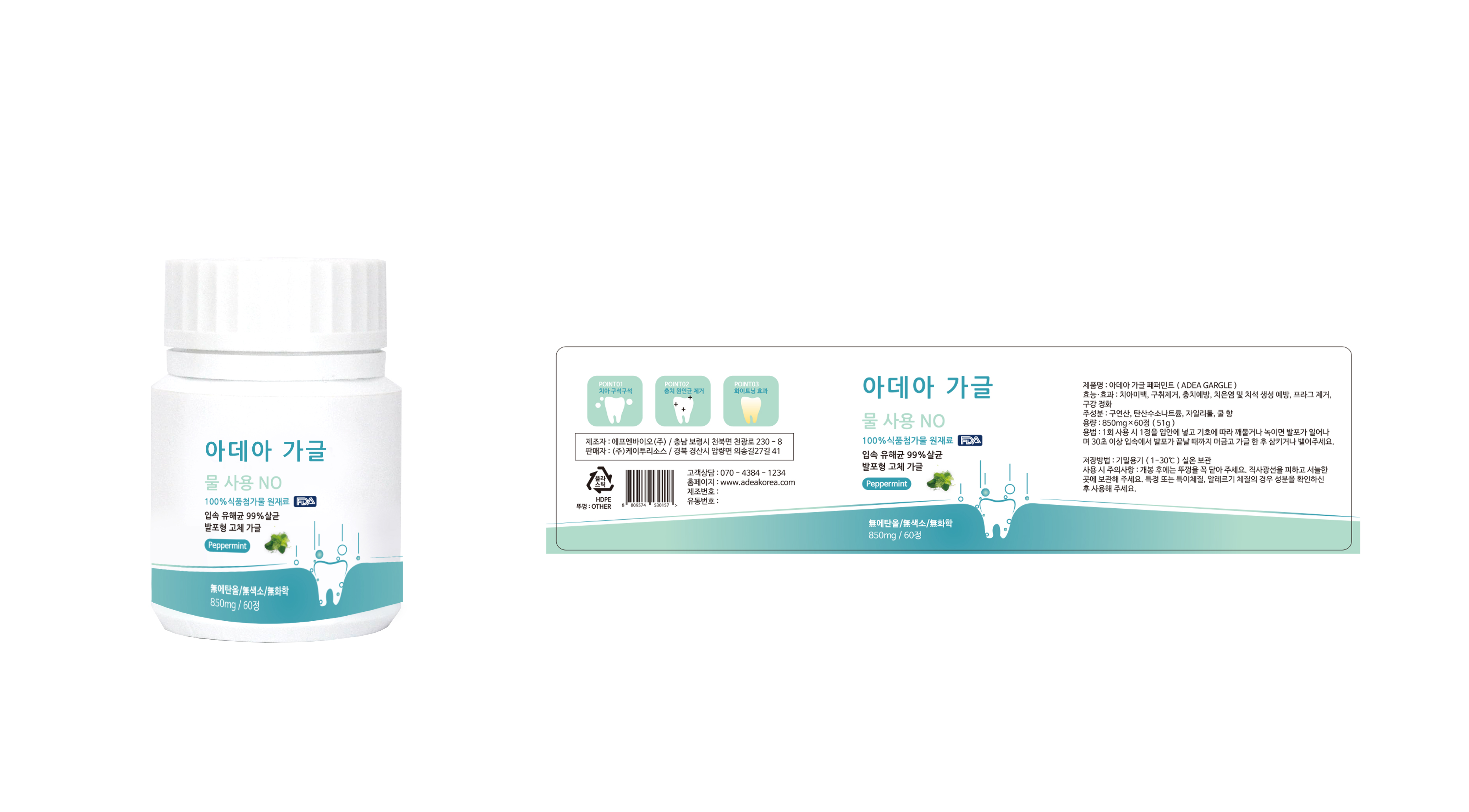
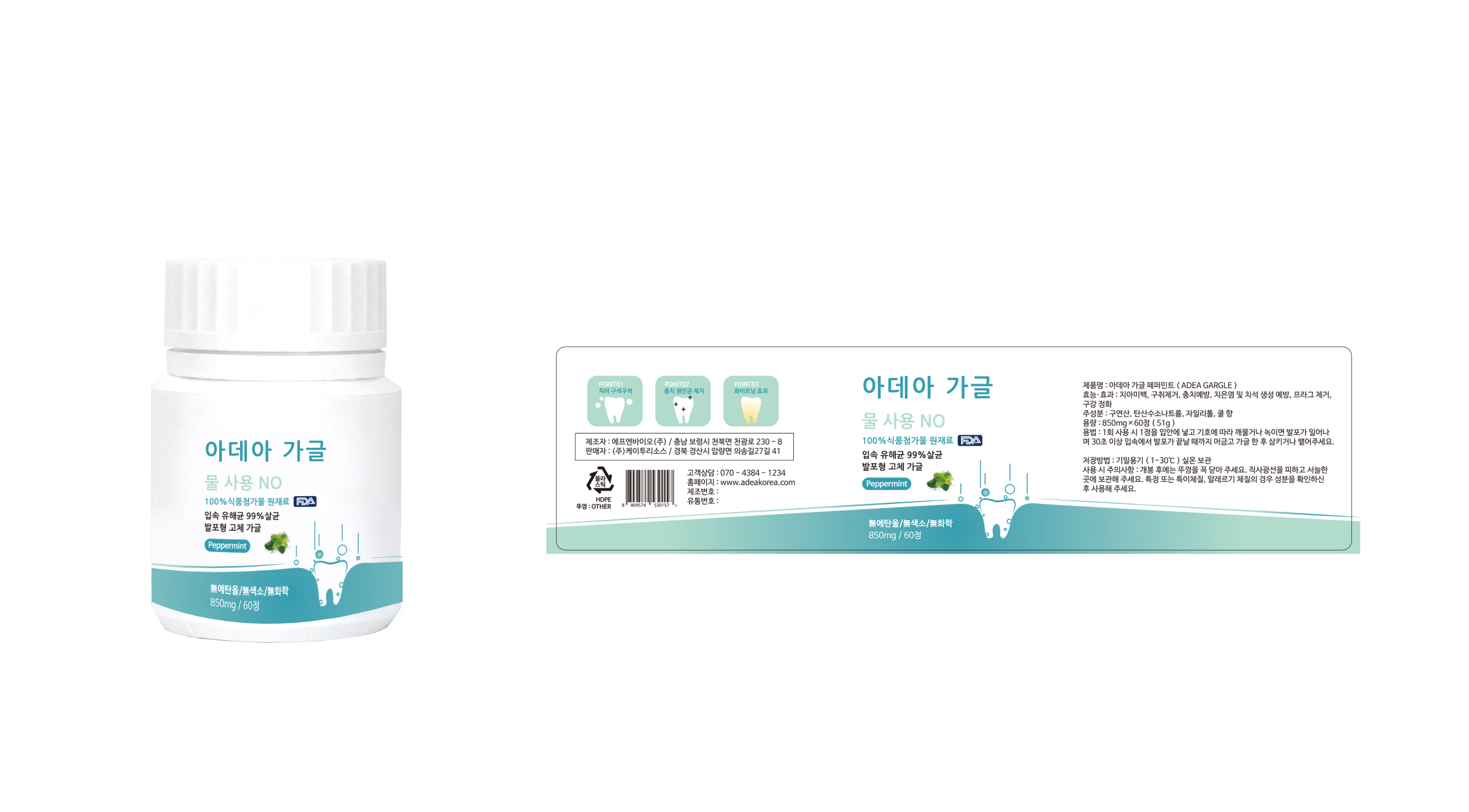
Label: ADEA GARGLE- citric acid, sodium hydrogen carbonate, xylitol tablet, chewable
-
Contains inactivated NDC Code(s)
NDC Code(s): 72191-0100-1 - Packager: K2resource co., ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated March 26, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- INACTIVE INGREDIENT
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ADEA GARGLE
citric acid, sodium hydrogen carbonate, xylitol tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72191-0100 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM BICARBONATE 36.2 g in 100 mg XYLITOL (UNII: VCQ006KQ1E) (XYLITOL - UNII:VCQ006KQ1E) XYLITOL 5 g in 100 mg Inactive Ingredients Ingredient Name Strength MENTHOL (UNII: L7T10EIP3A) Product Characteristics Color white Score score with uneven pieces Shape BULLET Size 17mm Flavor PEPPERMINT Imprint Code None Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72191-0100-1 850 mg in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/01/2018 Labeler - K2resource co., ltd. (694503238) Registrant - K2resource co., ltd. (694503238) Establishment Name Address ID/FEI Business Operations K2resource co., ltd. 694503238 manufacture(72191-0100)