Label: DIFFERIN- adapalene lotion
- NDC Code(s): 0299-5912-01, 0299-5912-02
- Packager: Galderma Laboratories, L.P.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated October 24, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DIFFERIN Lotion safely and effectively. See full prescribing information for DIFFERIN Lotion.
DIFFERIN ®(adapalene) lotion, for topical use Initial U.S. Approval: 1996INDICATIONS AND USAGE
DIFFERIN Lotion is a retinoid product indicated for the topical treatment of acne vulgaris in patients 12 years and older.. (1)
DOSAGE AND ADMINISTRATION
- Apply once daily, after washing gently with a mild soapless cleanser. ( 2)
- Dispense a nickel size amount of DIFFERIN Lotion (3-4 actuations of the pump) to cover the entire face and other affected areas of the skin. ( 2)
- Avoid application to the areas of skin around eyes, lips and mucous membranes. ( 2)
- For topical use only and not for oral, ophthalmic, or intravaginal use. ( 2)
DOSAGE FORMS AND STRENGTHS
Lotion, 0.1% in 2 oz bottle with pump. (3)
CONTRAINDICATIONS
Contraindicated in patients with known hypersensitivity to adapalene or any excipient of DIFFERIN Lotion. ( 4)
WARNINGS AND PRECAUTIONS
- Allergic/ Hypersensitivity Reactions:Allergy/hypersensitivity reactions include anaphylaxis, angioedema, urticaria, and pruritis. Discontinue DIFFERIN Lotion in the event of an allergic/hypersensitivity reaction. ( 5.1)
- Ultraviolet Light and Environmental Exposure:Avoid exposure to sunlight and sunlamps. Wear sunscreen when sun exposure cannot be avoided. ( 5.2)
- Local Cutaneous reactions:Erythema, scaling, dryness, and stinging/burning may occur with use of DIFFERIN Lotion. Avoid application of DIFFERIN Lotion to cuts, abrasions, eczematous or sunburned skin. Avoid concomitant use of other potentially irritating topical products (abrasive soaps and cleansers, soaps and cosmetics that have strong skin-drying effect and products with high concentrations of alcohol, astringents, spices, or limes). ( 5.3)
ADVERSE REACTIONS
The most common adverse reactions reported (≥ 1%) are dry skin and skin irritation. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact Galderma Laboratories, L.P. at 1-866-735-4137 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 4/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Allergic/ Hypersensitivity Reactions
5.2 Ultraviolet Light and Environmental Exposure
5.3 Local Cutaneous Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
Wash affected areas gently with a mild soapless cleanser.
Dispense a nickel size amount of DIFFERIN Lotion (3-4 actuations of the pump) and apply a thin film to the entire face and other affected areas of the skin once daily.
Avoid application to the areas of skin around eyes, lips and mucous membranes.
DIFFERIN Lotion is for topical use only and not for oral, ophthalmic, or intravaginal use. - 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
DIFFERIN Lotion is contraindicated in patients who have known hypersensitivity to adapalene or any excipient of DIFFERIN Lotion [ see WARNINGS AND PRECAUTIONS ( 5.1) ]
-
5 WARNINGS AND PRECAUTIONS
5.1 Allergic/ Hypersensitivity Reactions
Anaphylaxis, angioedema, urticaria, face edema, eyelid edema, lip swelling, and pruritus that sometimes required medical treatment have been reported during postmarketing use of adapalene. Advise a patient to stop using DIFFERIN Lotion and seek medical attention if experiencing allergic or anaphylactoid/anaphylactic reactions during treatment.
5.2 Ultraviolet Light and Environmental Exposure
Avoid exposure to sunlight, including sunlamps, during the use of DIFFERIN Lotion. Patients with high levels of sun exposure and those with inherent sensitivity to sun should be warned to exercise caution. Use of sunscreen products and protective apparel (e.g., hat) are recommended when exposure cannot be avoided. Weather extremes, such as wind or cold, may be irritating to patients under treatment with DIFFERIN Lotion..
5.3 Local Cutaneous Reactions
Signs and symptoms of local skin irritation (such as erythema, scaling, dryness, stinging/burning) may be experienced with use of DIFFERIN Lotion. These are most likely to occur during the first 2 weeks of treatment, are mostly mild to moderate in severity, and usually lessen with continued use of DIFFERIN Lotion. Depending upon the severity of these side effects, patients should be instructed to use a moisturizer, reduce the frequency of the application of DIFFERIN Lotion or discontinue use.
Avoid application of DIFFERIN Lotion to cuts, abrasions, eczematous or sunburned skin. As with other retinoids, use of “waxing” as a depilatory method should be avoided on skin treated with DIFFERIN Lotion. Avoid concomitant use of other potentially irritating topical products (abrasive soaps and cleansers, soaps and cosmetics that have strong skin-drying effect and products with high concentrations of alcohol, astringents, spices, or limes). -
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 2141 subjects with acne vulgaris, 12 years and older, were treated once daily for 12 weeks. Of these, 1068 were exposed to DIFFERIN Lotion during the clinical trials. A total of 1057 subjects completed at least one post treatment evaluation. Related adverse reactions that were reported in at least 1% of subjects treated with DIFFERIN Lotion or with the Vehicle Lotion are presented in Table 1. The majority of cases were transient, mild to moderate in severity and were managed with moisturizers.
Table 1: Adverse Reactions Reported in Clinical Trials by At Least 1% of Subjects System Organ Class/Preferred Term Adapalene Lotion 0.1%
N = 1068Vehicle Lotion
N = 1073Subjects with Related AR(s) 10.2% 4.6% Dry Skin 7.7% 3.0% Skin irritation 1.5% 0.7% Skin burning/skin discomfort 0.9% 0.0% Sunburn 0.6% 0.6% Local tolerability evaluations, presented in Table 2, were conducted at each study visit in clinical trials. Erythema, scaling, dryness, burning/stinging were assessed:
Table 2: Incidence of Local Cutaneous Irritation, for Subjects Whose Irritation Score was Higher than at Baseline, in Controlled Clinical Trials Adverse Reactions (DIFFERIN Lotion Group N = 1057*) Combined Trial 1 and Trial 2 Maximum Severity During Treatment
(N = 1057)Week 12 Treatment Severity
(N = 950)Local Cutaneous Irritation (skin irritation) Mild Moderate Severe Mild Moderate Severe Erythema 21.8% 8.0% 0.2% 7.9% 2.6% 0.2% Scaling 25.3% 6.5% 0.1% 5.3% 1.1% 0% Dryness 36.1% 7.3% 0.3% 7.6% 2.0% 0% Stinging/burning 22.1% 7.0% 0.9% 4.6% 1.0% 0.4% * Data from 11 subjects with missing data are not included
Local tolerability scores for erythema, scaling, dryness, burning/stinging rose during the first two weeks of treatment and generally decreased thereafter.
In an open label postmarketing pharmacokinetic trial of 13 adolescent subjects, the adverse reaction of pruritus was reported in 8 out of 13 subjects.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of adapalene:
Immune system disorders:angioedema, face edema, lip swelling
Skin disorders:application site pain
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from clinical trials with DIFFERIN Lotion use in pregnant women are insufficient to establish a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes. In animal reproduction studies, oral administration of adapalene to pregnant rats and rabbits during organogenesis at dose exposures 122 and 243 times, respectively, the human exposure at the maximum recommended human dose (MRHD) of 2 g resulted in fetal skeletal and visceral malformations ( see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies are 2 to 4% and 15 to 20%, respectively.Data
Animal Data
No malformations were observed in rats treated with oral adapalene doses of 0.15 to 5.0 mg/kg/day, up to 24 times the MRHD based on a mg/m2 comparison. However, malformations were observed in rats and rabbits when treated with oral doses of ≥ 25 mg/kg/day adapalene (122 and 243 times the MRHD, respectively, based on a mg/m 2comparison). Findings included cleft palate, microphthalmia, encephalocele, and skeletal abnormalities in rats and umbilical hernia, exophthalmos, and kidney and skeletal abnormalities in rabbits.
Dermal adapalene embryofetal development studies in rats and rabbits at doses up to 6.0 mg/kg/day (29 and 58 times the MRHD, respectively, based on a mg/m 2comparison) exhibited no fetotoxicity and only minimal increases in skeletal variations (supernumerary ribs in both species and delayed ossification in rabbits).8.2 Lactation
Risk Summary
There are no data on the presence of topical adapalene lotion or its metabolite in human milk, the effects on the breastfed infant, or the effects on milk production. In animal studies, adapalene is present in rat milk with oral administration of the drug. When a drug is present in animal milk, it is likely that the drug will be present in human milk. It is possible that topical administration of large amounts of adapalene could result in sufficient systemic absorption to produce detectable quantities in human milk ( see Clinical Considerations). The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for DIFFERIN Lotion and any potential adverse effects on the breastfed child from DIFFERIN Lotion or from the underlying maternal condition.Clinical Considerations
To minimize potential exposure to the breastfed infant via breastmilk, use DIFFERIN Lotion on the smallest area of skin and for the shortest duration possible while breastfeeding. Avoid application of DIFFERIN Lotion to areas with increased risk for potential ingestion by or ocular exposure to the breastfeeding child. -
11 DESCRIPTION
DIFFERIN (adapalene) Lotion, for topical use, contains adapalene in a white to off-white oil-in-water emulsion.
Adapalene is a naphthoic acid derivative with retinoid-like properties. The chemical name for adapalene is (6-[3-(1-adamantyl)-4methoxyphenyl]- 2-naphthoic acid). Adapalene has the following structural formula:
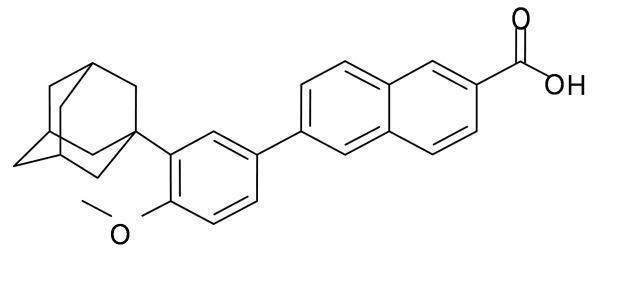
Adapalene:
Molecular formula: C 28H 28O 3 Molecular weight: 412.5
Each gram of DIFFERIN Lotion contains 1 mg of adapalene. The lotion also contains the following inactive ingredients: carbomer 941, disodium edetate, medium chain triglycerides, methylparaben, phenoxyethanol, poloxamer 124, polyoxyl-6-polyoxyl-32 palmitostearate, PPG12/SMDI copolymer, propylene glycol, propylparaben, purified water, sodium hydroxide, and stearyl alcohol. -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Adapalene binds to specific retinoic acid nuclear receptors but does not bind to cytosolic receptor protein. Biochemical and pharmacological profile studies have demonstrated that adapalene is a modulator of cellular differentiation, keratinization and inflammatory processes. However, the significance of these findings with regard to the mechanism of action of adapalene for the treatment of acne is unknown.
12.3 Pharmacokinetics
Systemic exposure of adapalene following a topical application of DIFFERIN Lotion was studied in two pharmacokinetic (PK) clinical trials. The first trial was conducted in 14 adult subjects 18 to 29 years of age with severe acne and the second trial was conducted in 13 adolescent subjects 12 to 17 years of age with moderate to severe acne.
In each trial, subjects were treated with 2 g of DIFFERIN Lotion applied once daily to approximately 1000 cm² of acne involved skin for 28 days (adolescent subjects) or 30 days (adult subjects). Serial plasma samples were collected at 24 or 72 hours following application on days 1, 15 and 28/30.
Daily topical application of DIFFERIN Lotion resulted in low systemic exposure to adapalene in the two populations (adult and adolescent subjects). In the adult population, all plasma concentrations in 12 out of 14 subjects were below the limit of quantification (LOQ=0.1 ng/mL). One subject had one sample above LOQ at day 30 and the other subject had four plasma samples above LOQ on both days 1 and 15, which ranged from 0.102 and 0.131 ng/mL.
In the adolescent population, plasma concentrations were quantifiable (>0.1 ng/mL) in five subjects. On Day 28, the mean C maxwas 0.128 ± 0.049 ng/mL (range: <0.100 to 0.244 ng/mL) and the mean of AUC 0-24hrwas 3.07 ± 1.21 ng.hr/mL (range: 1.86 to 4.93 ng.hr/mL). Adapalene plasma concentrations in all subjects were below the limit of quantification (<0.1 ng/mL) 48 hours after the last application on Day 28.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity, genotoxicity, or impairment of fertility studies were conducted with DIFFERIN Lotion.
Carcinogenicity studies with adapalene were conducted in mice at topical doses of 0.4, 1.3, and 4.0 mg/kg/day (1.2, 3.9, and 12 mg/m 2/day) and in rats at oral doses of 0.15, 0.5, and 1.5 mg/kg/day (0.9, 3.0, and 9.0 mg/m 2/day). The highest dose levels are 9.8 (mice) and 7.4 (rats) times the MRHD based on a mg/m 2comparison. In the rat study, an increased incidence of benign and malignant pheochromocytomas in the adrenal medulla of male rats was observed.
Adapalene was not mutagenic or genotoxic in vitro (Ames test, Chinese hamster ovary cell assay, or mouse lymphoma TK assay) or in vivo (mouse micronucleus test). In rat oral studies, 20 mg/kg/day adapalene (97 times the MRHD based on a mg/m 2comparison) did not affect the reproductive performance and fertility of F 0males and females or the growth, development, or reproductive function of F 1offspring.
-
14 CLINICAL STUDIES
The safety and efficacy of DIFFERIN Lotion applied once daily for the treatment of acne vulgaris were assessed in two 12-week, multicenter, controlled clinical trials of similar design, comparing DIFFERIN Lotion to the lotion vehicle in acne subjects.
In Trial 1, 1075 subjects were randomized to DIFFERIN Lotion or vehicle. The median age of these subjects was 16.7 years old and 53.1% were females. At baseline subjects had between 20 to 50 inflammatory lesions and 30 to 100 non-inflammatory lesions. The majority of subjects (91.0%) had a baseline IGA score of ‘Moderate’.
In Trial 2, 1066 subjects were randomized to DIFFERIN Lotion or vehicle. The median age of subjects was 16.7 years old and 53.7% were females. At baseline subjects had the same inclusion criteria as in Trial 1 and 95.7% of subjects had a baseline IGA score of ‘Moderate’. The outcome of the two trials is presented in Table 3.
Table 3: Clinical Trial Primary Efficacy Results at Week 12 Trial 1 DIFFERIN Lotion
(N = 533)Vehicle Lotion
(N = 542)IGA Success 140 (26.3%) 95 (17.3%) Total Lesions: Mean Absolute (Percent) Change 37.9 (51.5%) 26.7 (37.1%) Inflammatory Lesions: Mean Absolute (Percent) Change 14.7 (54.9%) 10.6 (40.3%) Non-inflammatory Lesions: Mean Absolute (Percent) Change 23.2 (49.6%) 16.1 (35.7%) Trial 2 DIFFERIN Lotion
(N = 535)Vehicle Lotion
(N = 531)IGA Success 129 (24.1%) 87 (16.4%) Total Lesions: Mean Absolute (Percent) Change 32.4 (44.6%) 23.4 (32.8%) Inflammatory Lesions: Mean Absolute (Percent) Change 12.7 (46.0%) 10.2 (36.9%) Non-inflammatory Lesions: Mean Absolute (Percent) Change 19.6 (43.1%) 13.1 (30.2%) -
16 HOW SUPPLIED/STORAGE AND HANDLING
DIFFERIN (adapalene) Lotion, 0.1%, is a white to off-white liquid packaged in a 2 oz (59 mL) bottle which is equipped with a lotion dispensing pump.
DIFFERIN Lotion is supplied as follows:
2 oz bottle pump NDC 0299-5912-02
Storage and handling
- Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [see USP Controlled Room Temperature]
- Do not freeze.
- Do not refrigerate.
- Protect from light.
- Keep out of reach of children.
- Keep away from heat.
- Keep bottle tightly closed.
- Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [see USP Controlled Room Temperature]
-
17 PATIENT COUNSELING INFORMATION
Patients using DIFFERIN Lotion should receive the following information and instructions:
- Wash affected areas gently with a mild soapless cleanser.
- Dispense a nickel size amount of DIFFERIN Lotion (3-4 actuations of the pump) and apply a thin film to the entire face and other affected areas of the skin once daily.
- Avoid application to the areas of skin around eyes, lips and mucous membranes.
- Do not apply to cuts, abrasions, eczematous, or sunburned skin.
- Wax depilation should not be performed on treated skin due to the potential for skin erosions.
- DIFFERIN Lotion may cause irritation such as erythema, scaling, dryness, stinging or burning. Minimize exposure to sunlight including sunlamps. Recommend the use of sunscreen products and protective apparel (e.g., hat) when exposure cannot be avoided.
- Moisturizers may be used if necessary; however, products containing alpha hydroxy or glycolic acids should be avoided.
- This product is for external use only.
- Contact the doctor if skin rash, pruritus, hives, chest pain, edema, and shortness of breath occurs, as these may be signs of allergy or hypersensitivity.
- Lactation: Use DIFFERIN Lotion on the smallest area of skin and for the shortest duration possible while breastfeeding. Advise breastfeeding women not to apply DIFFERIN Lotion to areas with increased risk for potential ingestion by or ocular exposure to the breastfeeding child. [See
Use in Specific Populations, Lactation (8.2)]
Marketed by:
GALDERMA LABORATORIES, L.P., Dallas, Texas 75201 USA
Made in Canada.
All trademarks are property of their respective owners.
P51503-X -
PATIENT MEDICATION INFORMATION SECTION
Patient Information
DIFFERIN® (Dif-er-in)
(adapalene)
LotionImportant:DIFFERIN Lotion is for use on the skin only (topical). Do not use DIFFERIN Lotion in or on your mouth, eyes, or vagina.
What is DIFFERIN Lotion?
DIFFERIN Lotion is a prescription medicine used on the skin (topical) to treat acne vulgaris in people 12 years of age and older.
It is not known if DIFFERIN Lotion is safe and effective in children under 12 years of age.Do not use DIFFERIN Lotion if youare allergic to adapalene or any of the ingredients in DIFFERIN Lotion. See the end of this Patient Information leaflet for a complete list of ingredients in DIFFERIN Lotion.
Before using DIFFERIN Lotion, tell your healthcare provider about all your medical conditions, including if you:
- have other skin problems, including cuts, abrasions, sunburn, or skin that is dry, itchy or red
- are pregnant or plan to become pregnant. It is not known if DIFFERIN Lotion can harm your unborn baby
- are breastfeeding or plan to breastfeed. It is not known if DIFFERIN Lotion passes into your breast milk and if it can harm your baby. Talk to your healthcare provider about the best way to feed your baby if you use DIFFERIN Lotion. If you use DIFFERIN Lotion while breastfeeding, use DIFFERIN Lotion on the smallest area of the skin and for the shortest time needed. Do not apply DIFFERIN Lotion to areas that may increase the risk of getting DIFFERIN Lotion in your child’s mouth or eyes.
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins and herbal supplements.
How should I use DIFFERIN Lotion?
- Use DIFFERIN Lotion exactly as your healthcare provider tells you to use it.
- Apply DIFFERIN Lotion 1 time a day.
Applying DIFFERIN Lotion:
- Wash the area where DIFFERIN Lotion will be applied gently with a mild soapless cleanser and pat dry.
- DIFFERIN Lotion comes in a pump. Dispense a nickel size amount of DIFFERIN Lotion (3 to 4 pumps) and apply a thin layer over the entire face and any other affected area of the skin. Do notapply DIFFERIN Lotion on areas of the skin around your eyes, lips, nose and mouth.
- Wash your hands after applying DIFFERIN Lotion.
What should I avoid while using DIFFERIN Lotion?
- Avoid spending time in sunlight, including sunlamps. DIFFERIN Lotion can make your skin sensitive to the sun and the light from sunlamps. Use sunscreen and wear a hat and clothes that cover the areas treated with DIFFERIN Lotion if you have to be in sunlight.
- Cold weather and wind may irritate your skin treated with DIFFERIN Lotion.
- Do notapply DIFFERIN Lotion to cuts, abrasions, sunburned skin, or skin that is dry, itchy or red.
- Avoid skin products that may dry or irritate your skin such as harsh soaps or cleansers, soaps and cosmetics that make your skin dry, and products that contain high levels of alcohol, astringents, spices, or limes.
- Avoid the use of “waxing” as a hair removal method on skin treated with DIFFERIN Lotion.
What are the possible side effects of DIFFERIN Lotion?
DIFFERIN Lotion may cause serious side effects, including:-
Allergic reactions. DIFFERIN Lotion may cause serious allergic reactions that sometimes may require medical treatment. Stop using DIFFERIN Lotion and tell your healthcare provider or get medical help right away if you have any of these symptoms of an allergic reaction:
- skin rash, itching or hives
- trouble breathing or chest pain
- swelling of your face, eyes, lips, tongue or throat
-
Skin reactions at the treated site.DIFFERIN Lotion may cause skin reactions including redness, scaling, dryness, stinging, or burning. These skin reactions are most likely to happen during the first 2 weeks of treatment, and usually lessen with continued use of DIFFERIN Lotion. Your healthcare provider may tell you to use a moisturizer, decrease how often you use DIFFERIN Lotion, or stop treatment with DIFFERIN Lotion if you get any skin reactions. If you use a moisturizer, you should avoid moisturizers that contain alpha hydroxy or glycolic acid. Ask your healthcare provider or pharmacist if you are not sure.
The most common side effects of DIFFERIN Lotion includedry skin and skin irritation.
These are not all the possible side effects of DIFFERIN Lotion.Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Galderma Laboratories, L.P. at 1-866-735-4137.How should I store DIFFERIN Lotion?
- Store DIFFERIN Lotion at room temperature between 68° F to 77° F (20° C to 25° C).
- Do notfreeze or refrigerate DIFFERIN Lotion.
- Keep DIFFERIN Lotion out of light and away from heat.
- Keep the DIFFERIN Lotion bottle tightly closed.
Keep DIFFERIN Lotion and all medicines out of the reach of children.
General information about the safe and effective use of DIFFERIN Lotion.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. Do not use DIFFERIN Lotion for a condition for which it was not prescribed. Do not give DIFFERIN Lotion to other people, even if they have the same symptoms you have. It may harm them. You can also ask your healthcare provider or pharmacist for information about DIFFERIN Lotion that is written for health professionals.What are the ingredients in DIFFERIN Lotion?
Active ingredient:adapalene
Inactive ingredients:carbomer 941, disodium edetate, medium chain triglycerides, methylparaben, phenoxyethanol, poloxamer 124, polyoxyl-6-polyoxyl-32 palmitostearate, PPG-12/SMDI copolymer, propylene glycol, propylparaben, purified water, sodium hydroxide, and stearyl alcohol.Marketed by: GALDERMA LABORATORIES, L.P., Dallas, Texas 75201 USA
Made in Canada.
GALDERMA is a registered trademark.
P51503-X
This Patient Information has been approved by the U.S. Food and Drug Administration.
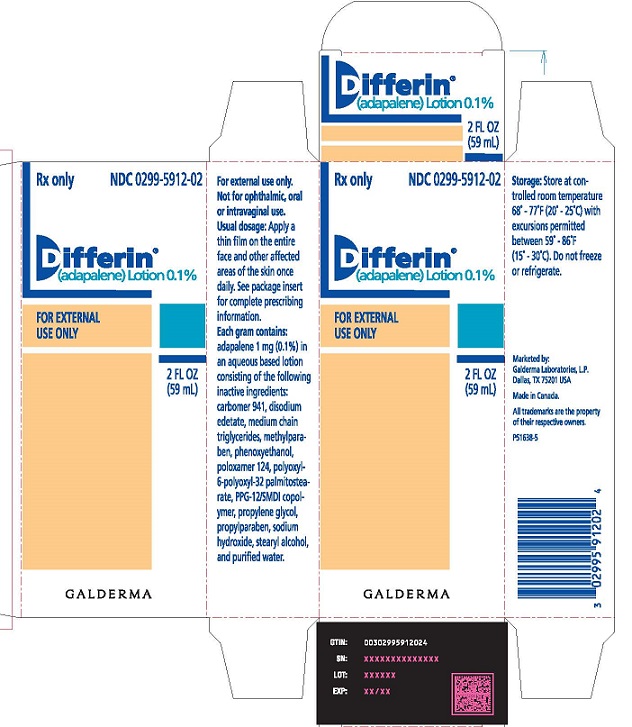
Issued: 04/2023 - PACKAGE LABEL - 2 FL OZ CARTON
-
PRINCIPAL DISPLAY PANEL
Rx Only
NDC0299-5912-02
Differin®
(adapalene) Lotion 0.1%
For External
Use Only
2 FL OZ
(59 mL)
GALDERMA
For external use only.
Not for ophthalmic, oral or intravaginal use.
Usual dosage:Apply a thin film on the entire face and other affected areas of the skin once daily. See package insert for complete prescribing information.
Each gram contains:adapalene 1 mg (0.1%) in an aqueous based lotion consisting of the following inactive ingredients:
carbomer 941, disodium edetate, medium chain triglycerides, methylparaben, phenoxyethanol, poloxamer 124, polyoxyl-6-polyoxyl-32 palmitostearate, PPG-12/SMDI copolymer, propylene glycol, propylparaben, sodium hydroxide, stearyl alcohol, and purified water.
Storage:Store at controlled room temperature 68° - 77°F (20° - 25°C) with excursions permitted between 59° - 86°F (15° - 30°C). Do not freeze or refrigerate.
Marketed by:
GALDERMA LABORATORIES, L.P.
Dallas, TX 75201
Made in Canada.
All trademarks are the property of their respective owners.
P51638-5 -
INGREDIENTS AND APPEARANCE
DIFFERIN
adapalene lotionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0299-5912 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ADAPALENE (UNII: 1L4806J2QF) (ADAPALENE - UNII:1L4806J2QF) ADAPALENE 0.1 g in 100 mL Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE A (UNII: F68VH75CJC) EDETATE DISODIUM (UNII: 7FLD91C86K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) METHYLPARABEN (UNII: A2I8C7HI9T) PHENOXYETHANOL (UNII: HIE492ZZ3T) POLOXAMER 124 (UNII: 1S66E28KXA) POLYETHYLENE GLYCOL 300 (UNII: 5655G9Y8AQ) POLYETHYLENE GLYCOL 1500 (UNII: 1212Z7S33A) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM HYDROXIDE (UNII: 55X04QC32I) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) WATER (UNII: 059QF0KO0R) Product Characteristics Color white (white to off-white) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0299-5912-02 1 in 1 CARTON 04/27/2010 12/31/2025 1 59 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 2 NDC:0299-5912-01 15 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/27/2010 12/31/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022502 03/31/2010 12/31/2025 Labeler - Galderma Laboratories, L.P. (047350186) Establishment Name Address ID/FEI Business Operations G Production Inc. 251676961 manufacture(0299-5912)