Label: DECCOX M- decoquinate powder
- NDC Code(s): 54771-1510-4, 54771-1510-5
- Packager: Zoetis Inc.
- Category: OTC TYPE A MEDICATED ARTICLE ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated October 15, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
None
Decoquinate
Medicated Powder for Whole Milk
To be added to whole milk for the prevention of coccidiosis in ruminating and non-ruminating calves, including veal calves caused by Eimeria bovis and E. zuernii.
Feed for at least 28 days during periods of coccidiosis exposure or when experience indicates that coccidiosis is likely to be a hazard.
- Active ingredient(s)
- Inactive ingredients
-
Directions
Dosage:
Feed at the rate of 22.7 mg per 100 lb (0.5 mg per kg) body weight daily for at least 28 days.
Mixing Directions - Individual Animal
Prepare fresh daily as shown below.
½ teaspoon per 60 lb body weight
1 teaspoon per 120 lb body weight
1 ½ teaspoons per 180 lb body weight
One level measuring teaspoon contains 28 mg of decoquinate.
Important:
Completely stir daily dose of decoquinate powder into whole milk.
Feed entire portion immediately.
Mixing Directions - Continuous Agitation
Mix Deccox-M into the total amount of whole milk (0.5 to 1.5 gallon per calf daily) needed for the number of calves to be fed, using level measuring cups, teaspoons (tsp) or tablespoons (TBS). Prepare fresh daily and feed each calf an equal portion of the total amount of medicated whole milk prepared as shown below.
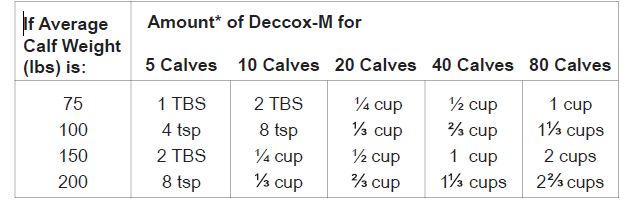
Mixing Table
*This is a measurement of volume not weight (do not convert to ounces).
Important:
Stir or agitate continuously.
Feed immediately in containers for individual animals only.
Do not use in bulk feeding containers (i.e., Calf-Bars).
Failure to properly mix and feed this product may result in underdosing or overdosing of some animals due to settling out of the drug.
- Other information
- Storage
- SPL UNCLASSIFIED SECTION
- Principal Display Panel 50lb bag
- Principal Display Panel 5lb bag
-
INGREDIENTS AND APPEARANCE
DECCOX M
decoquinate powderProduct Information Product Type OTC TYPE A MEDICATED ARTICLE ANIMAL DRUG Item Code (Source) NDC:54771-1510 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DECOQUINATE (UNII: 534I52PVWH) (DECOQUINATE - UNII:534I52PVWH) DECOQUINATE 3632 mg in 0.45 kg Inactive Ingredients Ingredient Name Strength SOYBEAN OIL (UNII: 241ATL177A) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) Product Characteristics Color WHITE (White to Tan) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54771-1510-4 2.27 kg in 1 BAG 2 NDC:54771-1510-5 22.68 kg in 1 BAG Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141060 01/01/2009 Labeler - Zoetis Inc. (828851555) Establishment Name Address ID/FEI Business Operations Hikal Limited 918602129 API MANUFACTURE Establishment Name Address ID/FEI Business Operations Zoetis LLC 099219842 MANUFACTURE




