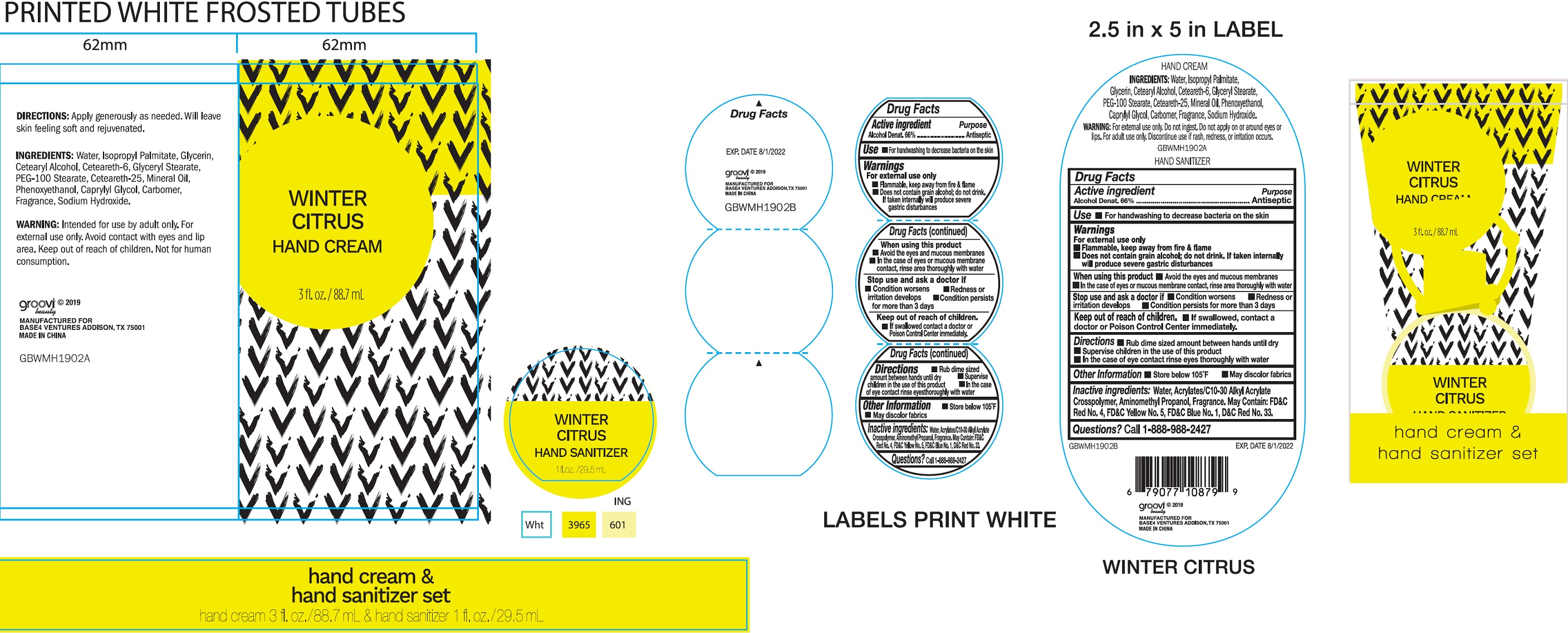
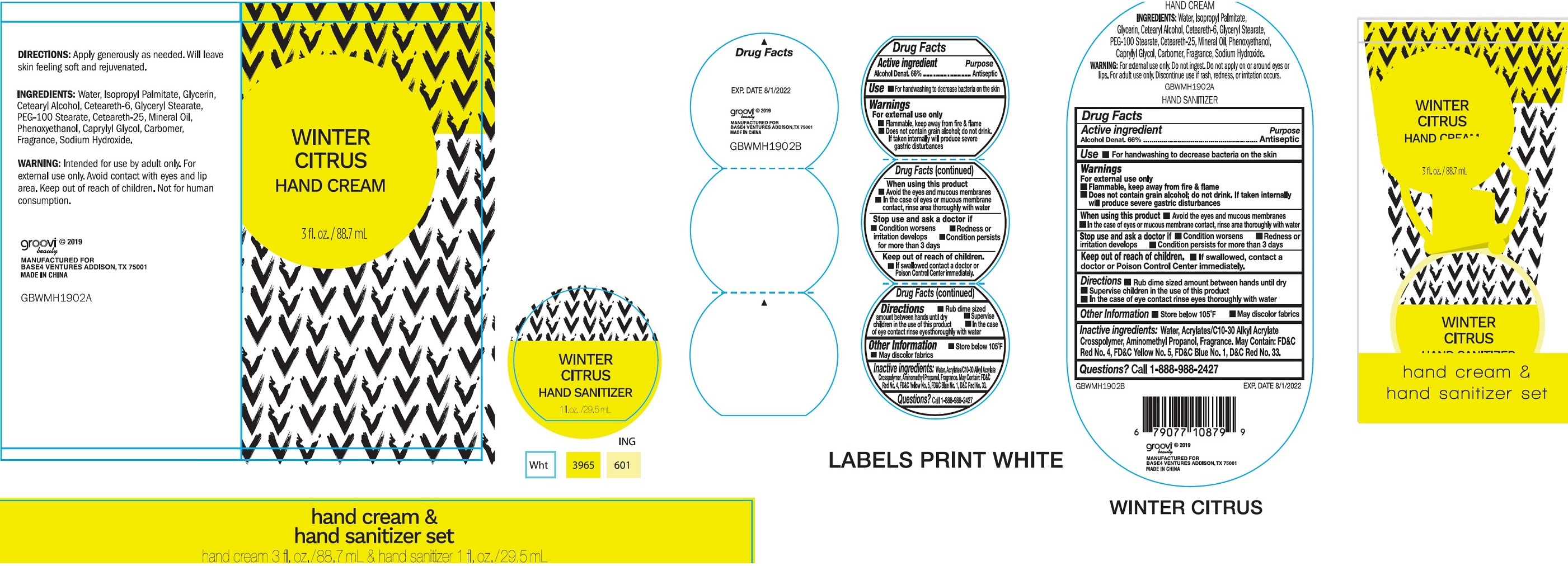
Label: HAND CARE SET WINTER CITRUS- alcohol kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 62651-008-01, 62651-022-00 - Packager: Base4 Ventures, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 26, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Use
-
Warnings
For external use only
- Flammable. Keep away from fire & flame
- Does not contain grain alcohol; do not drink. If taken internally will produce severe gastric disturbances
When using this product
- Avoid the eyes and mucous membranes
- In the case of eyes or mucous membrane contact, rinse area thoroughly with water
- Directions
- Other information
- Inactive ingredients:
- Questions?
- Hand Care Set Winter Citrus (62651-022-00)
- Hand Sanitizer Winter Citrus, 1 oz (62651-008-01)
-
INGREDIENTS AND APPEARANCE
HAND CARE SET WINTER CITRUS
alcohol kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62651-022 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62651-022-00 1 in 1 KIT 10/15/2019 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 29.5 mL Part 1 of 1 HAND SANITIZER WINTER CITRUS
alcohol gelProduct Information Item Code (Source) NDC:62651-008 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 660 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) FD&C RED NO. 4 (UNII: X3W0AM1JLX) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) D&C RED NO. 33 (UNII: 9DBA0SBB0L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62651-008-01 29.5 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 10/15/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 10/15/2019 Labeler - Base4 Ventures, LLC (137316126)