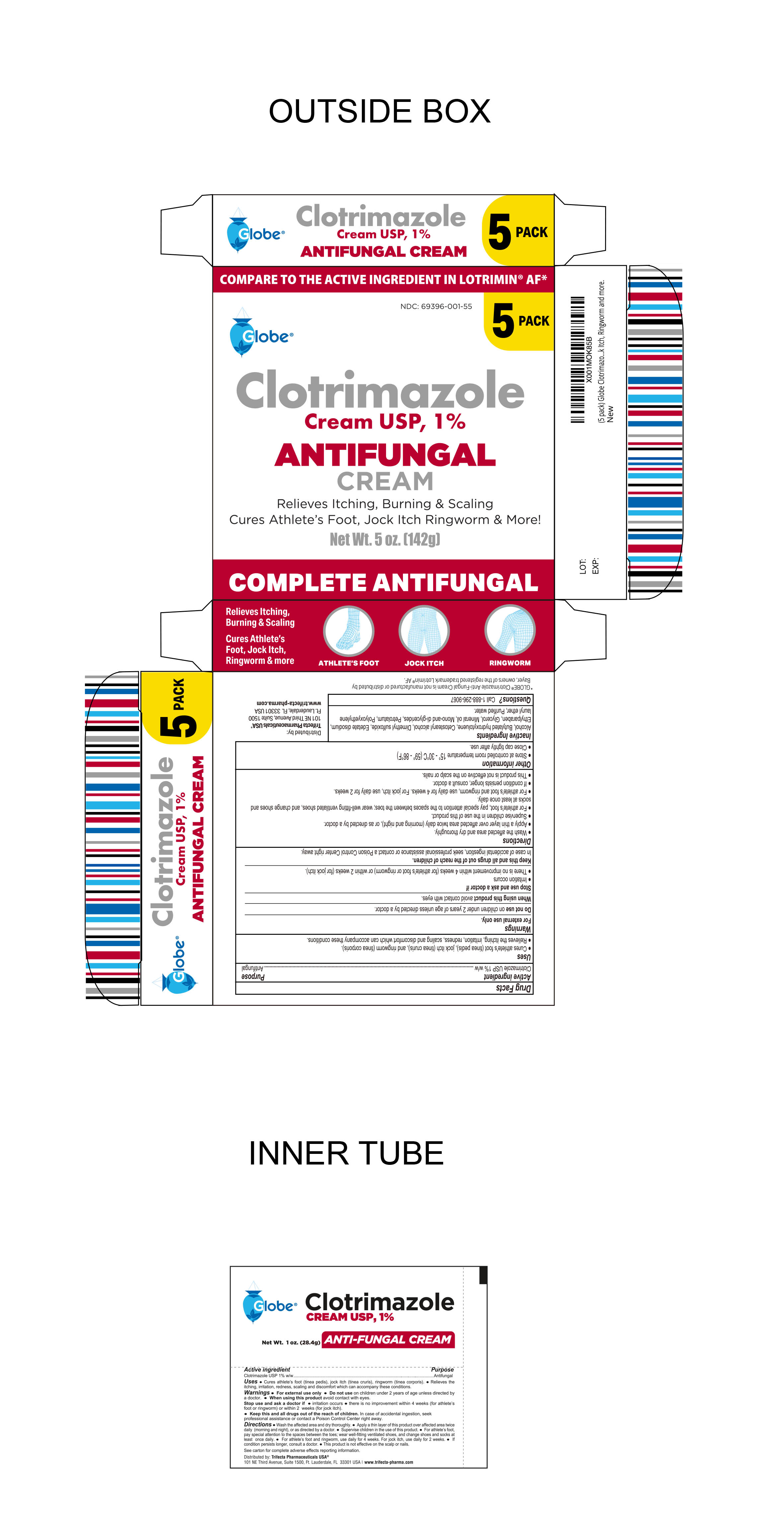
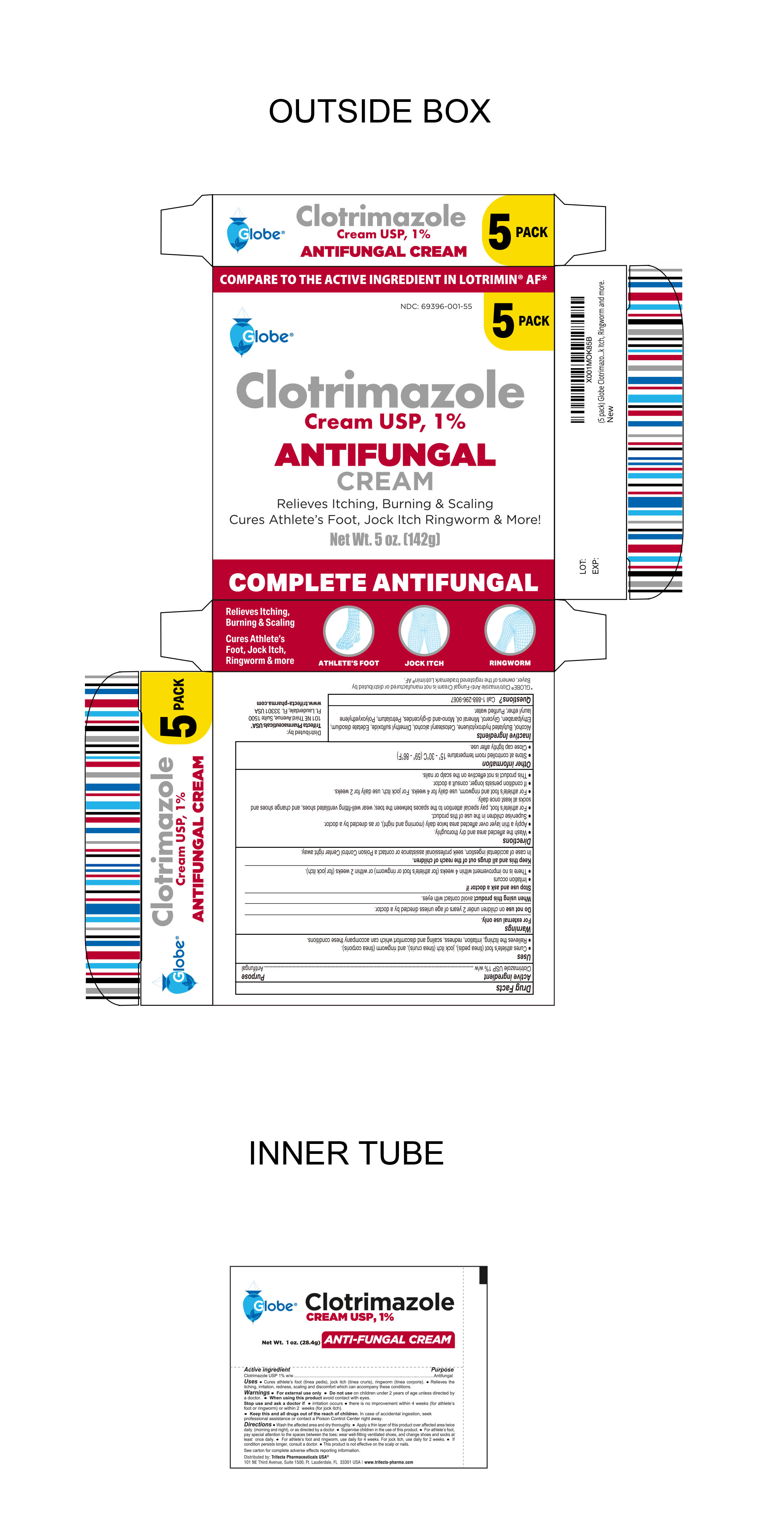
Label: CLOTRIMAZOLE cream
-
NDC Code(s):
69396-001-04,
69396-001-05,
69396-001-09,
69396-001-20, view more69396-001-55
- Packager: TRIFECTA PHARMACEUTICALS USA LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- Do Not Use
- When using this product
- Stop use and ask doctor if
- Keep this and all drugs out of the reach of children
-
Directions
- Wash the affected area and dry thoroughly.
● Apply a thin layer of this product over affected area twice daily (morning and night), or as directed by a doctor.
● Supervise children in the use of this product.
● For athlete’s foot, pay special attention to the spaces between the toes; wear well-fitting ventilated shoes, and change shoes and socks at least once daily.
● For athlete’s foot and ringworm, use daily for 4 weeks. For jock itch, use daily for 2 weeks.
● If conditions persists longer, consult a doctor.
● This product is not effective on the scalp or nails.
- Other Information
- Inactive Ingredients
- Questions?
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CLOTRIMAZOLE
clotrimazole creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69396-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLOTRIMAZOLE (UNII: G07GZ97H65) (CLOTRIMAZOLE - UNII:G07GZ97H65) CLOTRIMAZOLE 1 g in 100 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) DIMETHYL SULFOXIDE (UNII: YOW8V9698H) ETHYLPARABEN (UNII: 14255EXE39) GLYCERIN (UNII: PDC6A3C0OX) LAURETH-23 (UNII: N72LMW566G) PETROLATUM (UNII: 4T6H12BN9U) WATER (UNII: 059QF0KO0R) EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCERYL MONO AND DIPALMITOSTEARATE (UNII: KC98RO82HJ) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) ALCOHOL (UNII: 3K9958V90M) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69396-001-55 5 in 1 BOX 11/06/2023 1 28.4 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:69396-001-05 1 in 1 BOX 08/23/2016 2 15 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:69396-001-09 144 in 1 BOX 03/09/2022 3 0.9 g in 1 PACKAGE; Type 0: Not a Combination Product 4 NDC:69396-001-04 1 in 1 BOX 05/03/2022 4 113.4 g in 1 TUBE; Type 0: Not a Combination Product 5 NDC:69396-001-20 1 in 1 BOX 03/10/2015 5 28.4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M005 03/10/2015 Labeler - TRIFECTA PHARMACEUTICALS USA LLC (079424163) Registrant - Trifecta Pharmaceuticals USA (079424163)