Label: CVS HEALTH STOOL SOFTENER- docusate sodium capsule
- NDC Code(s): 69842-523-01
- Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 6, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each softgel)
- Purpose
- Uses
- Warnings
- ASK DOCTOR/PHARMACIST
- DO NOT USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- QUESTIONS
- WARNINGS
-
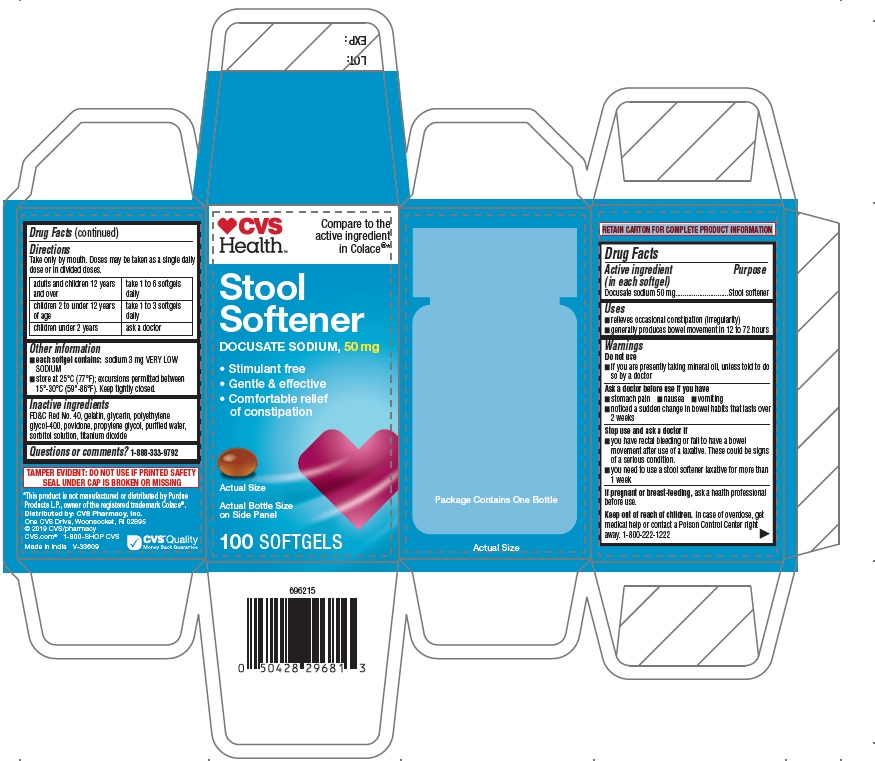
PRINCIPAL DISPLAY PANEL
CVS Health
Compare to the
active ingredient
in ColaceStool
Softener
DOCUSATE SODIUM, 50 mg
• Stimulant free
• Gentle & effective
• Comfortable relief
of constipationActual Bottle Size
on Side Pane100 SOFTGELS
RETAIN CARTON FOR COMPLETE PRODUCT INFORMATION
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY
SEAL UNDER CAP IS BROKEN OR MISSING*This product is not manufactured or distributed by Purdue
Products L.P., owner of the registered trademark Colace®.Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2019 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
Made in India V-33609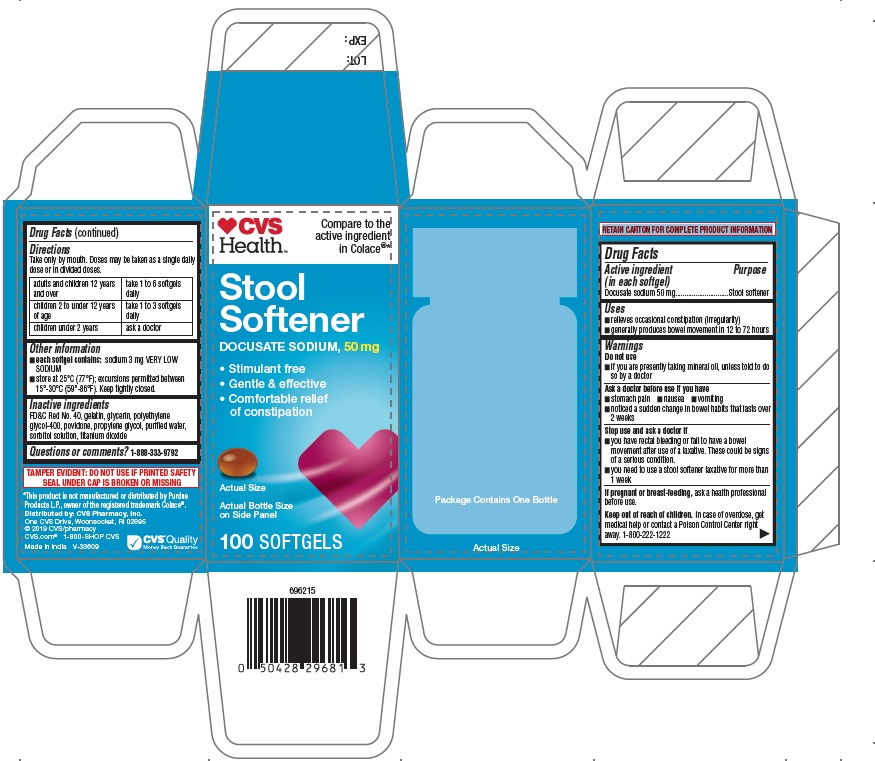
-
INGREDIENTS AND APPEARANCE
CVS HEALTH STOOL SOFTENER
docusate sodium capsuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-523 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 50 mg Inactive Ingredients Ingredient Name Strength FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POVIDONE K30 (UNII: U725QWY32X) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color red Score no score Shape OVAL Size 10mm Flavor Imprint Code S10 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-523-01 1 in 1 CARTON 06/05/2019 1 100 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 06/05/2019 Labeler - CVS Pharmacy (062312574)