Label: CREST 3D WHITE BRILLIANCE BLAST- sodium fluoride paste, dentifrice
-
NDC Code(s):
37000-916-35,
37000-916-36,
37000-916-39,
37000-916-41, view more37000-916-82
- Packager: The Procter & Gamble Manufacturing Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
- Warning
-
Directions
- adults and children 2 yrs. & older: brush teeth thoroughly after meals or at least twice a day or use as directed by a dentist
- do not swallow
- to minimize swallowing use a pea-sized amount in children under 6
- supervise children's brushing until good habits are established
- children under 2 yrs.: ask a dentist
- adults and children 2 yrs. & older: brush teeth thoroughly after meals or at least twice a day or use as directed by a dentist
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
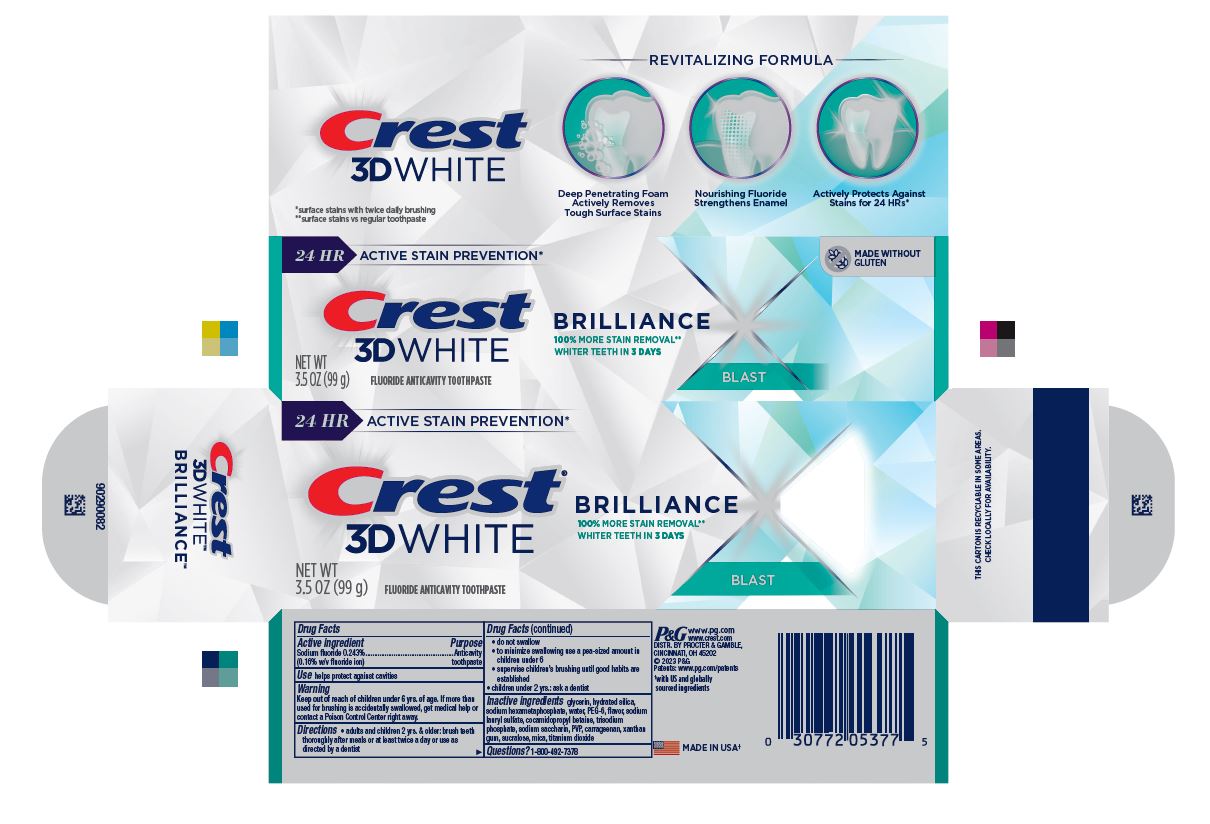
- PRINCIPAL DISPLAY PANEL - 99 g Tube Carton
-
INGREDIENTS AND APPEARANCE
CREST 3D WHITE BRILLIANCE BLAST
sodium fluoride paste, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:37000-916 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 1.6 mg in 1 g Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) HYDRATED SILICA (UNII: Y6O7T4G8P9) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) SACCHARIN SODIUM (UNII: SB8ZUX40TY) XANTHAN GUM (UNII: TTV12P4NEE) SUCRALOSE (UNII: 96K6UQ3ZD4) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) MICA (UNII: V8A1AW0880) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM PHOSPHATE, TRIBASIC, ANHYDROUS (UNII: SX01TZO3QZ) CARRAGEENAN (UNII: 5C69YCD2YJ) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM POLYMETAPHOSPHATE (UNII: P1BM4ZH95L) POLYETHYLENE GLYCOL 300 (UNII: 5655G9Y8AQ) Product Characteristics Color white Score Shape Size Flavor PEPPERMINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:37000-916-41 1 in 1 CARTON 07/01/2018 1 116 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:37000-916-82 2 in 1 CELLO PACK 07/01/2018 2 1 in 1 CARTON 2 116 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:37000-916-39 1 in 1 CARTON 09/15/2021 3 110 g in 1 TUBE; Type 0: Not a Combination Product 4 NDC:37000-916-35 1 in 1 CARTON 09/15/2021 4 99 g in 1 TUBE; Type 0: Not a Combination Product 5 NDC:37000-916-36 2 in 1 CELLO PACK 07/01/2018 5 1 in 1 CARTON 5 99 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M021 07/01/2018 Labeler - The Procter & Gamble Manufacturing Company (004238200)

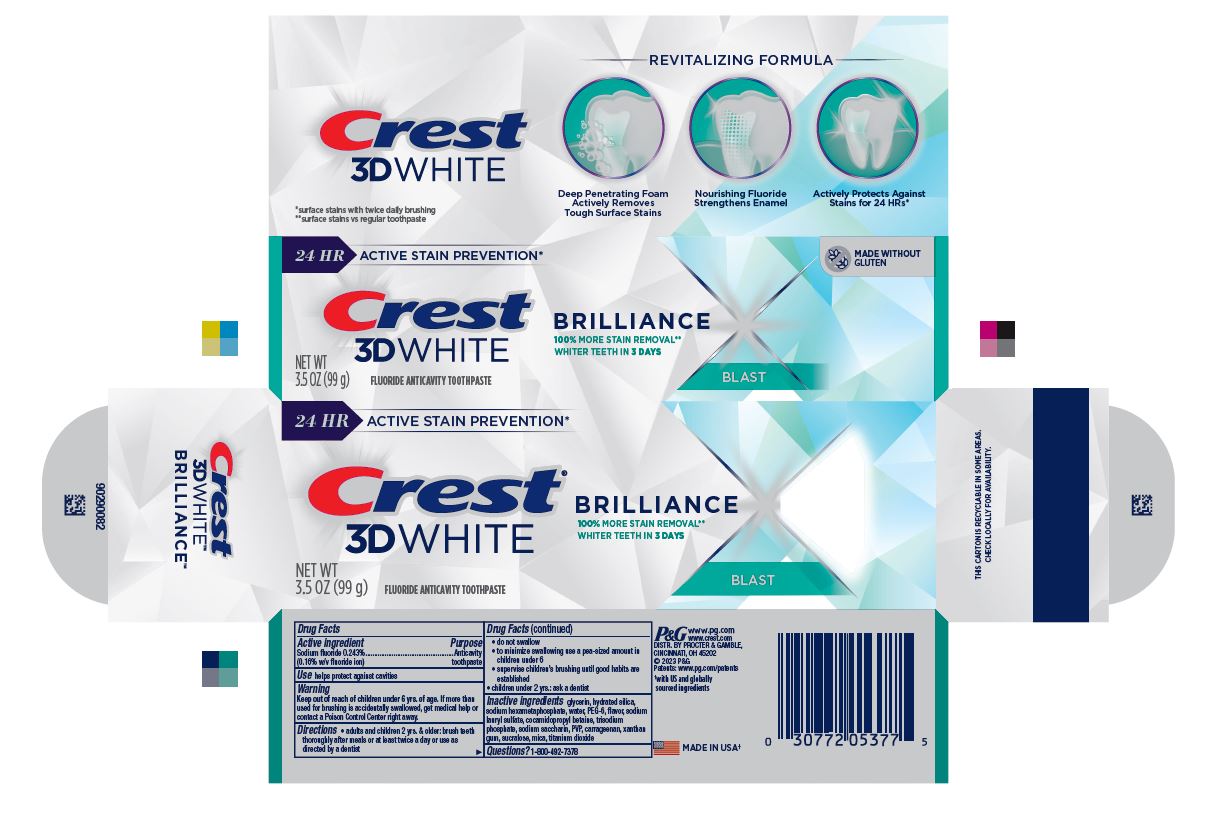
 24 HR ACTIVE STAIN PREVENTION*
24 HR ACTIVE STAIN PREVENTION*