Label: FOOT WORKS HEALTHY ANTIFUNGAL- tolnaftate spray
-
Contains inactivated NDC Code(s)
NDC Code(s): 10096-0272-1 - Packager: New Avon LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 1, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
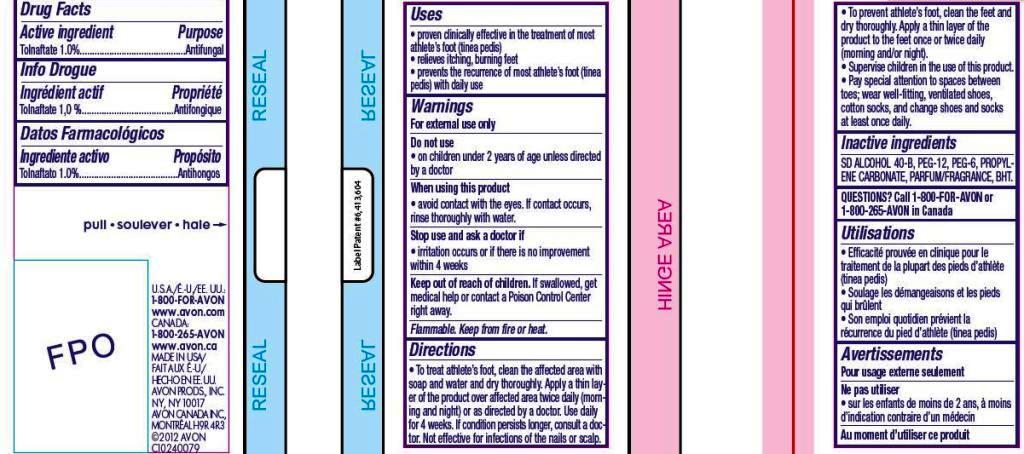
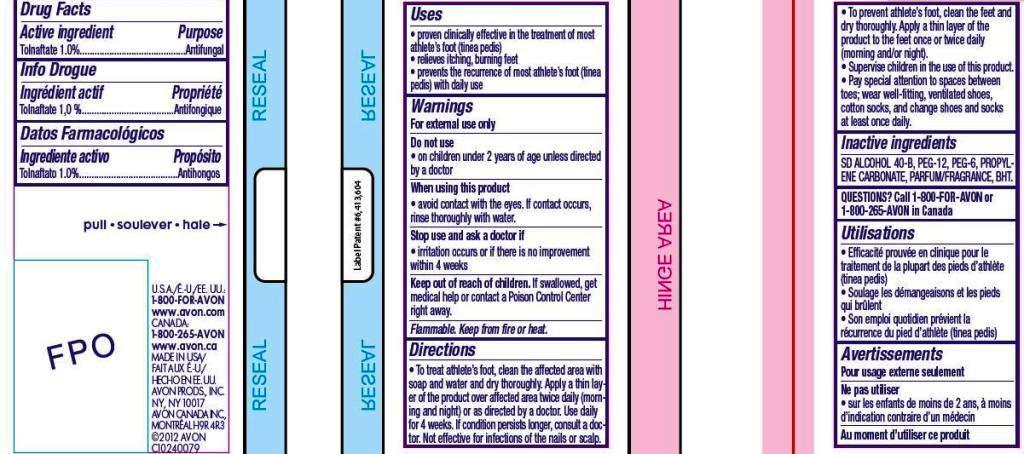
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
-
DOSAGE & ADMINISTRATION
Directions
• To treat athlete’s foot, clean the affected area with soap and water and dry thoroughly. Apply a thin layer of the product over affected area twice daily (morning and night) or as directed by a doctor. Use daily for 4 weeks. If condition persists longer, consult a doctor. Not effective for infections of the nails or scalp.• To prevent athlete’s foot, clean the feet and dry thoroughly. Apply a thin layer of the product to the feet once or twice daily (morning and/or night).
• Supervise children in the use of this product.
• Pay special attention to spaces between toes; wear well-fitting, ventilated shoes, cotton socks, and change shoes and socks at least once daily. - INACTIVE INGREDIENT
- QUESTIONS
- WARNINGS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FOOT WORKS HEALTHY ANTIFUNGAL
tolnaftate sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10096-0272 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOLNAFTATE (UNII: 06KB629TKV) (TOLNAFTATE - UNII:06KB629TKV) TOLNAFTATE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10096-0272-1 100 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 01/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333C 12/19/2011 Labeler - New Avon LLC (080143520)