Label: CARDIOVASCULAR PROCEDURE KIT- kit
- NHRIC Code(s): 24840-1101-2, 24840-1101-1
- NDC Code(s): 0409-4713-65
- Packager: Centurion Medical Products
- Category: MEDICAL DEVICE
- DEA Schedule: None
- Marketing Status: Exempt device
Drug Label Information
Updated June 4, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Lidocaine Hydrochloride Injection, USP is a sterile, nonpyrogenic solution of lidocaine hydrochloride in water for injection for parenteral administration in various concentrations. Multiple-dose vials contain 0.1% of methylparaben added as preservative. May contain sodium hydroxide and/or hydrochloric acid for pH adjustment. The pH is 6.5 (5.0 to 7.0). Lidocaine is a local anesthetic of the amide type. Lidocaine Hydrochloride, USP is a white powder freely soluable in water. The colecular weight is 288.82.
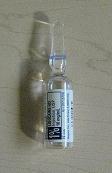
- Cardiovascular Procedure Kit Primary Label
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CARDIOVASCULAR PROCEDURE KIT
cardiovascular procedure kit kitProduct Information Product Type MEDICAL DEVICE Item Code (Source) NHRIC:24840-1101 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:24840-1101-2 10 in 1 CASE 1 NHRIC:24840-1101-1 1 in 1 PACKAGE, COMBINATION Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 AMPULE 5 mL Part 1 of 1 LIDOCAINE HYDROCHLORIDE
lidocaine hydrochloride anhydrous injection, solutionProduct Information Item Code (Source) NDC:0409-4713 Route of Administration INFILTRATION Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE ANHYDROUS (UNII: EC2CNF7XFP) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 7 mg in 1 mL WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0409-4713-65 5 mL in 1 AMPULE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA080408 03/30/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date exempt device OEZ 01/01/2012 Labeler - Centurion Medical Products (017246562) Establishment Name Address ID/FEI Business Operations Centurion Medical Products 017246562 manufacture, repack Establishment Name Address ID/FEI Business Operations Centurion Medical Products 148522279 manufacture, repack Establishment Name Address ID/FEI Business Operations Centurion Medical Products 626660810 manufacture, repack Establishment Name Address ID/FEI Business Operations Hospira, Inc 093132819 manufacture


 MM1
MM1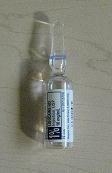 MM1
MM1