Label: TRISEPTIN HAND AND BODY ANTISEPTIC- alcohol lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 0064-1050-04 - Packager: HEALTHPOINT, LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 4, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- PURPOSE
- Uses
-
Warnings
- Flammable, keep away from fire or flame.
- For external use only.
- Do not use
- if you are allergic to or have a known or suspected hypersensitivity to any ingredient in this product.
- on or near the eyes, mucous membranes, genital skin, or mouth. If product gets into the eyes, flush immediately with water.
- Avoid contact with broken skin.
- Stop use and ask doctor if irritation and redness develop. If condition persists for more than 72 hours consult a physician.
- Keep out of reach of children.
- If swallowed, get medical help or contact a Poison Control Center right away.
-
Directions
Surgical Hand Scrub:
- Clean under nails with a nail pick. Nails should be maintained with a 1 mm free edge.
- Wet hands and forearms.
- Dispense palmful (approx. 7 ml) of TRISEPTIN Scrub into the palm of one hand.
- Twist fingertips of the opposite hand, working TRISEPTIN under the nails.
- Transfer TRISEPTIN to the opposite hand and repeat with fingertips of the other hand.
- Rub TRISEPTIN over the hands and up the forearms to just past the elbow, paying particular attention to the nails, cuticles and interdigital spaces for 90 seconds.
- Rinse and repeat the procedure to just below the elbows. Rinse completely.
Patient Preoperative Skin Preparation:
- Apply to cleanse the surgical site and surrounding area 12 hours and 6 hours prior to surgery.
- Clean the surgical area.
- Place a palmful (approx. 7 ml) of TRISEPTIN to the surgical site and surrounding area and rub for 90 seconds paying particular attention to the surgical area.
- Rinse and repeat.
- Rinse completely.
-
Inactive ingredients
Benzethonium chloride, citric acid, cocamine oxide, fragrance, glyceryl laurate, hydroxypropylcellulose, phenylethyl alcohol, phosphoric acid, polyoxyl 40 stearate, propyl gallate, propylene glycol, purified water, zinc pyrithione.
Questions?
Please call 1-800-523-0502 (M-F 8 a.m.-5 p.m. CST) or visit our web site www.triseptin.com.
Marketed by:
CareFusion
Leawood, KS 66211Manufactured for:
Healthpoint, Ltd.
Fort Worth, TX 76107
by DPT Laboratories, Ltd.
San Antonio, Texas 78215REORDER NO. 0064-1050-04
PATENT NO. 6,110,9084 fl oz (118 ml)
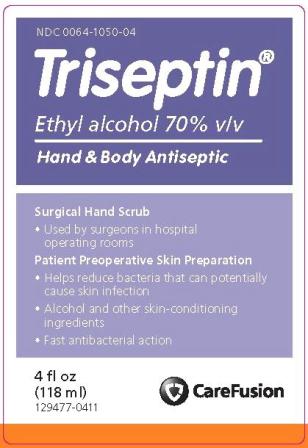
129478-0411PRINCIPAL DISPLAY PANEL
NDC 0064-1050-04
Triseptin®
Ethyl alcohol 70% v/v
Hand & Body AntisepticSurgical Hand Scrub
- Used by surgeons in hospital operating rooms
Patient Preoperative Skin Preparation
- Helps reduce bacteria that can potentially cause skin infection
- Alcohol and other skin-conditioning ingredients
- Fast antibacterial action
4 fl oz
(118 ml)
129477-0411CareFusion
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TRISEPTIN HAND AND BODY ANTISEPTIC
alcohol lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0064-1050 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL .7 mL in 1 mL Inactive Ingredients Ingredient Name Strength BENZETHONIUM CHLORIDE (UNII: PH41D05744) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) COCAMINE OXIDE (UNII: QWA2IZI6FI) GLYCERYL LAURATE (UNII: Y98611C087) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) PHENYLETHYL ALCOHOL (UNII: ML9LGA7468) PHOSPHORIC ACID (UNII: E4GA8884NN) POLYOXYL 40 STEARATE (UNII: 13A4J4NH9I) PROPYL GALLATE (UNII: 8D4SNN7V92) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) PYRITHIONE ZINC (UNII: R953O2RHZ5) Product Characteristics Color WHITE (white to off-white) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0064-1050-04 118 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 04/01/2002 Labeler - HEALTHPOINT, LTD (965634504) Establishment Name Address ID/FEI Business Operations DPT LABORATORIES, LTD 832224526 MANUFACTURE