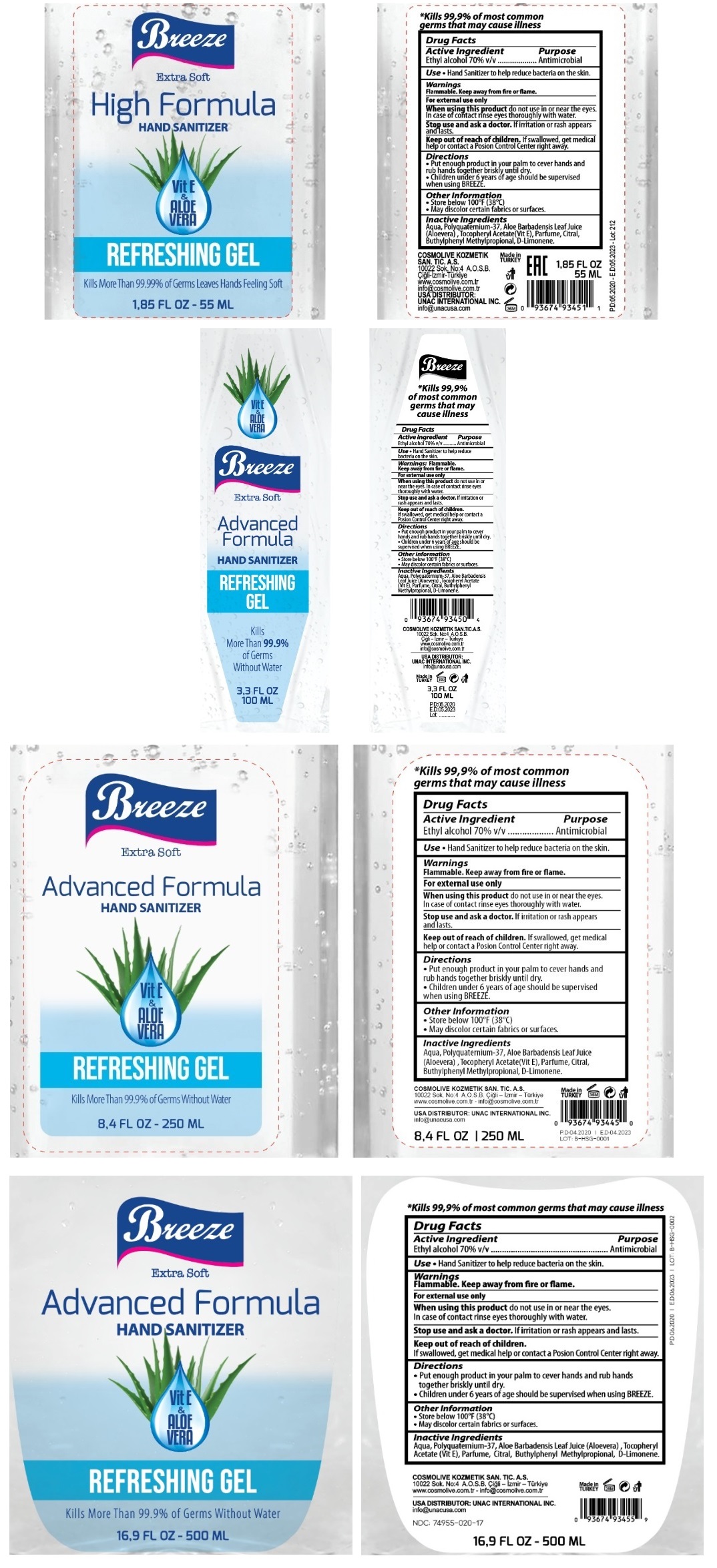
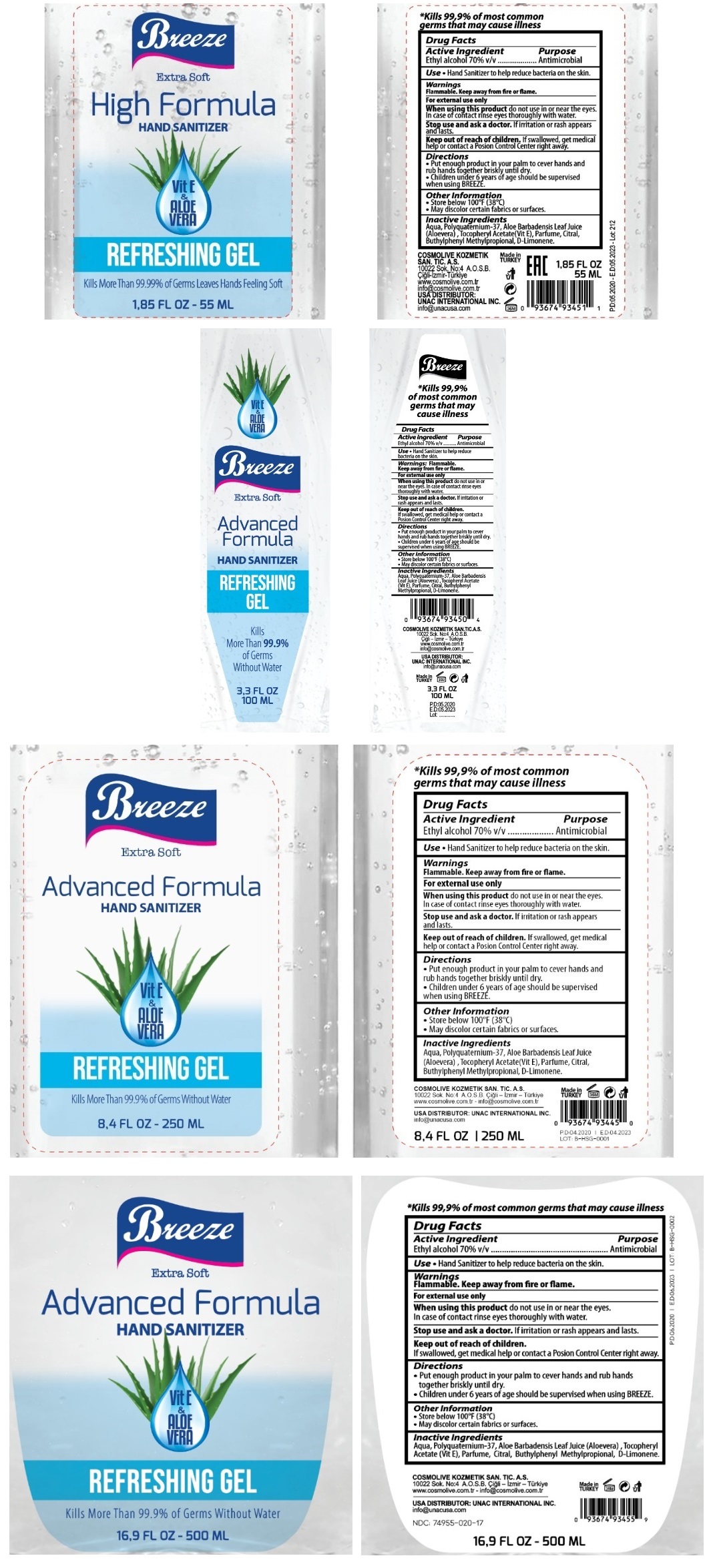
Label: BREEZE HAND SANITIZER WITH VITAMIN E AND ALOE VERA- ethyl alcohol gel
- NDC Code(s): 74955-020-17, 74955-020-18, 74955-020-33, 74955-020-84
- Packager: COSMOLIVE KOZMETIK SANAYI VE TICARET ANONIM SIRKETI
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 5, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient
- Purpose
- INDICATIONS & USAGE
- Warnings
- Directions
- Other Information
- Inactive ingredients
-
SPL UNCLASSIFIED SECTION
Extra Soft
Vit E & ALOE VERA
REFRESHING GEL
Kills More than 99.9% of Germs Without Water
*Kills 99.9% of most common germs that may cause illness
COSMOLIVE KOZMETIK SAN. TIC. A. S.
10022 Sok No: 4 A.O.S.B. Cigli- Izmir -Turkiye
www.cosmolive.com.tr - info@cosmolive.cpm.tr
USA DISTRIBUTOR: UNAC INTERNATIONAL INC.
info@unacusa.com
Made in TURKEY
- Packaging
-
INGREDIENTS AND APPEARANCE
BREEZE HAND SANITIZER WITH VITAMIN E AND ALOE VERA
ethyl alcohol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:74955-020 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 70 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) POLYQUATERNIUM-37 (25000 MPA.S) (UNII: 31L31U8285) ALOE VERA LEAF (UNII: ZY81Z83H0X) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) SULFURYL FLUORIDE (UNII: 64B59K7U6Q) CITRAL (UNII: T7EU0O9VPP) BUTYLPHENYL METHYLPROPIONAL (UNII: T7540GJV69) LIMONENE, (+)- (UNII: GFD7C86Q1W) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:74955-020-17 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 06/01/2020 2 NDC:74955-020-84 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/10/2020 3 NDC:74955-020-33 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/14/2020 4 NDC:74955-020-18 55 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/14/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 04/10/2020 Labeler - COSMOLIVE KOZMETIK SANAYI VE TICARET ANONIM SIRKETI (595671824) Establishment Name Address ID/FEI Business Operations COSMOLIVE KOZMETIK SANAYI VE TICARET ANONIM SIRKETI 595671824 manufacture(74955-020)