Label: KIDS NASAL ALLERGY RELIEF- cardiospermum halicacabum flowering top, galphimia glauca flowering top, luffa operculata fruit and schoenocaulon officinale seed spray
- NDC Code(s): 59262-245-20
- Packager: Similasan Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated April 23, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
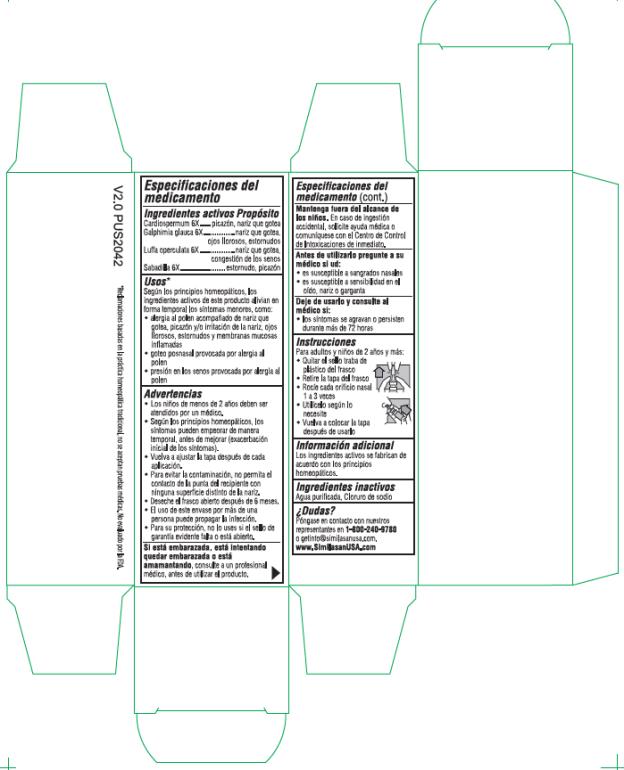
- Active Ingredients
- Purpose
-
Uses*
According to homeopathic principles, the active ingredients in this product temporarily relieve minor symptoms such as:
• allergies accompanied by runny nose, itching and/or burning of the nose, watery eyes, sneezing and swollen mucous membranes (congestion)
• post nasal drip caused by allergies
• sinus pressure caused by allergies
-
Warnings
• Children under 2 years of age should be seen by a physician.
- According to homeopathic principles, symptoms may temporarily worsen before improving (Initial exacerbation of symptoms).
• Replace cap tightly after every use.
• To avoid contamination, do not touch the tip of the container to any surface besides nose.
• Discard open bottle after 6 months.
• The use of this container by more than one person may spread infection.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
- Directions
- Other information
- Inactive Ingredients
- Questions?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
KIDS NASAL ALLERGY RELIEF
cardiospermum halicacabum flowering top, galphimia glauca flowering top, luffa operculata fruit and schoenocaulon officinale seed sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59262-245 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARDIOSPERMUM HALICACABUM FLOWERING TOP (UNII: MZP2508BRR) (CARDIOSPERMUM HALICACABUM FLOWERING TOP - UNII:MZP2508BRR) CARDIOSPERMUM HALICACABUM FLOWERING TOP 6 [hp_X] in 20 mL GALPHIMIA GLAUCA FLOWERING TOP (UNII: 93PH5Q8M7E) (GALPHIMIA GLAUCA FLOWERING TOP - UNII:93PH5Q8M7E) GALPHIMIA GLAUCA FLOWERING TOP 6 [hp_X] in 20 mL LUFFA OPERCULATA FRUIT (UNII: C4MO6809HU) (LUFFA OPERCULATA FRUIT - UNII:C4MO6809HU) LUFFA OPERCULATA FRUIT 6 [hp_X] in 20 mL SCHOENOCAULON OFFICINALE SEED (UNII: 6NAF1689IO) (SCHOENOCAULON OFFICINALE SEED - UNII:6NAF1689IO) SCHOENOCAULON OFFICINALE SEED 6 [hp_X] in 20 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59262-245-20 1 in 1 CARTON 02/15/2019 1 20 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 02/15/2019 Labeler - Similasan Corporation (111566530)