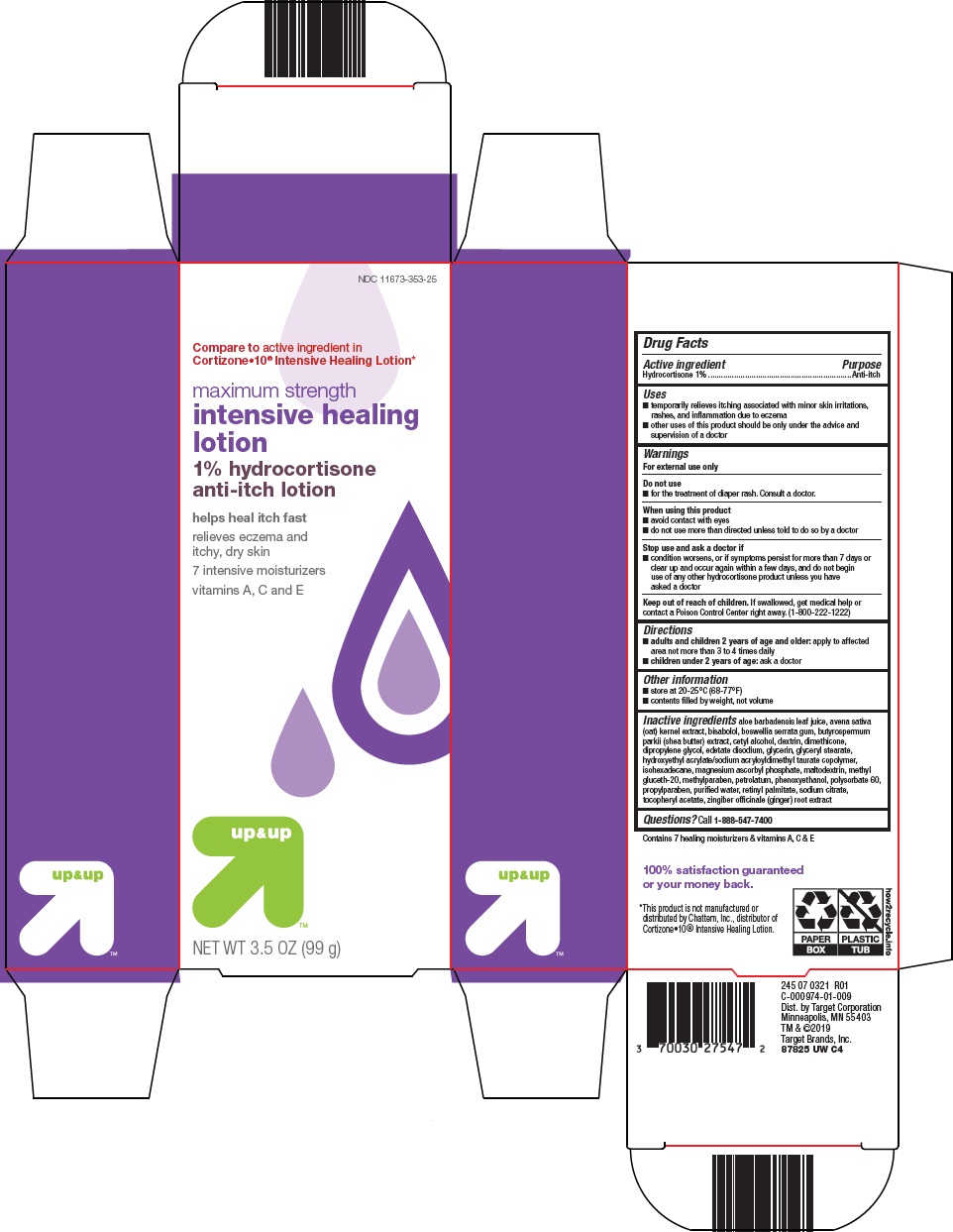
Label: UP AND UP INTENSIVE HEALING- hydrocortisone lotion
- NDC Code(s): 11673-353-25
- Packager: Target Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 1, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
- Directions
- Other information
-
Inactive ingredients
aloe barbadensis leaf juice, avena sativa (oat) kernel extract, bisabolol, boswellia serrata gum, butyrospermum parkii (shea butter) extract, cetyl alcohol, dextrin, dimethicone, dipropylene glycol, edetate disodium, glycerin, glyceryl stearate, hydroxyethyl acrylate/sodium acryloyldimethyl taurate copolymer, isohexadecane, magnesium ascorbyl phosphate, maltodextrin, methyl gluceth-20, methylparaben, petrolatum, phenoxyethanol, polysorbate 60, propylparaben, purified water, retinyl palmitate, sodium citrate, tocopheryl acetate, zingiber officinale (ginger) root extract
- Questions?
- Package/Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
UP AND UP INTENSIVE HEALING
hydrocortisone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11673-353 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE (UNII: WI4X0X7BPJ) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 1 g in 100 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) OAT (UNII: Z6J799EAJK) LEVOMENOL (UNII: 24WE03BX2T) INDIAN FRANKINCENSE (UNII: 4PW41QCO2M) CETYL ALCOHOL (UNII: 936JST6JCN) ICODEXTRIN (UNII: 2NX48Z0A9G) DIMETHICONE (UNII: 92RU3N3Y1O) DIPROPYLENE GLYCOL (UNII: E107L85C40) EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ISOHEXADECANE (UNII: 918X1OUF1E) MAGNESIUM ASCORBYL PHOSPHATE (UNII: 0R822556M5) MALTODEXTRIN (UNII: 7CVR7L4A2D) METHYL GLUCETH-20 (UNII: J3QD0LD11P) METHYLPARABEN (UNII: A2I8C7HI9T) PETROLATUM (UNII: 4T6H12BN9U) PHENOXYETHANOL (UNII: HIE492ZZ3T) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) SODIUM CITRATE (UNII: 1Q73Q2JULR) GINGER (UNII: C5529G5JPQ) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11673-353-25 1 in 1 CARTON 01/23/2019 1 99 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 01/23/2019 Labeler - Target Corporation (006961700)