Label: DIPHENHYDRAMINE- diphenhydramine hydrochloride solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 76413-333-01 - Packager: Central Texas Community Health Centers
- This is a repackaged label.
- Source NDC Code(s): 49348-045
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 28, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each 5 mL)
- Purpose
- Uses
-
Warnings
Do not use
- •
- with any other product containing diphenhydramine, even one used on skin
- •
- to make a child sleepy
Ask a doctor before use if the child has
- •
- a breathing problem such as chronic bronchitis
- •
- glaucoma
- •
- a sodium-restricted diet
-
Directions
- •
- find right dose on chart below
- •
- mL = milliliter
- •
- take every 4 to 6 hours, or as directed by a doctor
- •
- do not take more than 6 doses in 24 hours
Age (yr)
Dose (mL)
children under 2 years
do not use
children 2 to 5 years
do not use unless directed by a doctor
children 6 to 11 years
5 mL to 10 mL
Attention: use only enclosed dosing cup specifically designed for use with this product. Do not use any other dosing device.
- Other information
- Inactive ingredients
- Questions or comments?
-
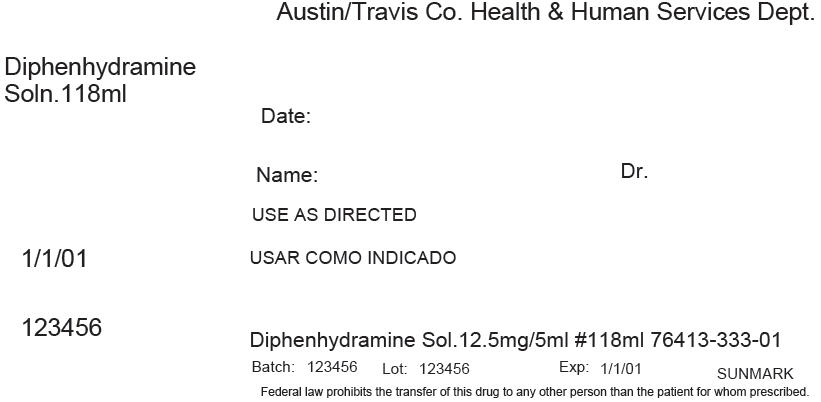
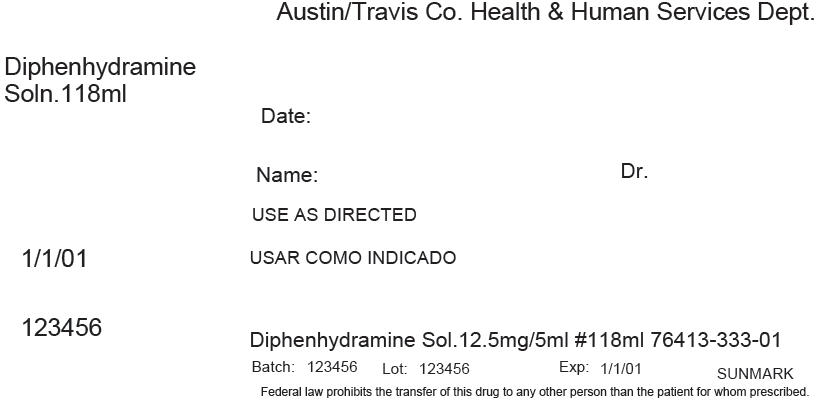
PRINCIPAL DISPLAY PANEL - 118 ml Bottle Label
Austin/Travis Co. Health & Human Services Dept.
Diphenhydramine
Soln.118mlDate:
Name:
Dr.USE AS DIRECTED
1/1/01
123456
Diphenhydramine Sol.12.5mg/5ml #118ml 76413-333-01
Batch: 123456
Lot: 123456
Exp: 1/1/01
SUNMARKFederal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
-
INGREDIENTS AND APPEARANCE
DIPHENHYDRAMINE
diphenhydramine hydrochloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76413-333(NDC:49348-045) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 12.5 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) D&C RED NO. 33 (UNII: 9DBA0SBB0L) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) POLOXAMER 407 (UNII: TUF2IVW3M2) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SORBITOL (UNII: 506T60A25R) Product Characteristics Color RED (Bluish-Red) Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76413-333-01 118 mL in 1 BOTTLE; Type 0: Not a Combination Product 08/21/2003 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 08/21/2003 Labeler - Central Texas Community Health Centers (079674019) Establishment Name Address ID/FEI Business Operations Central Texas Community Health Centers 079674019 REPACK(76413-333) , RELABEL(76413-333)