Label: R-GENE- arginine hydrochloride injection, solution
- NDC Code(s): 0009-0436-01
- Packager: Pharmacia & Upjohn Company LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated April 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Each 100 mL of R-Gene® 10 (Arginine Hydrochloride Injection, USP) for intravenous use contains 10 g of L-Arginine Hydrochloride, USP in Water for Injection, USP (equivalent to a 10% solution) (0.1 g/mL). L-arginine is a naturally occurring amino acid.
R-Gene® 10 is hypertonic (950 mOsmol/liter) and contains 47.5 mEq of chloride ion per 100 mL of solution. The pH is adjusted to 5.6 (5.0–6.5) with arginine base or hydrochloric acid.
-
CLINICAL PHARMACOLOGY
Intravenous infusion of R-Gene® 10 often induces a pronounced rise in the plasma level of human growth hormone (HGH) in subjects with intact pituitary function. This rise is usually diminished or absent in patients with impairment of this function.
Expected Plasma Levels of HGH in ng/mL Patient Control Range Range of Peak Response to Arginine Normal
0–6
10–30
Pituitary
deficient
0–4
0–10
These ranges are based on the mean values of plasma HGH levels calculated from the data of several clinical investigators and reflect their experiences with various methods of radioimmunoassay. Upon gaining experience with this diagnostic test, each clinician will establish his/her own ranges for control and peak levels of HGH.
L-arginine is a normal metabolite in animals and man and has a low order of toxicity.
-
INDICATIONS AND USAGE
R-Gene® 10 is indicated as an intravenous stimulant to the pituitary for the release of human growth hormone in patients where the measurement of pituitary reserve for HGH can be of diagnostic usefulness. It can be used as a diagnostic aid in such conditions as panhypopituitarism, pituitary dwarfism, chromophobe adenoma, postsurgical craniopharyngioma, hypophysectomy, pituitary trauma, acromegaly, gigantism and problems of growth and stature.
If the insulin hypoglycemia test has indicated a deficiency of pituitary reserve for HGH, a test with R-Gene® 10 is advisable to confirm the negative response. This can be done after a waiting period of one day. As patients may not respond to R-Gene® 10 (Arginine Hydrochloride Injection, USP) during the first test, the unresponsive patient should be tested again to confirm the negative result. A second test can be performed after a waiting period of one day. Some patients who respond to R-Gene® 10 do not respond to insulin and vice versa. The rate of false positive responses for R-Gene® 10 is approximately 32%, and the rate of false negatives is approximately 27%.
- CONTRAINDICATIONS
-
WARNINGS
There have been reports of overdosage of R-Gene® 10 in pediatric patients leading to death. EXTREME CAUTION MUST BE EXERCISED WHEN INFUSING R-GENE® 10 INTO PEDIATRIC PATIENTS. OVERDOSAGE OF R-GENE® 10 IN PEDIATRIC PATIENTS CAN RESULT IN HYPERCHLOREMIC METABOLIC ACIDOSIS, CEREBRAL EDEMA, OR POSSIBLY DEATH.
Hypersensitivity reactions, including anaphylaxis have been reported. Appropriate medical support should be available during R-Gene® 10 administration. If anaphylaxis or other serious hypersensitivity reaction occurs, R-Gene® 10 should be discontinued and appropriate medical treatment initiated.
R-Gene® 10 should always be administered by intravenous infusion because of its hypertonicity.
R-Gene® 10 is a diagnostic aid and is not intended for therapeutic use.
-
PRECAUTIONS
General
R-Gene® 10 is a hypertonic (950 mOsmol/liter) and acidic (average pH of 5.6) solution that can cause irritation and damage to tissues. Care should be used to ensure administration of R-Gene® 10 through a patent catheter within a patent vein. Excessive rates of infusion may result in local irritation and in flushing, nausea, or vomiting. Inadequate dosing or prolongation of the infusion period may diminish the stimulus to the pituitary and nullify the test.
The arginine in R-Gene® 10 can be metabolized resulting in nitrogen-containing products for excretion. The effect of an acute amino acid or nitrogen burden upon patients with impairment of renal function should be considered when R-Gene® 10 is to be administered.
The chloride content of R-Gene® 10 is 47.5 mEq per 100 mL of solution, and the effect of infusing this amount of chloride into patients with electrolyte imbalance should be evaluated before the test is undertaken.
It should be noted that the basal and post stimulation levels of growth hormone are elevated in patients who are pregnant or are taking oral contraceptives.
Carcinogenesis, mutagenesis, and impairment of fertility
Long term animal studies have not been performed to evaluate the carcinogenic potential, the mutagenic potential or the effect on fertility of intravenously administered R-Gene® 10.
Pregnancy
Reproduction studies have been performed in rabbits and mice at doses 12 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to R-Gene® 10 (10% Arginine Hydrochloride Injection, USP). There have been no adequate or well controlled studies for the use of R-Gene® 10 in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should not be used during pregnancy.
Nursing Mothers
It is not known whether intravenous administration of R-Gene® 10 could result in significant quantities of arginine in breast milk. Systemically administered amino acids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, caution should be exercised when R-Gene® 10 is to be administered to nursing women.
-
ADVERSE REACTIONS
Adverse reactions associated with 1670 infusions in premarketing studies were as follows:
Non-specific side effects consisting of nausea, vomiting, headache, flushing, numbness and local venous irritation were reported in approximately 3% of the patients.
One patient had an allergic reaction which was manifested as a confluent macular rash with reddening and swelling of the hands and face. The rash subsided rapidly after the infusion was terminated and 50 mg of diphenhydramine were administered. One patient had an apparent decrease in platelet count from 150,000 to 60,000. One patient with a history of acrocyanosis had an exacerbation of this condition following infusion of R-Gene® 10.
Post Marketing Experience:
The following adverse events have been reported during post-marketing use: extravasation leading to burn-like reaction and/or skin necrosis requiring surgical intervention, hypersensitivity reactions including anaphylaxis, and hematuria that in some cases occurred 1–2 days after an R-Gene® 10 administration. Because these adverse events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
OVERDOSAGE
An overdosage may cause a transient metabolic acidosis with hyperventilation, which could lead to death (see "WARNINGS"). In most cases the acidosis will self-compensate and the base deficit will return to normal following completion of the infusion. If the condition persists, the deficit should be determined and corrected by a calculated dose of an alkalizing agent.
-
DOSAGE AND ADMINISTRATION
Adult Dosage
The recommended adult dose is 30 g arginine hydrochloride (300 mL of R-Gene® 10) administered by intravenous infusion over 30 minutes. The total dose should not exceed 30 g arginine hydrochloride. See Directions for Use for preparation instructions.
Pediatric Dosage
The recommended pediatric dose is 0.5 g/kg arginine hydrochloride (5 mL/kg of R-Gene® 10) administered by intravenous infusion over 30 minutes. The total dose should not exceed 30 g arginine hydrochloride.
- •
- For patients weighing 59 kg or less, withdraw a weight based dose from a sealed R-Gene® 10 bottle and place in a separate container for intravenous infusion to avoid the inadvertent delivery and administration of the total volume from the commercially available container. See Directions for Use for preparation instructions.
- •
- For patients weighing 60 kg or more, the recommended dose is 30 g arginine hydrochloride (300 mL of R-Gene® 10). See Directions for Use for preparation instructions
Test Procedure
The intravenous infusion of R-Gene® 10 is a part of the test for measurement of pituitary reserve of human growth hormone and, for successful administration of the test, clinical conditions and procedures should be as follows:
- 1.
- The test should be scheduled in the morning following a normal night's sleep, and an overnight fast should continue through the test period.
- 2.
- Patients must be placed at bed rest for at least 30 minutes before the infusion begins. Care should be taken to minimize apprehension and distress. This is particularly important in children.
- 3.
- R-Gene® 10 (Arginine Hydrochloride Injection, USP) is a hypertonic solution and should only be infused through an indwelling needle or soft catheter placed in an antecubital vein or other suitable vein (see PRECAUTIONS). Blood samples should be taken by venipuncture from the contra-lateral arm.
- 4.
- A desirable schedule for drawing blood samples is at −30, 0, 30, 60, 90, 120 and 150 minutes.
- 5.
- R-Gene® 10 should be infused beginning at zero time at a uniform rate which will permit the recommended dose to be administered over 30 minutes.
- 6.
- Blood samples should be promptly centrifuged and the plasma stored at −20°C until assayed by one of the published radioimmunoassay procedures.
- 7.
- Diagnostic test results showing a deficiency of pituitary reserve for HGH should be confirmed by a second test with R-Gene® 10, or one may elect to confirm with the insulin hypoglycemia test. A waiting period of one day is advised between tests.
Directions for Use
R-Gene® 10 is provided as a ready-to-use solution for patients weighing 60 kg (132 lbs) or more and should not be further diluted. For pediatric patients weighing 59 kg (130 lbs) or less a dose must be placed in a separate container. Follow the preparation instructions below.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
For Pediatric Patients weighing 59 kg (130 lbs) or less:
Withdraw a weight-based dose from an intact sealed bottle of R-Gene® 10. The entire 300 mL bottle of R-Gene® 10 for infusion is not intended for use in patients weighing 59 kg or less. The dose must be placed in a separate container, such as an evacuated sterile glass container designed for intravenous administration, using aseptic technique.
Additionally, R-Gene® 10 is stable in polypropylene syringes and plastic containers made of either polyvinyl chloride (PVC) or ethylene vinyl acetate (EVA).
The post-penetration storage period is not more than 4 hours including infusion time at room temperature or 24 hours at refrigerated temperature (2-8°C).
The healthcare professional administering the dose should verify the accuracy of the dose prior to administration.
Use only if the solution is clear. Discard any unused drug product.
For Adults and Pediatric Patients weighing 60kg (132 lbs) or more:
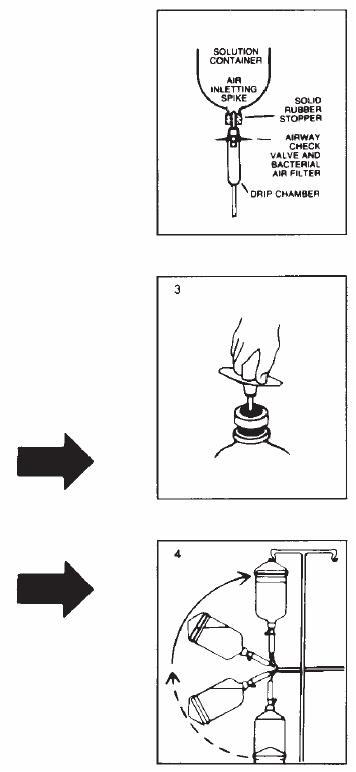
Follow these directions using aseptic technique. As R-Gene® 10 for intravenous use is provided in glass containers, a standard air-inletting, air-filtering intravenous infusion set with a bacterial air filter is required.
- 1.
- Use only if solution is clear and seal is intact. Carefully examine bottle for evidence of damage, e.g., small cracks, dents in seal, or areas of dried powder on exterior. Do not administer contents if such damage is found.
- 2.
- Remove plastic flip off lid from bottle to expose rubber stopper, taking care that you do not contaminate the target site of the stopper with fingers, hair, clothing, etc. Immediately perform step #3.
- 3.
- With shut-off clamp closed, remove sterility protector from spike of administration set and immediately insert set with a quick thrust into center of stopper with bottle upright on table. (Push straight in — don't twist — twisting may cause stopper coring.)
- 4.
- Promptly invert bottle to automatically establish fluid level in drip chamber and to check for vacuum by observing rising filtered air bubbles. Discard bottle if there is no vacuum or if the solution is not clear.
- 5.
- Clear tubing of air. Proceed with infusion.
-
HOW SUPPLIED
R-Gene® 10 is supplied as a 30 g/300 mL (0.1 g/mL) fill in 500 mL glass containers.
Preservative Free: Discard any unused portion.
NDC 0009-0436-01
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. It is recommended that the product be stored at room temperature (25°C); however, brief exposure up to 40°C does not adversely affect the product. Solution that has been frozen must not be used.
LAB-0020-13.0
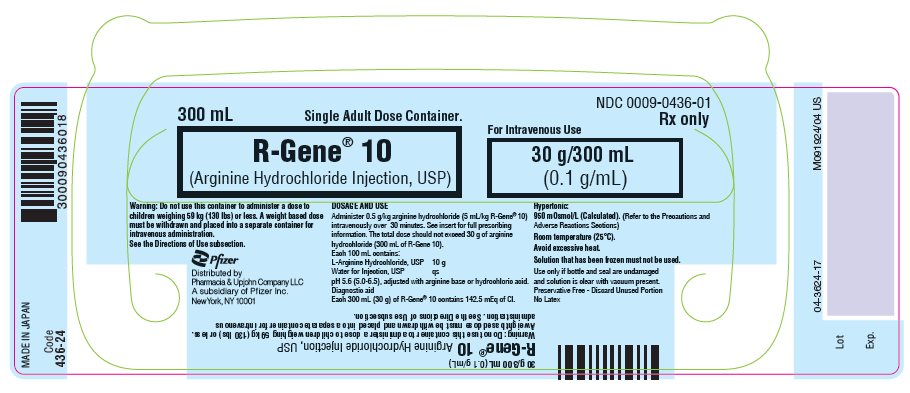
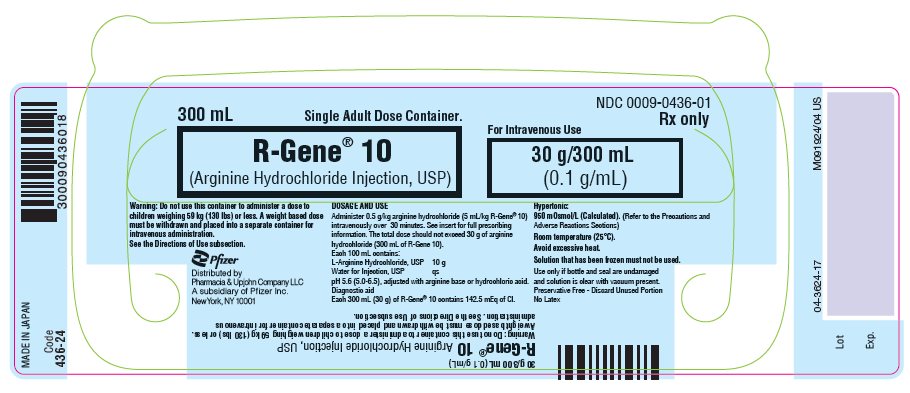
Revised Aug 2023 - PRINCIPAL DISPLAY PANEL - 300 mL Container Label
-
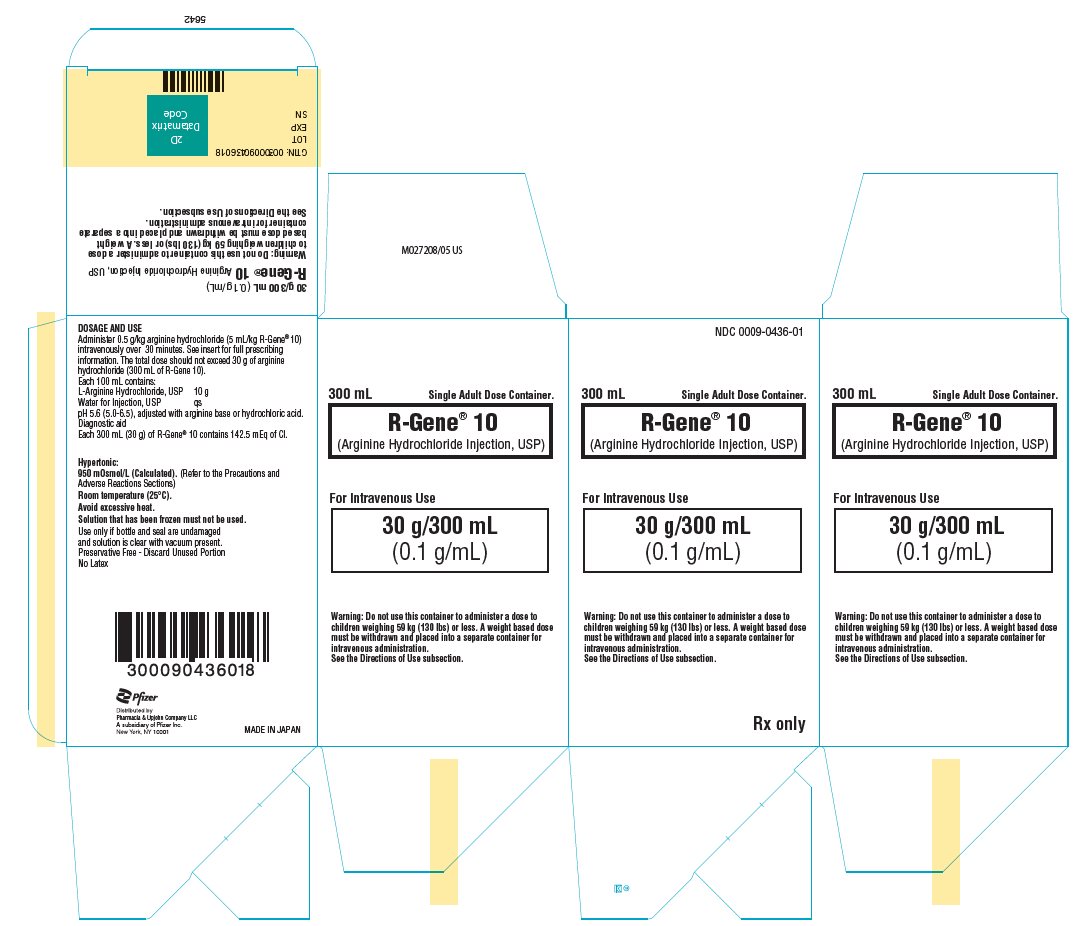
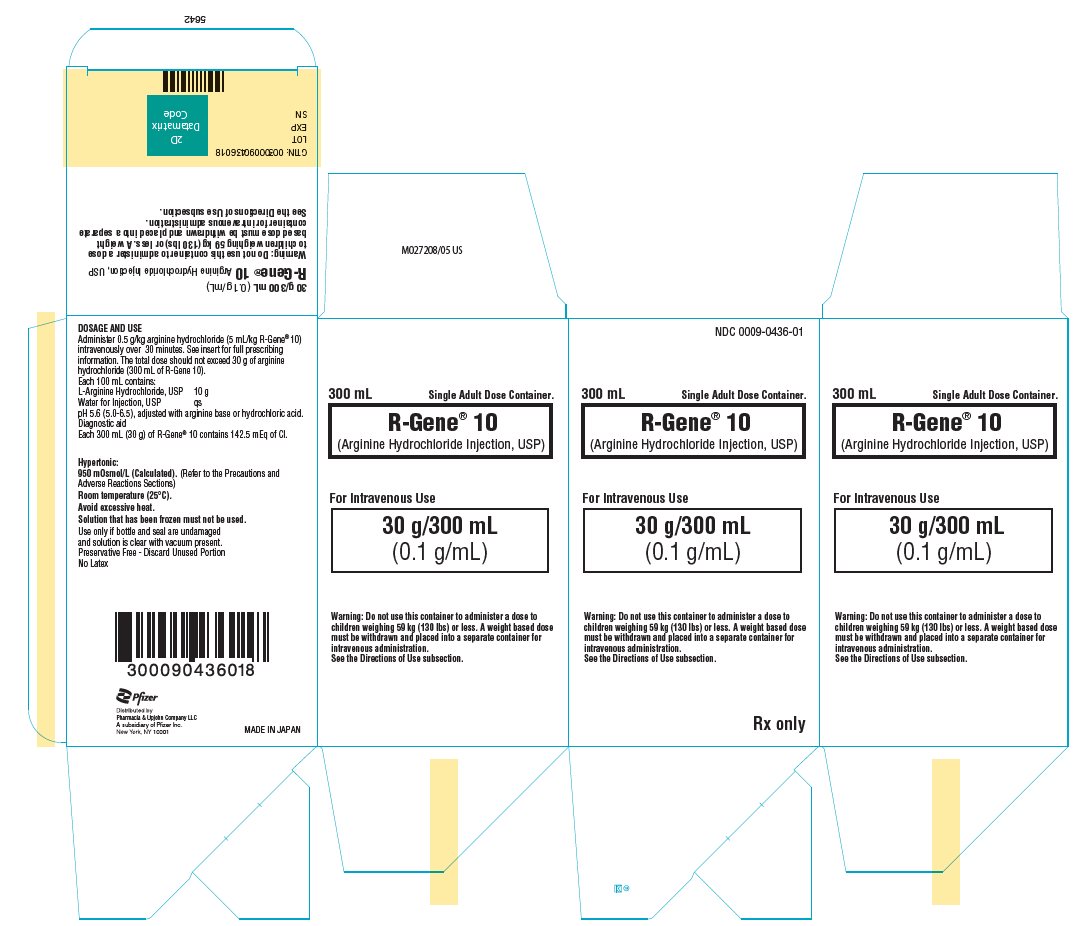
PRINCIPAL DISPLAY PANEL - 300 mL Container Carton
NDC 0009-0436-01
300 mL
Single Adult Dose Container.R-Gene® 10
(Arginine Hydrochloride Injection, USP)For Intravenous Use
30 g/300 mL
(0.1 g/mL)Warning: Do not use this container to administer a dose to
children weighing 59 kg (130 lbs) or less. A weight based dose
must be withdrawn and placed into a separate container for
intravenous administration.
See the Directions of Use subsection.Rx only
-
INGREDIENTS AND APPEARANCE
R-GENE
arginine hydrochloride injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0009-0436 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARGININE HYDROCHLORIDE (UNII: F7LTH1E20Y) (ARGININE - UNII:94ZLA3W45F) ARGININE HYDROCHLORIDE 10 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0009-0436-01 1 in 1 CARTON 06/15/1976 1 300 mL in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA016931 06/15/1976 Labeler - Pharmacia & Upjohn Company LLC (618054084) Registrant - Pfizer Inc (113480771)