Label: DRYMARINE AGE CONTROL- octinoxate, titanium dioxide, avobenzone cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 42248-109-02 - Packager: Zenith Medicosm SL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 21, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
-
Description
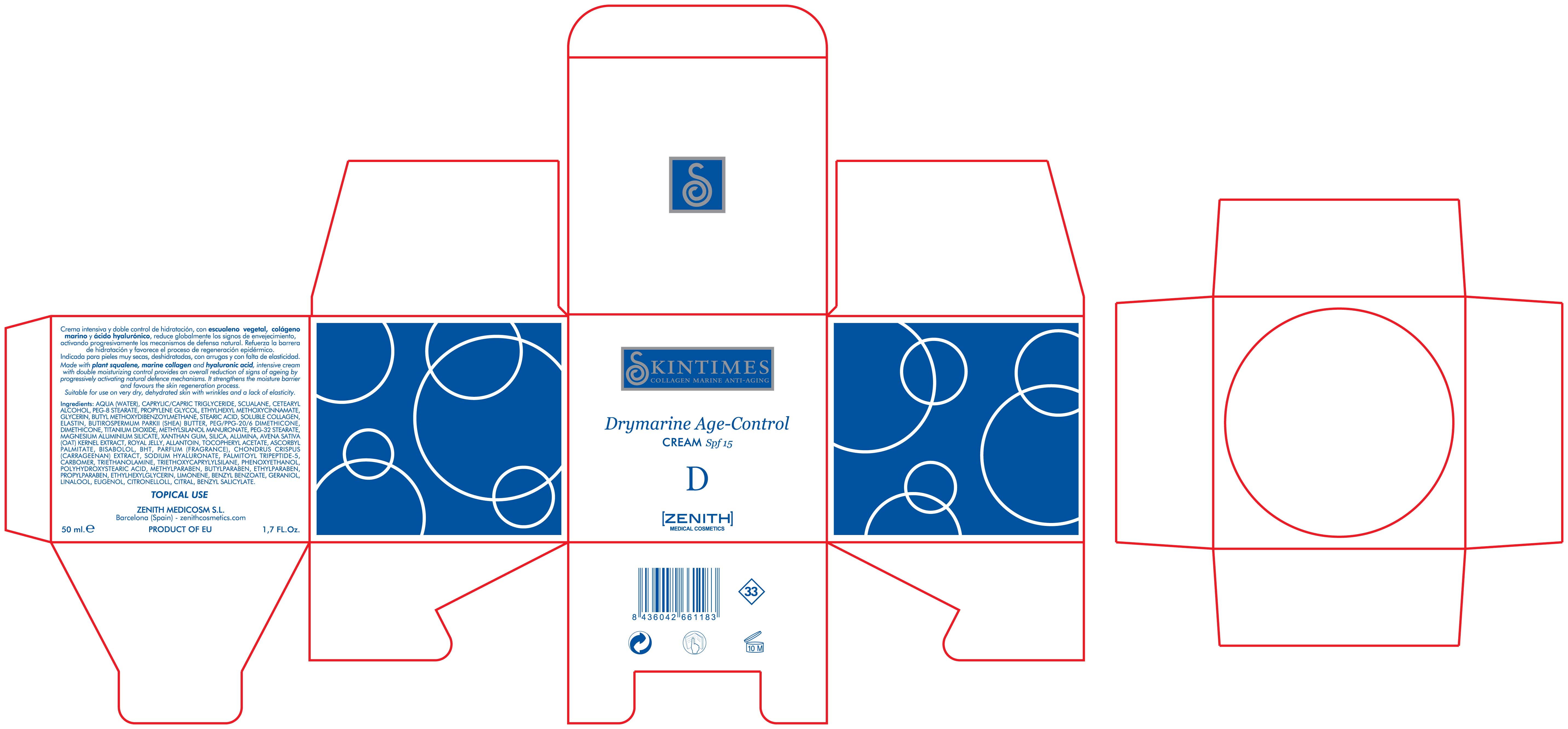
Made with plant squalene, marine collagen and hyaluronic acid, intensive cream with double moisturizing control provides an overall reduction of signs of ageing by progressively activating natural defence mechanisms. It strenghtens the moisture barrier and favours the skin regeneration process. Suitable for use on very dry, dehydrated skin with wrinkles and lack of elasticity.
50ml. 1.7 fl. Oz
- Warning
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DRYMARINE AGE CONTROL
octinoxate, titanium dioxide, avobenzone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42248-109 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 3.75 mL in 50 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 12.5 mL in 50 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 1.5 mL in 50 mL Inactive Ingredients Ingredient Name Strength MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) PROPYLPARABEN (UNII: Z8IX2SC1OH) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) DIMETHICONE (UNII: 92RU3N3Y1O) XANTHAN GUM (UNII: TTV12P4NEE) TRICAPRYLIN (UNII: 6P92858988) WATER (UNII: 059QF0KO0R) SQUALANE (UNII: GW89575KF9) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PEG-8 STEARATE (UNII: 2P9L47VI5E) TROLAMINE (UNII: 9O3K93S3TK) GLYCERIN (UNII: PDC6A3C0OX) CHONDRUS CRISPUS (UNII: OQS23HUA1X) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) HYDROLYZED ELASTIN, BOVINE, ALKALINE (1000 MW) (UNII: ZR28QKN0WT) CARBOMER 934 (UNII: Z135WT9208) SHEA BUTTER (UNII: K49155WL9Y) PEG/PPG-20/6 DIMETHICONE (UNII: PWZ7N4UIKE) METHYLPARABEN (UNII: A2I8C7HI9T) COLLAGEN, SOLUBLE, FISH SKIN (UNII: 8JC99XGU4W) PEG-32 STEARATE (UNII: 33GX5WQC0M) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ALUMINUM OXIDE (UNII: LMI26O6933) STEARIC ACID (UNII: 4ELV7Z65AP) OAT (UNII: Z6J799EAJK) ROYAL JELLY (UNII: L497I37F0C) ASCORBYL PALMITATE (UNII: QN83US2B0N) CHONDRUS CRISPUS (UNII: OQS23HUA1X) HYALURONATE SODIUM (UNII: YSE9PPT4TH) PALMITOYL TRIPEPTIDE-5 (UNII: 2A3916MQHO) ALLANTOIN (UNII: 344S277G0Z) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) LEVOMENOL (UNII: 24WE03BX2T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PHENOXYETHANOL (UNII: HIE492ZZ3T) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) BUTYLPARABEN (UNII: 3QPI1U3FV8) ETHYLPARABEN (UNII: 14255EXE39) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) LIMONENE, (+/-)- (UNII: 9MC3I34447) BENZYL BENZOATE (UNII: N863NB338G) GERANIOL (UNII: L837108USY) LINALOOL, (+/-)- (UNII: D81QY6I88E) EUGENOL (UNII: 3T8H1794QW) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) CITRAL (UNII: T7EU0O9VPP) BENZYL SALICYLATE (UNII: WAO5MNK9TU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42248-109-02 1 in 1 BOX 1 50 mL in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 12/01/2011 Labeler - Zenith Medicosm SL (464239694) Registrant - Zenith Medicosm SL (464239694) Establishment Name Address ID/FEI Business Operations Zenith Medicosm SL 464239694 manufacture