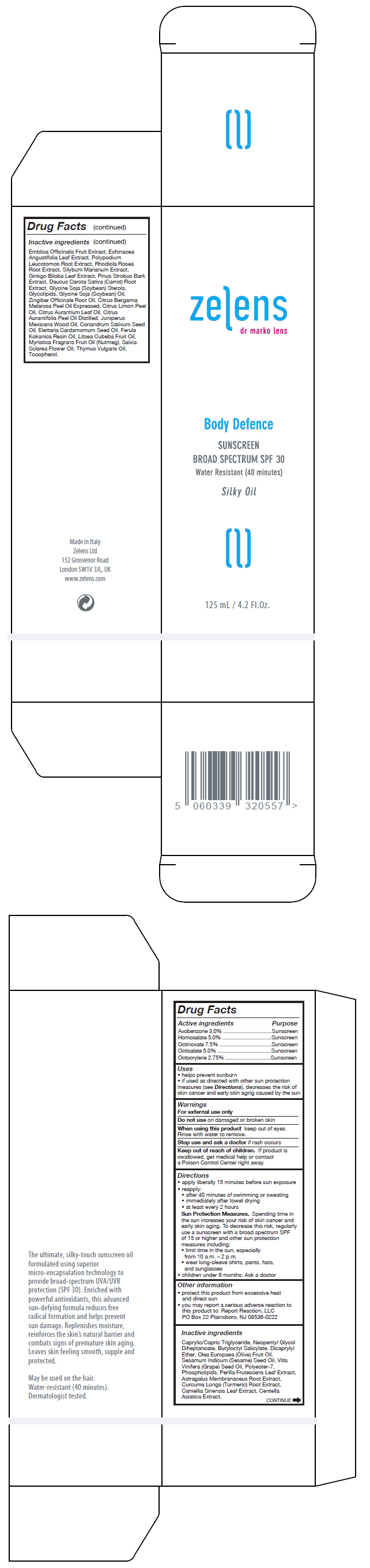
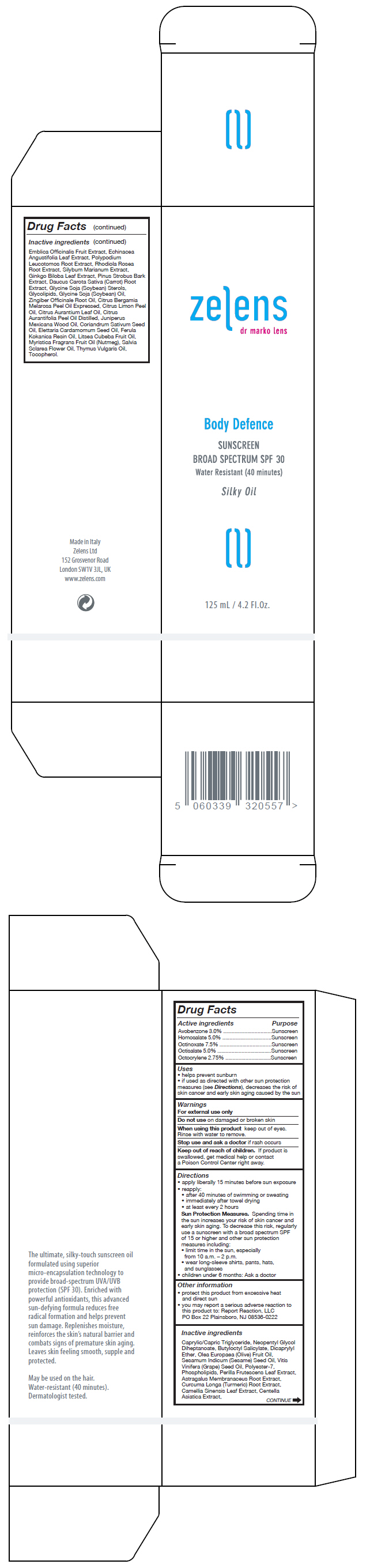
Label: BODY DEFENCE SUNSCREEN BROAD SPECTRUM SPF30- octinoxate, octisalate, homosalate, avobenzone, and octocrylene oil
-
Contains inactivated NDC Code(s)
NDC Code(s): 63712-201-01 - Packager: ZELENS Limited
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 5, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
- apply liberally 15 minutes before sun exposure
- reapply:
- after 40 minutes of swimming or sweating
- immediately after towel drying
- at least every 2 hours
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
- Other information
-
Inactive ingredients
Caprylic/Capric Triglyceride, Neopentyl Glycol Diheptanoate, Butyloctyl Salicylate, Dicaprylyl Ether, Olea Europaea (Olive) Fruit Oil, Sesamum Indicum (Sesame) Seed Oil, Vitis Vinifera (Grape) Seed Oil, Polyester-7, Phospholipids, Perilla Frutescens Leaf Extract, Astragalus Membranaceus Root Extract, Curcuma Longa (Turmeric) Root Extract, Camellia Sinensis Leaf Extract, Centella Asiatica Extract, Emblica Officinalis Fruit Extract, Echinacea Angustifolia Leaf Extract, Polypodium Leucotomos Root Extract, Rhodiola Rosea Root Extract, Silybum Marianum Extract, Ginkgo Biloba Leaf Extract, Pinus Strobus Bark Extract, Daucus Carota Sativa (Carrot) Root Extract, Glycine Soja (Soybean) Sterols, Glycolipids, Glycine Soja (Soybean) Oil, Zingiber Officinale Root Oil, Citrus Bergamia Melarosa Peel Oil Expressed, Citrus Limon Peel Oil, Citrus Aurantium Leaf Oil, Citrus Aurantifolia Peel Oil Distilled, Juniperus Mexicana Wood Oil, Coriandrum Sativum Seed Oil, Elettaria Cardamomum Seed Oil, Ferula Kokanica Resin Oil, Litsea Cubeba Fruit Oil, Myristica Fragrans Fruit Oil (Nutmeg), Salvia Sclarea Flower Oil, Thymus Vulgaris Oil, Tocopherol.
- PRINCIPAL DISPLAY PANEL - 125 mL Bottle Box
-
INGREDIENTS AND APPEARANCE
BODY DEFENCE SUNSCREEN BROAD SPECTRUM SPF30
octinoxate, octisalate, homosalate, avobenzone, and octocrylene oilProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63712-201 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 50 mg in 1 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 27.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) DICAPRYLYL ETHER (UNII: 77JZM5516Z) OLIVE OIL (UNII: 6UYK2W1W1E) SESAME OIL (UNII: QX10HYY4QV) GRAPE SEED OIL (UNII: 930MLC8XGG) POLYESTER-7 (UNII: 0841698D2F) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) PERILLA FRUTESCENS LEAF (UNII: T4L5881Y68) ASTRAGALUS PROPINQUUS ROOT (UNII: 922OP8YUPF) TURMERIC (UNII: 856YO1Z64F) GREEN TEA LEAF (UNII: W2ZU1RY8B0) CENTELLA ASIATICA (UNII: 7M867G6T1U) ECHINACEA ANGUSTIFOLIA LEAF (UNII: FS7G8S6PJ8) GINKGO (UNII: 19FUJ2C58T) PINUS STROBUS BARK (UNII: 8PJ4761097) PHYLLANTHUS EMBLICA FRUIT (UNII: YLX4CW2576) PHLEBODIUM AUREUM ROOT (UNII: VPS45WBU8U) SEDUM ROSEUM ROOT (UNII: 3S5ITS5ULN) MILK THISTLE (UNII: U946SH95EE) CARROT (UNII: L56Z1JK48B) SOY STEROL (UNII: PL360EPO9J) SOYBEAN OIL (UNII: 241ATL177A) GINGER (UNII: C5529G5JPQ) BERGAMOT OIL (UNII: 39W1PKE3JI) LEMON OIL (UNII: I9GRO824LL) BITTER ORANGE OIL (UNII: 9TLV70SV6I) LIME OIL (UNII: UZH29XGA8G) CEDRUS ATLANTICA BARK OIL (UNII: ZX5QRE4U60) CORIANDER OIL (UNII: 7626GC95E5) CARDAMOM OIL (UNII: JM0KJ091HZ) GALBANUM OIL (UNII: 211UF7M8N1) LITSEA OIL (UNII: 2XIW34BN6O) NUTMEG OIL (UNII: Z1CLM48948) CLARY SAGE OIL (UNII: 87L0D4U3M0) THYME OIL (UNII: 2UK410MY6B) TOCOPHEROL (UNII: R0ZB2556P8) Product Characteristics Color yellow Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63712-201-01 1 in 1 BOX 05/01/2016 1 125 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 05/01/2016 Labeler - ZELENS Limited (217149018) Establishment Name Address ID/FEI Business Operations Originitalia 441072253 manufacture(63712-201)