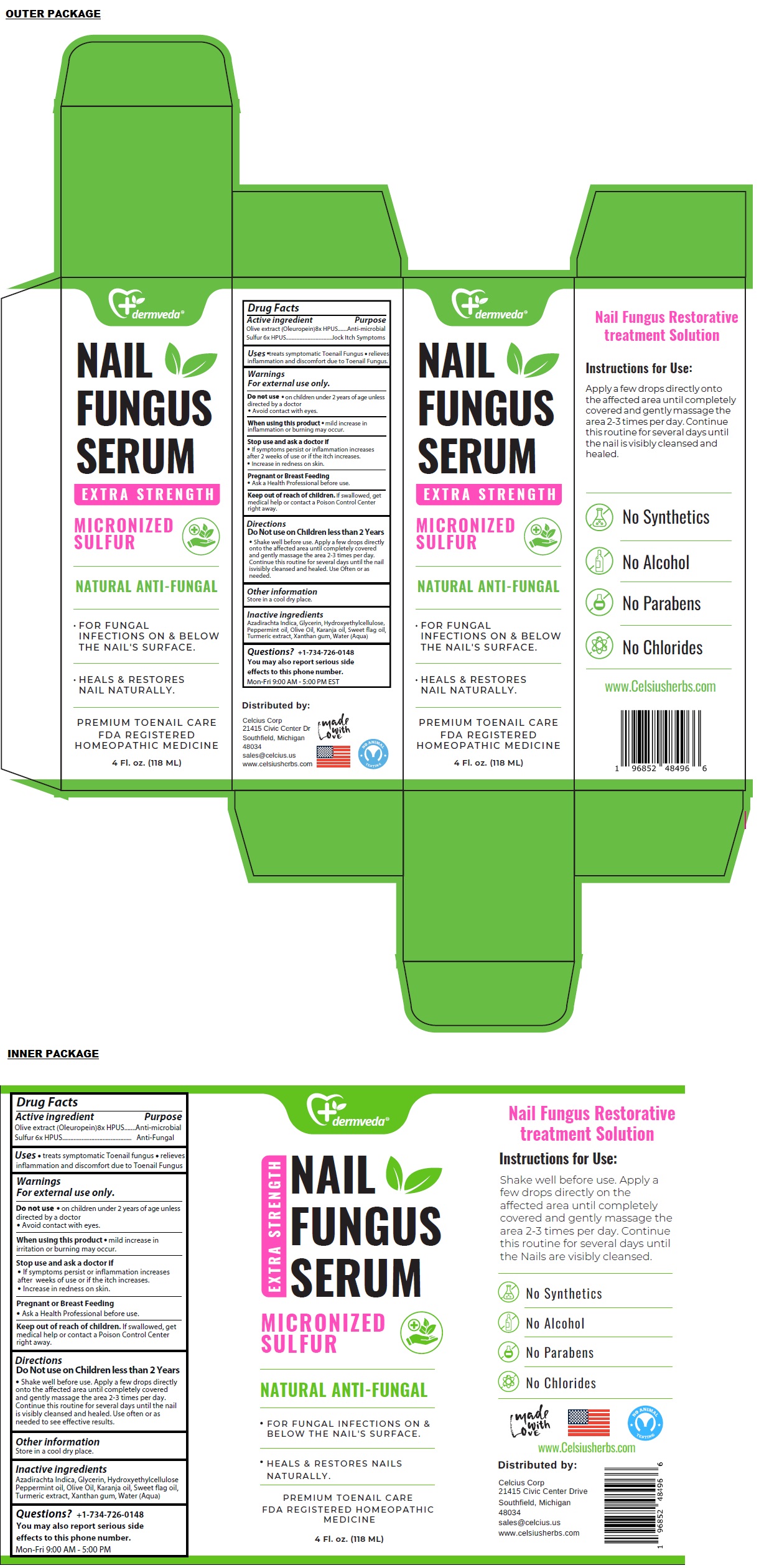
Label: DERMVEDA NAIL FUNGUS SERUM- olive extract, sulfur solution
- NDC Code(s): 83819-120-04
- Packager: Celcius Corp.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 25, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only.
Do not use • on children under 2 years of age unless directed by a doctor.
• Avoid contact with eyes.When using this product • mild increase in inflammation or burning may occur.
Stop use and ask a doctor if
• If symptoms persist or inflammation increases after 2 weeks of use or if the itch increases.
• Increase in redness on skin.Pregnant or Breast Feeding
• Ask a Health Professional before use. -
Directions
Do Not use on Children less than 2 Years
• Shake well before use. Apply a few drops directly onto the affected area until completely covered and gently massage the area 2-3 times per day. Continue this routine for several days until the nail is visibly cleansed and healed. Use Often or as needed to see effective results.
- Other information
- Inactive ingredients
- Questions?
-
SPL UNCLASSIFIED SECTION
EXTRA STRENGTH
MICRONIZED SULFUR
NATURAL ANTI-FUNGAL
• FOR FUNGAL INFECTIONS ON & BELOW THE NAIL'S SURFACE.
• HEALS & RESTORES NAIL NATURALLY.PREMIUM TOENAIL CARE
FDA REGISTERED
HOMEOPATHIC MEDICINENail Fungus Restorative treatment Solution
No Synthetics
No Alcohol
No Parabens
No ChloridesDistributed by:
Celcius Corp
21415 Civic Center Dr
Southfield, Michigan
48034
sales@celcius.us
www.celsiusherbs.commade with Love
NO ANIMAL TESTING
- Packaging
-
INGREDIENTS AND APPEARANCE
DERMVEDA NAIL FUNGUS SERUM
olive extract, sulfur solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83819-120 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OLEA EUROPAEA LEAF (UNII: MJ95C3OH47) (OLEA EUROPAEA LEAF - UNII:MJ95C3OH47) OLEA EUROPAEA LEAF 8 [hp_X] in 118 mL SULFUR (UNII: 70FD1KFU70) (SULFUR - UNII:70FD1KFU70) SULFUR 6 [hp_X] in 118 mL Inactive Ingredients Ingredient Name Strength AZADIRACHTA INDICA SEED OIL (UNII: 4DKJ9B3K2T) GLYCERIN (UNII: PDC6A3C0OX) HYDROXYETHYL CELLULOSE, UNSPECIFIED (UNII: T4V6TWG28D) PEPPERMINT OIL (UNII: AV092KU4JH) OLIVE OIL (UNII: 6UYK2W1W1E) KARUM SEED OIL (UNII: 62160PU6FJ) CALAMUS OIL (UNII: 5F9K5X640P) TURMERIC (UNII: 856YO1Z64F) XANTHAN GUM (UNII: TTV12P4NEE) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83819-120-04 1 in 1 BOX 12/25/2023 1 118 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 12/25/2023 Labeler - Celcius Corp. (117755609)