Label: NIZORAL- ketoconazole shampoo
- NDC Code(s): 55505-192-33, 55505-192-40, 55505-192-57, 55505-192-59
- Packager: Kramer Laboratories
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated December 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
- WARNINGS
- Do not use
- When using this product
- Stop use and ask a doctor if
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
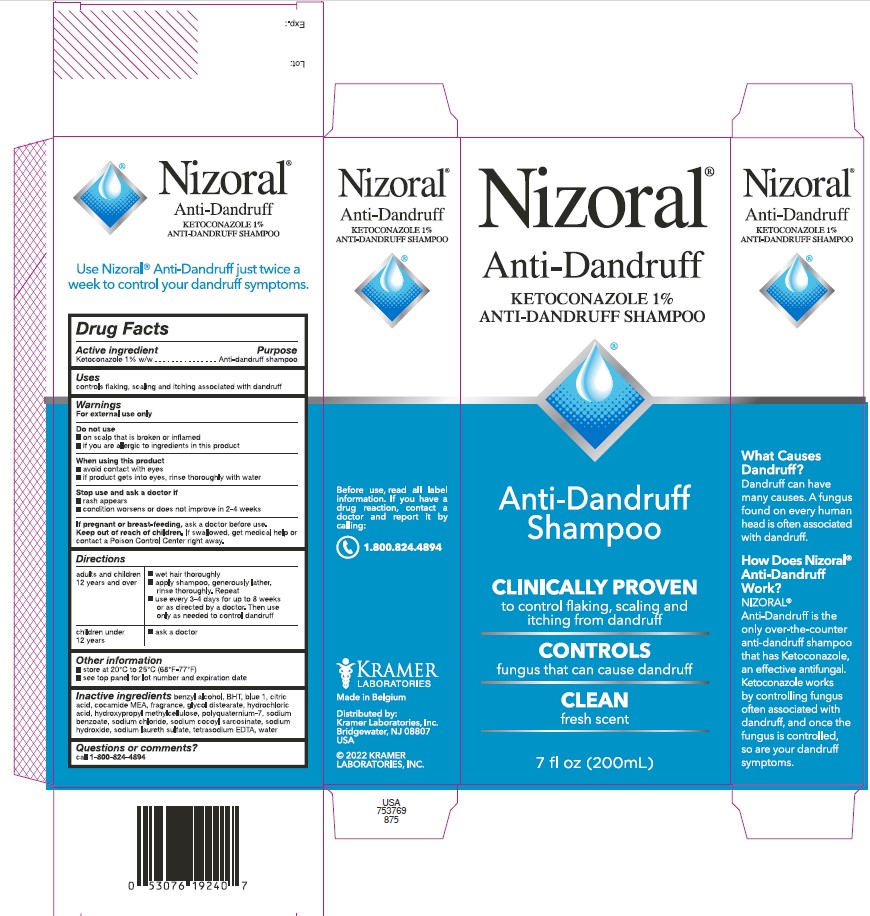
PRINCIPAL DISPLAY PANEL
Nizoral®
Anti-Dandruff
KETOCONAZOLE 1%
ANTI-DANDRUFF SHAMPOO
Anti-Dandruff
Shampoo
CLINICALLY PROVEN
to control flaking, scaling and
itching from dandruff
CONTROLS
fungus that can cause dandruff
CLEAN
fresh scent
7 fl oz (200mL)
What Causes
Dandruff?
Dandruff can have
many causes. A fungus
found on every human
head is often associated
with dandruff.
How Does Nizoral®
Anti-Dandruff
Work?
NIZORAL® is the
only over-the-counter
anti-dandruff shampoo
that has Ketoconazole,
an effective antifungal.
Ketoconazole works
by controlling fungus
often associated with
dandruff, and once the
fungus is controlled,
so are your dandruff
symptoms.
Before use, read all label
information. If you have a
drug reaction, contact a
doctor and report it by
calling:
1.800.824.4894
KRAMER
LABORATORIES
Made in Belgium
Distributed by:
Kramer Laboratories, Inc.
Bridgewater, NJ 08807
USA
© 2022 KRAMER
LABORATORIES, INC.
USA
753769
875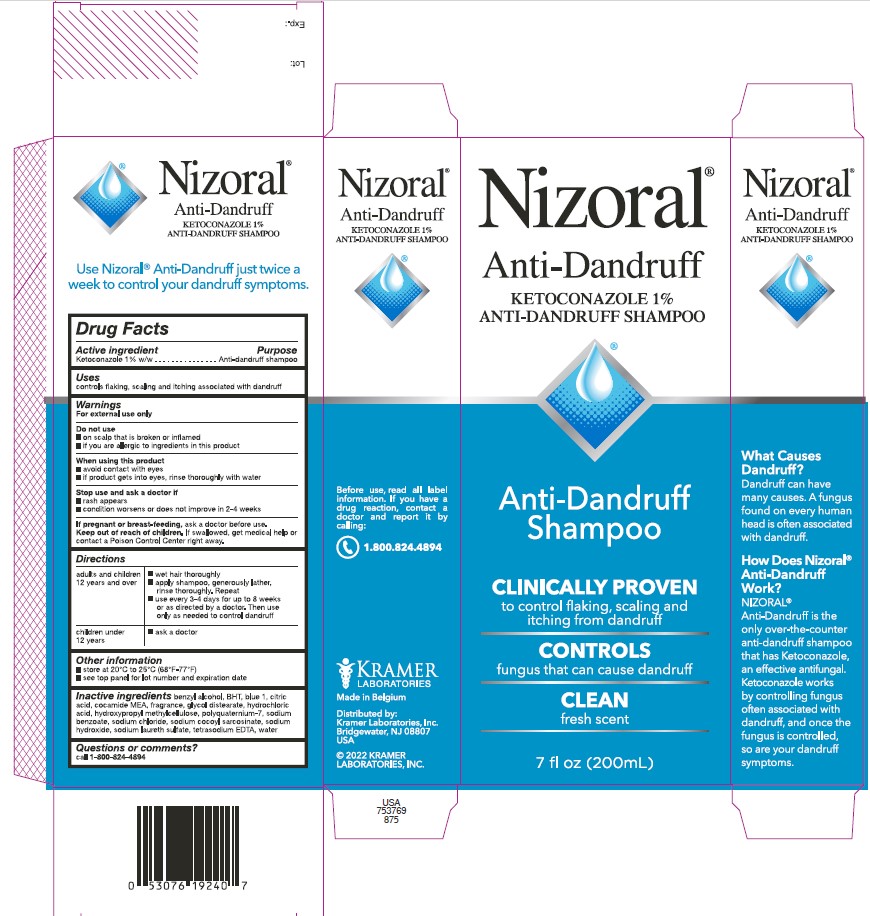
Nizoral®
Anti-Dandruff
KETOCONAZOLE 1%
ANTI-DANDRUFF SHAMPOO
Anti-Dandruff
Shampoo
CLINICALLY PROVEN
to control flaking, scaling and
itching from dandruff
CONTROLS
fungus that can cause dandruff
CLEAN
fresh scent
7 fl oz (200mL)
USA -753767
Dist. by: Kramer Laboratories, Inc., Bridgewater, NJ 08807 USA
© 2022 Kramer Laboratories, Inc.
Made in Belgium
USA - 753768
LOT
EXP

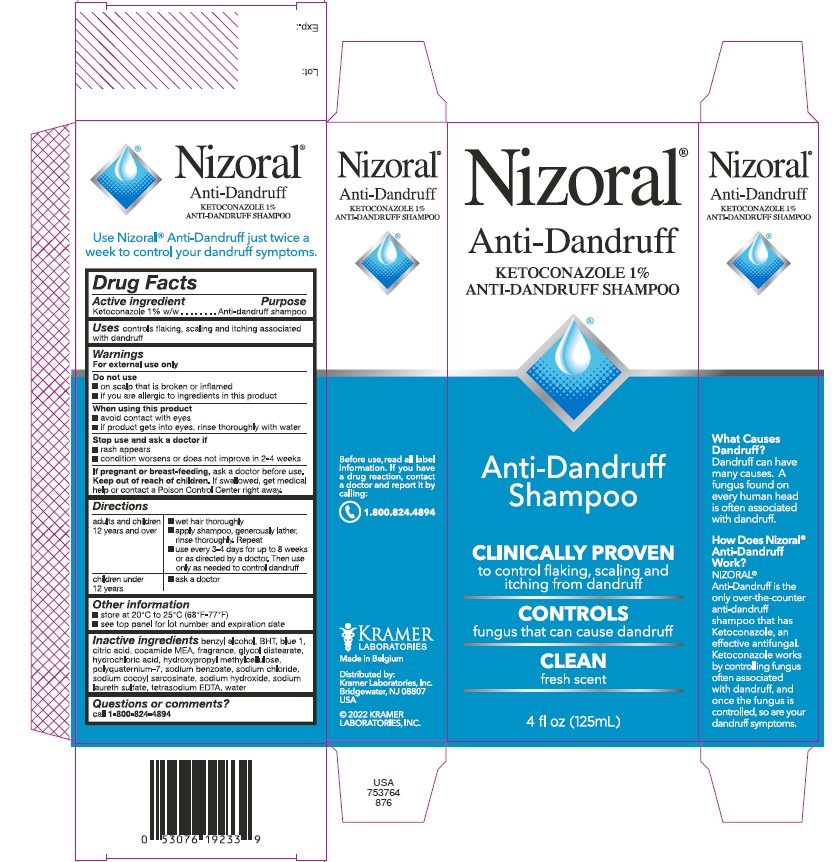
Nizoral®
Anti-Dandruff
KETOCONAZOLE 1%
ANTI-DANDRUFF SHAMPOO
Anti-Dandruff
Shampoo
CLINICALLY PROVEN
to control flaking, scaling and
itching from dandruff
CONTROLS
fungus that can cause dandruff
CLEAN
fresh scent
4 fl oz (125mL)
What Causes
Dandruff?
Dandruff can have
many causes. A
fungus found on
every human head
is often associated
with dandruff.
How Does Nizoral®
Anti-Dandruff
Work?
NIZORAL® is the
only over-the-counter
anti-dandruff
shampoo that has
Ketoconazole, an
effective antifungal.
Ketoconazole works
by controlling fungus
often associated
with dandruff, and
once the fungus is
controlled, so are your
dandruff symptoms.
Before use, read all label
information. If you have
a drug reaction, contact
a doctor and report it by
calling:
1.800.824.4894
KRAMER
LABORATORIES
Made in Belgium
Distributed by:
Kramer Laboratories, Inc.
Bridgewater, NJ 08807
USA
© 2022 KRAMER
LABORATORIES, INC.
USA
753764
876
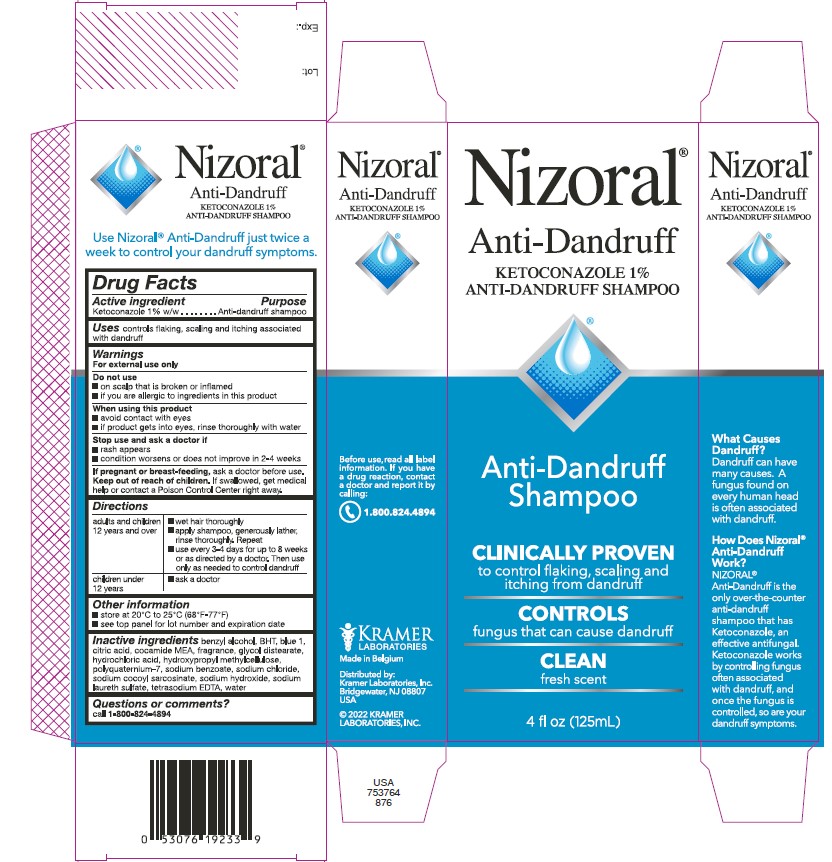
Nizoral®
Anti-Dandruff
KETOCONAZOLE 1%
ANTI-DANDRUFF SHAMPOO
Anti-Dandruff
Shampoo
CLINICALLY PROVEN
to control flaking, scaling and
itching from dandruff
CONTROLS
fungus that can cause dandruff
CLEAN
fresh scent
4 fl oz (125mL)
USA -753765
Dist. by: Kramer Laboratories, Inc. Bridgewater, NJ 08807 USA
© 2022 Kramer Laboratories, Inc.
Made in Belgium
USA - 753766
LOT
EXP

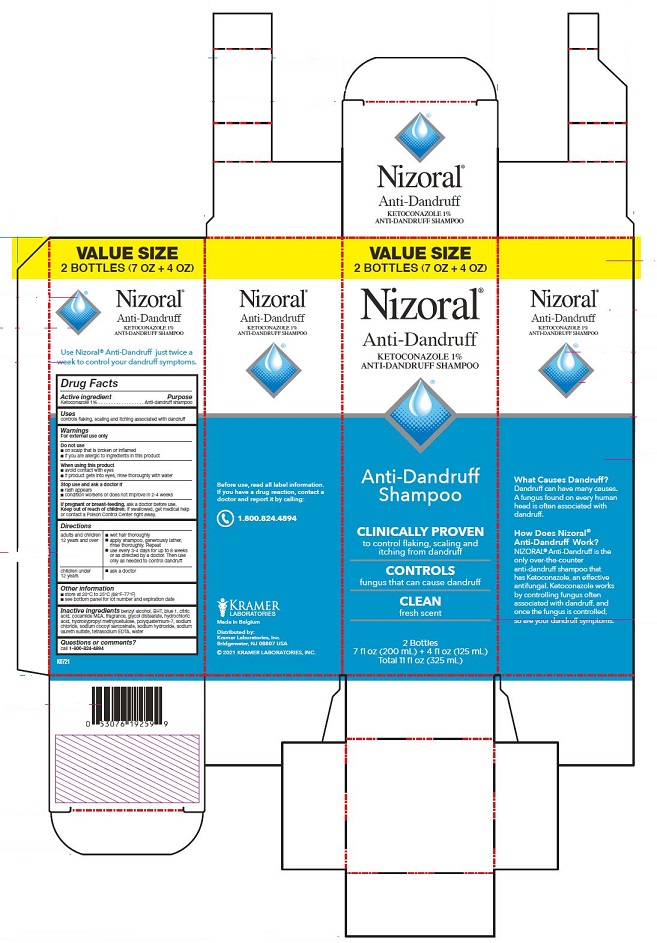
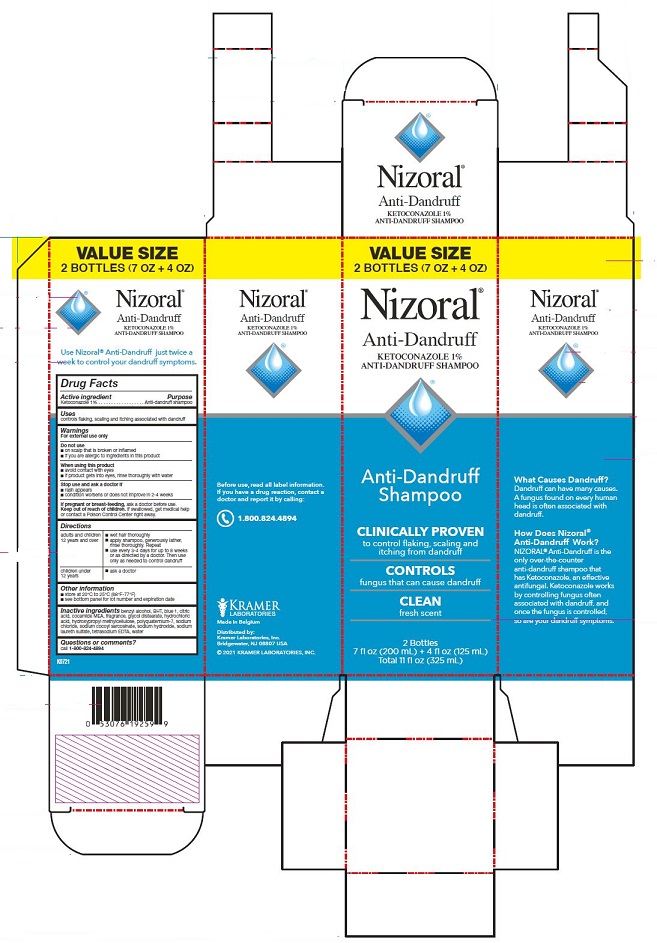
BUY 2 AND SAVE!
Nizoral®
Anti-Dandruff
KETOCONAZOLE 1%
ANTI-DANDRUFF SHAMPOO
Anti-Dandruff
Shampoo
CLINICALLY PROVEN
to control flaking, scaling and
itching from dandruff
CONTROLS
fungus that can cause dandruff
CLEAN
fresh scent
2 Bottles: 7 fl oz (200 mL) each
Total 14 fl oz (400 mL)
Use Nizoral® Anti-Dandruff just
twice a week to control your
dandruff symptoms.
What Causes Dandruff?
Dandruff can have many causes.
A fungus found on every human
head is often associated with
dandruff.
How Does Nizoral®
Anti-Dandruff Work?
NIZORAL® Anti-Dandruff is the
only over-the-counter
anti-dandruff shampoo that
has Ketoconazole, an effective
antifungal. Ketoconazole works
by controlling fungus often
associated with dandruff, and
once the fungus is controlled,
so are your dandruff symptoms.
Before use, read all label
information. If you have a
drug reaction, contact a
doctor and report it by
calling:
1.800.824.4894
KRAMER
LABORATORIES
Made in Belgium
Distributed by:
Kramer Laboratories, Inc.
Bridgewater, NJ 08807 USA
© 2021 KRAMER
LABORATORIES, INC.
value packs xxx
-
INGREDIENTS AND APPEARANCE
NIZORAL
ketoconazole shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55505-192 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Ketoconazole (UNII: R9400W927I) (Ketoconazole - UNII:R9400W927I) Ketoconazole 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength Benzyl Alcohol (UNII: LKG8494WBH) Butylated Hydroxytoluene (UNII: 1P9D0Z171K) Fd&C Blue No. 1 (UNII: H3R47K3TBD) Citric Acid Monohydrate (UNII: 2968PHW8QP) Coco Monoethanolamide (UNII: C80684146D) Glycol Distearate (UNII: 13W7MDN21W) Hydrochloric Acid (UNII: QTT17582CB) Hypromelloses (UNII: 3NXW29V3WO) Polyquaternium-7 (70/30 Acrylamide/Dadmac; 1600000 Mw) (UNII: 0L414VCS5Y) Sodium Chloride (UNII: 451W47IQ8X) Sodium Cocoyl Sarcosinate (UNII: 1R9DUY89CZ) Sodium Hydroxide (UNII: 55X04QC32I) Sodium Laureth-3 Sulfate (UNII: BPV390UAP0) Edetate Sodium (UNII: MP1J8420LU) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55505-192-40 1 in 1 CARTON 01/07/2019 1 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:55505-192-33 1 in 1 CARTON 11/01/2019 2 125 mL in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:55505-192-57 2 in 1 CARTON 11/01/2019 3 1 in 1 CARTON 3 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC:55505-192-59 2 in 1 CARTON 11/01/2019 4 325 mL in 1 CARTON; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020310 01/07/2019 Labeler - Kramer Laboratories (122720675)