Label: CETIRIZINE HYDROCHLORIDE tablet
- NDC Code(s): 16571-401-10
- Packager: Rising Pharma Holdings, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
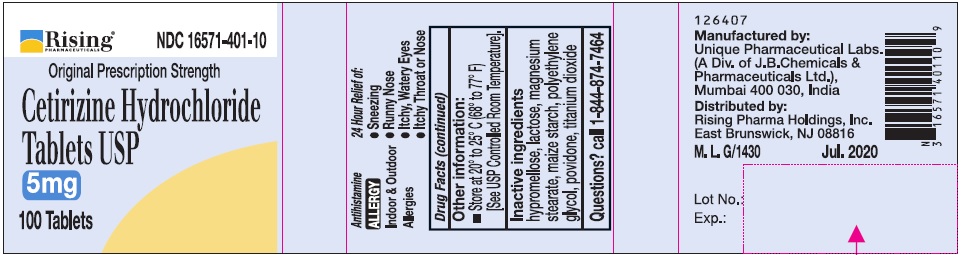
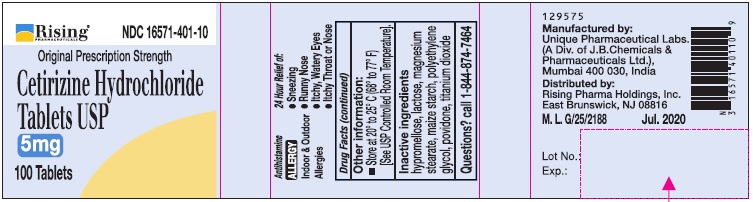
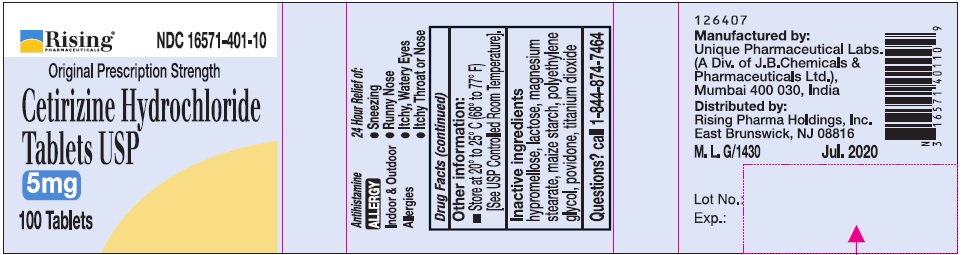
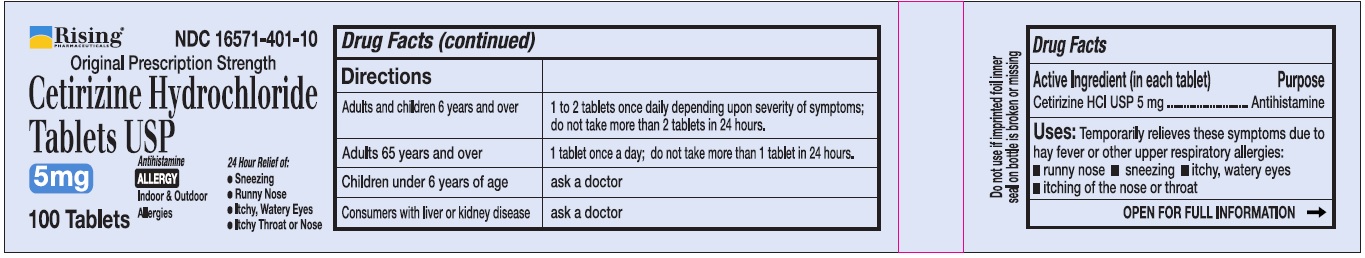
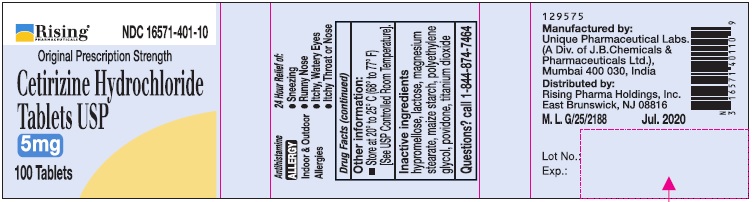
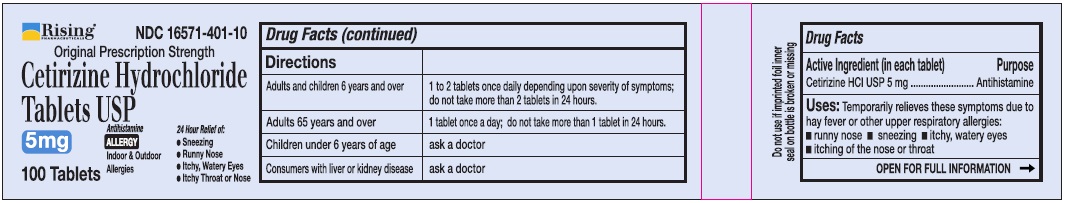
- Active Ingredient (in each tablet)
- Purpose
- Uses
-
Warnings:
Do Not Use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
- drowsines may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinary.
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
-
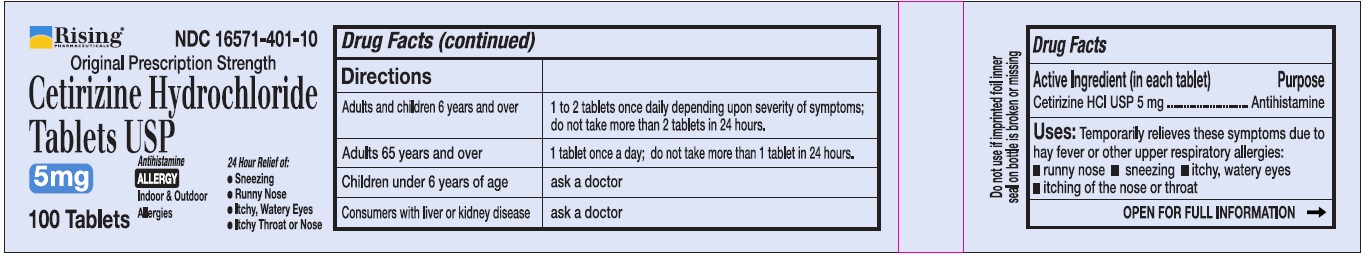
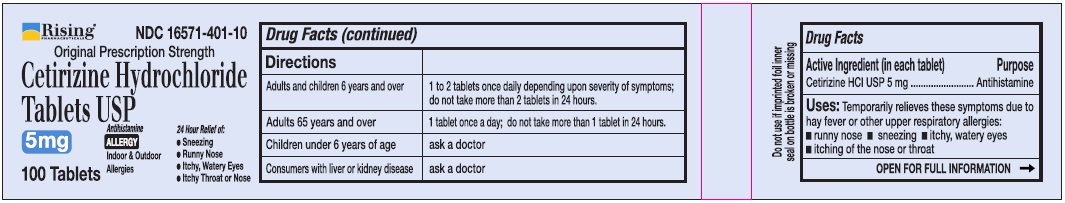
Directions
Adults and children 6 years and over
1 to 2 tablets once daily depending upon severity of symptoms; do not take more than 2 tablets in 24 hours
Adults 65 years and over
1 tablet once a day; do not take more than 1 tablet in 24 hours
Children under 6 years of age
Ask a doctor
Consumers with liver or kidney disease
Ask a doctor
- Other information:
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL-100'S COUNT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:16571-401 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Cetirizine Hydrochloride (UNII: 64O047KTOA) (Cetirizine - UNII:YO7261ME24) Cetirizine Hydrochloride 5 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSES (UNII: 3NXW29V3WO) lactose (UNII: J2B2A4N98G) magnesium stearate (UNII: 70097M6I30) starch, corn (UNII: O8232NY3SJ) polyethylene glycol (UNII: 3WJQ0SDW1A) povidone (UNII: FZ989GH94E) titanium dioxide (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (White) Score no score Shape BULLET (Barrel Shaped) Size 7mm Flavor Imprint Code CTN;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16571-401-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 10/01/2009 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077829 10/01/2009 Labeler - Rising Pharma Holdings, Inc. (116880195) Registrant - Unique Pharmaceutical Laboratories (917165052) Establishment Name Address ID/FEI Business Operations Unique Pharmaceutical Laboratories 650434645 ANALYSIS(16571-401) , MANUFACTURE(16571-401) Establishment Name Address ID/FEI Business Operations Unique Pharmaceutical Laboratories 864354608 analysis(16571-401) , manufacture(16571-401)