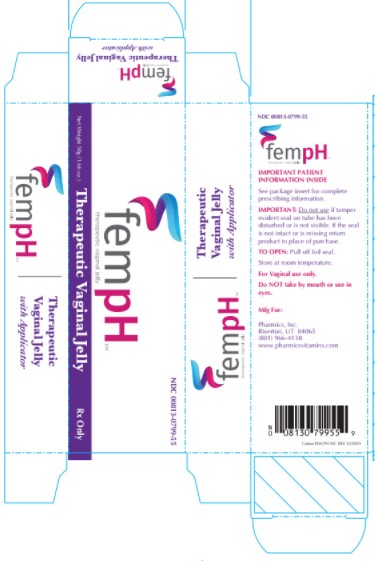
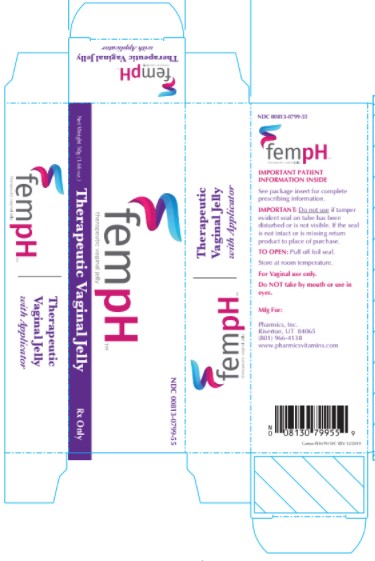
Label: FEM PH- acetic acid and oxyquinoline sulfate jelly
- NDC Code(s): 0813-0799-55
- Packager: Pharmics, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 18, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
Description
Fem pH Vaginal Jelly is a bland, non-irritating water dispersible, buffered acid jelly for intravaginal use. Fem pH is classified as a Vaginal Therapeutic Jelly. Fem pH contains 0.9% glacial acetic acid (C2H402) and 0.025% oxyquinoline sulfate (C18H16N206S) compounded with glycerin, lactic acid, poly ethylene glycol 4500 and purified water. Fem pH is formulated to pH 3.8-4.3 and is adjusted using 1 N potassium hydroxide.
- Clinical Pharmacology
- Indications and Usage
- Contraindications
- Warnings
-
Precautions
Drug Interactions
No incidence of drug interactions has been reported with concomitant use of Fem pH and any other medication.
Laboratory Tests
The monitoring of vaginal acidity (pH) may be helpful in following the patient's response. (The normal vaginal pH has been shown to be in the range of 4.0 to 5.0)
Carcinogenesis
No long-term studies in animals have been performed to evaluate carcinogenic potential.
- Adverse Reactions
-
Dosage and Administration
The usual dose is one applicator full, administered intra-vaginally, morning and evening. Duration of treatment may be determined by the patient's response to therapy. Each tube has a tamper evident seal at the opening of the tube. Replace cap after each use. To fill applicator screw applicator clockwise onto the tube. Squeeze tube forcing Fem pH jelly into barrel until it is full. Then unscrew applicator counter-clockwise to remove from tube. Lie on your back with knees drawn up. Hold filled applicator by the barrel and gently insert it into the vagina as far as it will comfortably go. Press plunger to empty the contents. Keep the plunger depressed and remove the applicator from vagina. After each use pull applicator apart and wash with warm soapy water, rinse well, dry and reassemble.
- How Supplied
-
Therapeutic Vaginal Jelly
Directions for using Fem pH applicator.
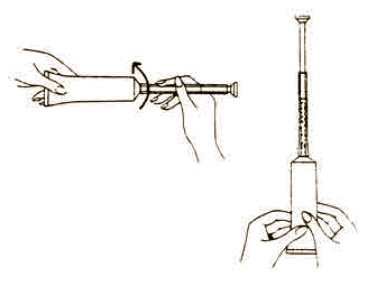
Each tube has a tamper evident seal at the opening of the tube. Replace cap after each use.
To fill applicator screw applicator clockwise onto the tube. Squeeze tube forcing Fem pH jelly into barrel until it is full. Then unscrew applicator counter-clockwise to remove from tube.
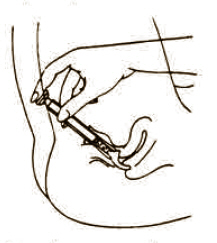
Lie on your back with knees drawn up. Hold filled applicator by the barrel and gently insert it into the vagina as far as it will comfortably go. Press plunger to empty the contents. Keep the plunger depressed and remove the applicator from vagina.
After each use pull applicator apart and wash with warm soapy water, rinse well, dry and reassemble.

- PRINCIPAL DISPLAY PANEL - 50g Tube Box
-
INGREDIENTS AND APPEARANCE
FEM PH
acetic acid and oxyquinoline sulfate jellyProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0813-0799 Route of Administration VAGINAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETIC ACID (UNII: Q40Q9N063P) (ACETIC ACID - UNII:Q40Q9N063P) ACETIC ACID 0.009 g in 1 g OXYQUINOLINE SULFATE (UNII: 61VUG75Y3P) (OXYQUINOLINE - UNII:5UTX5635HP) OXYQUINOLINE 0.00025 g in 1 g Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) LACTIC ACID (UNII: 33X04XA5AT) POLYETHYLENE GLYCOL 4500 (UNII: TVH7653921) WATER (UNII: 059QF0KO0R) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0813-0799-55 50 g in 1 TUBE; Type 0: Not a Combination Product 07/15/1999 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 07/15/1999 Labeler - Pharmics, Inc. (058560996) Registrant - Womens Choice (833067841)