Label: CVS ANTIFUNGAL RINGWORM- clotrimazole cream
- NDC Code(s): 59779-514-01
- Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 11, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
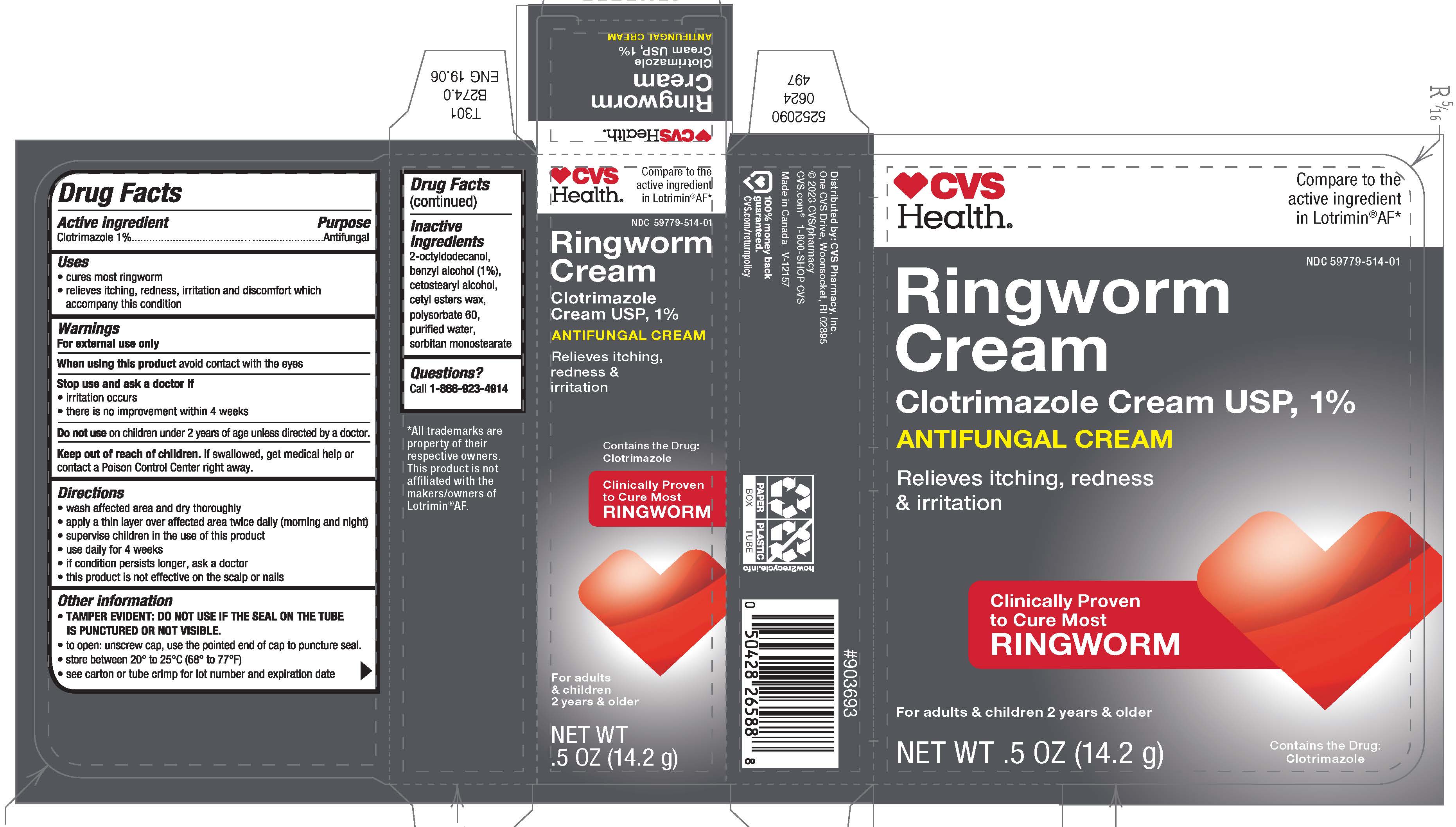
PRINCIPAL DISPLAY PANEL - 14.2 g Tube Carton
CVS
Health ®Compare to the
active ingredient
in Lotrimin ®AF*NDC 59779-514-01
Ringworm
Cream
Clotrimazole Cream USP, 1%
ANTIFUNGAL CREAMRelieves itching, redness
& irritationClinically Proven
to Cure Most
RINGWORMFor adults & children 2 years & older
Contains the Drug:
ClotrimazoleNET WT .5 OZ (14.2 g)
-
INGREDIENTS AND APPEARANCE
CVS ANTIFUNGAL RINGWORM
clotrimazole creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59779-514 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLOTRIMAZOLE (UNII: G07GZ97H65) (CLOTRIMAZOLE - UNII:G07GZ97H65) CLOTRIMAZOLE 1 g in 100 g Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CETYL ESTERS WAX (UNII: D072FFP9GU) OCTYLDODECANOL (UNII: 461N1O614Y) POLYSORBATE 60 (UNII: CAL22UVI4M) WATER (UNII: 059QF0KO0R) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59779-514-01 1 in 1 CARTON 12/28/2012 1 14.2 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M005 06/01/1995 Labeler - CVS Pharmacy (062312574) Establishment Name Address ID/FEI Business Operations Taro Pharmaceuticals Inc. 206263295 manufacture(59779-514)

