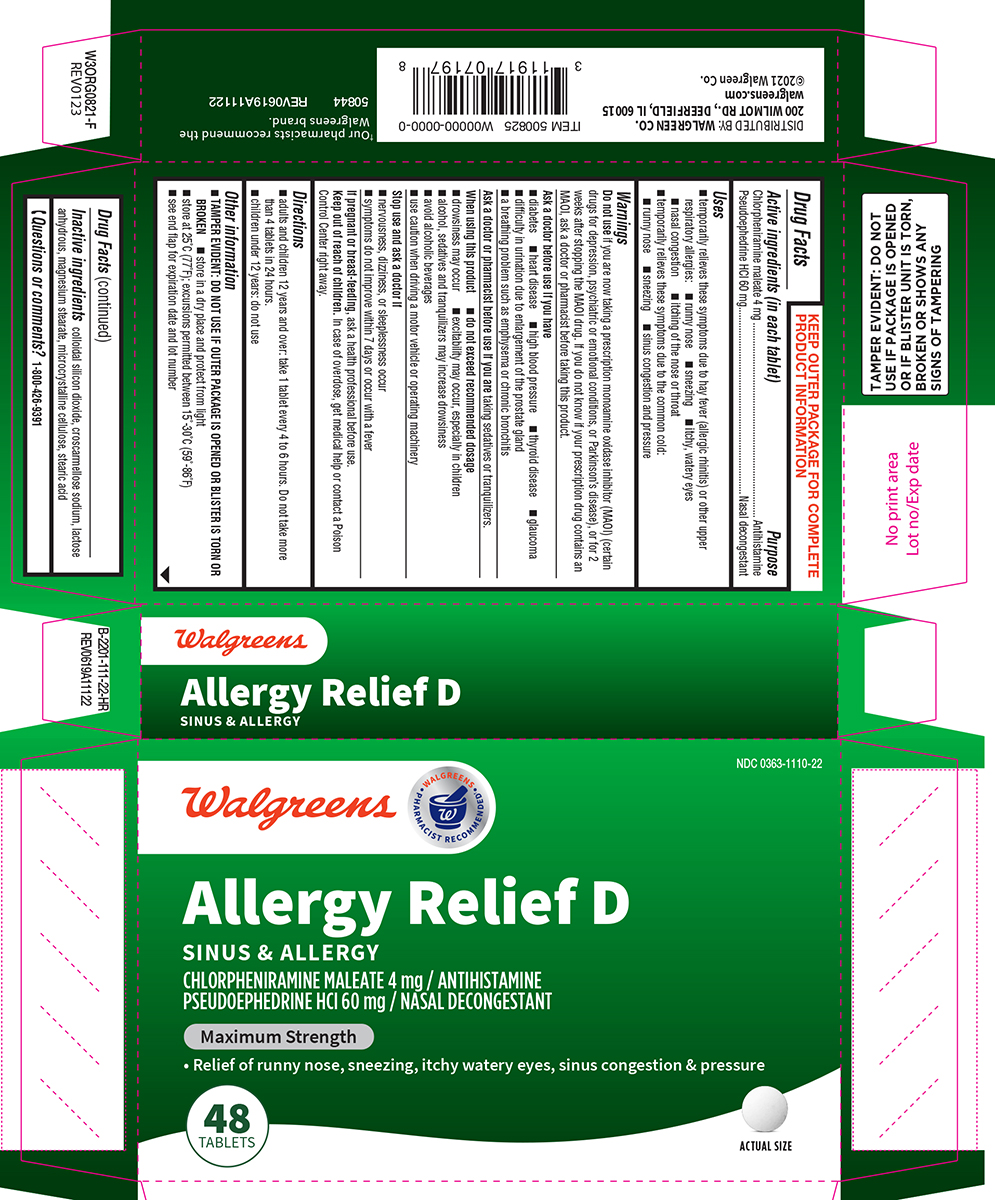
Label: ALLERGY RELIEF D- chlorpheniramine maleate, pseudoephedrine hcl tablet
- NDC Code(s): 0363-1110-22
- Packager: Walgreen Company
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated July 3, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each tablet)
- Purpose
-
Uses
- temporarily relieves these symptoms due to hay fever (allergic rhinitis) or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- nasal congestion
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
- sinus congestion and pressure
- temporarily relieves these symptoms due to hay fever (allergic rhinitis) or other upper respiratory allergies:
-
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- diabetes
- heart disease
- high blood pressure
- thyroid disease
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem such as emphysema or chronic bronchitis
When using this product
-
do not exceed recommended dosage
- drowsiness may occur
- excitability may occur, especially in children
- alcohol, sedatives and tranquilizers may increase drowsiness
- avoid alcoholic beverages
- use caution when driving a motor vehicle or operating machinery
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal display panel
NDC 0363-1110-22
Walgreens
• WALGREENS •
PHARMACIST RECOMMENDED†Allergy Relief D
SINUS & ALLERGY
CHLORPHENIRAMINE MALEATE 4 mg / ANTIHISTAMINE
PSEUDOEPHEDRINE HCI 60 mg / NASAL DECONGESTANTMaximum Strength
• Relief of runny nose, sneezing, itchy, watery eyes, sinus congestion & pressure
48
TABLETSACTUAL SIZE
TAMPER EVIDENT: DO NOT
USE IF PACKAGE IS OPENED
OR IF BLISTER UNIT IS TORN,
BROKEN OR SHOWS ANY
SIGNS OF TAMPERING†Our pharmacists recommend the Walgreens brand.
50844 REV0619A11122
DISTRIBUTED BY: WALGREEN CO.
200 WILMOT RD., DEERFIELD, IL 60015
walgreens.com
©2021 Walgreen Co.
Walgreens 44-111
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF D
chlorpheniramine maleate, pseudoephedrine hcl tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0363-1110 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 4 mg PSEUDOEPHEDRINE HYDROCHLORIDE (UNII: 6V9V2RYJ8N) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE HYDROCHLORIDE 60 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white Score 2 pieces Shape ROUND Size 7mm Flavor Imprint Code 44;111 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0363-1110-22 2 in 1 CARTON 08/24/2021 1 24 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 08/24/2021 Labeler - Walgreen Company (008965063) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(0363-1110) , pack(0363-1110) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(0363-1110)