Label: LOPERAMIDE HYDROCHLORIDE capsule
- NDC Code(s): 0615-8087-39
- Packager: NCS HealthCare of KY, LLC dba Vangard Labs
- This is a repackaged label.
- Source NDC Code(s): 0378-2100
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated November 16, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)
WARNING: TORSADES DE POINTES AND SUDDEN DEATH
- ● Cases of Torsades de Pointes, cardiac arrest, and death have been reported with the use of a higher than recommended dosage of loperamide hydrochloride capsules (see WARNINGS and OVERDOSAGE).
- ● Loperamide hydrochloride capsules are contraindicated in pediatric patients less than 2 years of age (see CONTRAINDICATIONS).
- ● Avoid loperamide hydrochloride capsules dosages higher than recommended in adults and pediatric patients 2 years of age and older due to the risk of serious cardiac adverse reactions (see DOSAGE AND ADMINISTRATION).
-
DESCRIPTION
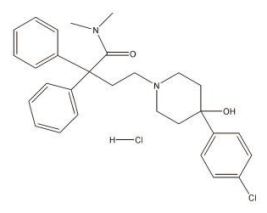
Loperamide hydrochloride, 4-(p-chlorophenyl)-4-hydroxy-N, N-dimethyl-α,α-diphenyl-1-piperidinebutyramide monohydrochloride, is a synthetic antidiarrheal for oral use.
Loperamide hydrochloride is available in 2 mg capsules.
Loperamide hydrochloride capsules, USP contain the following inactive ingredients: black iron oxide, colloidal silicon dioxide, gelatin, lactose monohydrate, magnesium stearate, microcrystalline cellulose, red iron oxide, sodium lauryl sulfate, sodium starch glycolate, titanium dioxide, and yellow iron oxide. In addition, the black imprinting ink also contains black iron oxide, D&C Yellow No. 10 Aluminum Lake, FD&C Blue No. 1 Aluminum Lake, FD&C Blue No. 2 Aluminum Lake, FD&C Red No. 40 Aluminum Lake, propylene glycol, and shellac glaze.
-
CLINICAL PHARMACOLOGY:
Mechanism of Action:
In vitro and animal studies show that loperamide hydrochloride capsules act by slowing intestinal motility and by affecting water and electrolyte movement through the bowel. Loperamide binds to the opiate receptor in the gut wall. Consequently, it inhibits the release of acetylcholine and prostaglandins, thereby reducing propulsive peristalsis, and increasing intestinal transit time. Loperamide increases the tone of the anal sphincter, thereby reducing incontinence and urgency.
Pharmacodynamics:
Loperamide prolongs the transit time of the intestinal contents. It reduces daily fecal volume, increases the viscosity and bulk density, and diminishes the loss of fluid and electrolytes. Tolerance to the antidiarrheal effect has not been observed.
Pharmacokinetics:
Absorption
Plasma concentrations of unchanged drug remain below 2 ng/mL after the intake of a 2 mg capsule of loperamide hydrochloride capsules. Plasma loperamide concentrations are highest approximately 5 hours after administration of the capsule and 2.5 hours after the liquid. The peak plasma concentrations of loperamide were similar for both formulations.
Distribution
Based on literature information, the plasma protein binding of loperamide is about 95%. Loperamide is a P-glycoprotein substrate.
Elimination:
The apparent elimination half-life of loperamide is 10.8 hours with a range of 9.1 to 14.4 hours. Elimination of loperamide mainly occurs by oxidative N-demethylation.
Metabolism
In vitro loperamide is metabolized mainly by cytochrome P450 (CYP450) isozymes, CYP2C8 and CYP3A4, to form- N-demethyl loperamide. In an in vitro study, quercetin (CYP2C8 inhibitor) and ketoconazole (CYP3A4 inhibitor) significantly inhibited the N-demethylation process by 40% and 90%, respectively. In addition, CYP2B6 and CYP2D6 appear to play a minor role in loperamide N-demethylation.
Concomitant use of loperamide hydrochloride capsules with inhibitors of CYP3A4 (e.g., itraconazole) or CYP2C8 (e.g., gemfibrozil) or inhibitors of P-glycoprotein (e.g., quinidine, ritonavir) can increase exposure to loperamide (see PRECAUTIONS: Drug Interactions).
-
INDICATIONS AND USAGE:
Loperamide hydrochloride capsules are indicated for the control and symptomatic relief of acute nonspecific diarrhea in patients 2 years of age and older and of chronic diarrhea in adults associated with inflammatory bowel disease. Loperamide hydrochloride capsules are also indicated for reducing the volume of discharge from ileostomies.
-
CONTRAINDICATIONS:
Loperamide hydrochloride capsules are contraindicated in:
- pediatric patients less than 2 years of age due to the risks of respiratory depression and serious cardiac adverse reactions (see WARNINGS).
- patients with a known hypersensitivity to loperamide hydrochloride or to any of the excipients.
- patients with abdominal pain in the absence of diarrhea.
- patients with acute dysentery, which is characterized by blood in stools and high fever.
- patients with acute ulcerative colitis.
- patients with bacterial enterocolitis caused by invasive organisms including Salmonella, Shigella, and Campylobacter.
- patients with pseudomembranous colitis (e.g., Clostridium difficle) associated with the use of broad-spectrum antibiotics.
-
WARNINGS:
Cardiac Adverse Reactions, Including Torsades de Pointes and Sudden Death:
Cases of prolongation of the QT/QTc interval, Torsades de Pointes, other ventricular arrhythmias, cardiac arrest, some resulting in death, have been reported in adults with use of higher than recommended doses per day of loperamide hydrochloride capsules. Cases include patients who were abusing or misusing loperamide hydrochloride (see OVERDOSAGE and DRUG ABUSE AND DEPENDENCE). Cases of syncope and ventricular tachycardia have been reported in adult patients receiving the recommended dosage of loperamide hydrochloride capsules. Some of these patients were taking other drugs or had other risk factors that may have increased their risk of cardiac adverse reactions. Additionally, postmarketing cases of cardiac arrest, syncope, and respiratory depression have been reported in pediatric patients less than 2 years of age.
Loperamide hydrochloride capsules are contraindicated in pediatric patients less than 2 years of age due to the risks of respiratory depression and serious cardiac adverse reactions. Avoid loperamide hydrochloride capsules dosages higher than recommended in adults and pediatric patients 2 years of age and older due to the risk of serious cardiac adverse reactions (see DOSAGE AND ADMINISTRATION and OVERDOSAGE).
Avoid loperamide hydrochloride capsules in:
- combination with others drugs or herbal products that are known to prolong the QT interval, including Class 1A (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmics, antipsychotics (e.g., chlorpromazine, haloperidol, thioridazine, ziprasidone), antibiotics (e.g., moxifloxacin), or any other drug known to prolong the QT interval (e.g., pentamidine, levomethadyl acetate, methadone).
- patients with risk factors for QT prolongation, including patients with congenital long QT syndrome, with a history of cardiac arrhythmias or other cardiac conditions, elderly patients and those with electrolyte abnormalities.
Dehydration:
Fluid and electrolyte depletion often occur in patients who have diarrhea. In such cases, administration of appropriate fluid and electrolytes is very important. The use of loperamide hydrochloride capsules does not preclude the need for appropriate fluid and electrolyte therapy.
Gastrointestinal Disorders:
In general, loperamide hydrochloride capsules should not be used when inhibition of peristalsis is to be avoided due to the possible risk of significant sequelae including ileus, megacolon and toxic megacolon. Loperamide hydrochloride capsules must be discontinued promptly when constipation, abdominal distention or ileus develop.
Treatment of diarrhea with loperamide hydrochloride capsules is only symptomatic. Whenever an underlying etiology can be determined, specific treatment should be given when appropriate (or when indicated).
Patients with AIDS treated with loperamide hydrochloride capsules for diarrhea should have therapy stopped at the earliest signs of abdominal distention. There have been isolated reports of toxic megacolon in AIDS patients with infectious colitis from both viral and bacterial pathogens treated with loperamide hydrochloride.
Variability in Pediatric Response:
Loperamide hydrochloride capsules should be used with special caution in pediatric patients because of the greater variability of response in this age group. Dehydration, particularly in pediatric patients less than 6 years of age, may further influence the variability of response to loperamide hydrochloride capsules. Loperamide hydrochloride capsules are contraindicated in pediatric patients less than 2 years of age due to the risks of respiratory depression and serious cardiac adverse reactions.
-
PRECAUTIONS:
General:
Allergic Reactions:
Extremely rare allergic reactions including anaphylaxis and anaphylactic shock have been reported.
Hepatic Impairment:
The effects of hepatic impairment on the pharmacokinetics of loperamide have not been studied. Use loperamide hydrochloride capsules with caution in such patients because the systemic exposure to loperamide may be increased due to reduced metabolism. Monitor patients with hepatic impairment closely for signs of central nervous system (CNS) toxicity.
Renal Impairment:
No pharmacokinetic data are available in patients with renal impairment. Since it has been reported that the majority of the drug is metabolized and metabolites or the unchanged drug are excreted mainly in the feces, dosage adjustments in patients with renal impairment are not required.
Geriatric Use:
No formal studies have been conducted to evaluate the pharmacokinetics of loperamide in elderly subjects. However, in two studies that enrolled elderly patients, there were no major differences in the drug disposition in elderly patients with diarrhea relative to young patients.
In general, elderly patients may be more susceptible to drug-associated effects on the QT interval. Avoid loperamide hydrochloride capsules in elderly patients taking drugs that can result in prolongation of the QT interval (for example, Class IA or III antiarrhythmics) or in patients with risk factors for Torsades de Pointes (see WARNINGS).
Information for Patients:
Advise patients:
- to take loperamide hydrochloride capsules at the prescribed dosage. Use of a higher than prescribed dosage is not recommended (see WARNINGS). Report to a healthcare facility if you or someone you are caring for taking loperamide hydrochloride capsules experiences a fainting episode, a rapid or irregular heartbeat or becomes unresponsive.
- with acute diarrhea, that if clinical improvement is not observed in 48 hours, discontinue loperamide hydrochloride capsules and contact their healthcare provider.
- to contact their healthcare provider if they see blood in their stools, or if they develop a fever or abdominal distention.
- to use caution when driving a car or operating machinery, as tiredness, dizziness, or drowsiness may occur in the setting of diarrheal syndromes treated with loperamide hydrochloride capsules (see ADVERSE REACTIONS).
- to tell their healthcare provider about all the medications they are taking, including prescription and over-the-counter medications, vitamins and herbal supplements, especially if they take Class 1A (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmics, antipsychotics (e.g., chlorpromazine, haloperidol, thioridazine, ziprasidone), antibiotics (e.g., moxifloxacin), or any other drug known to prolong the QT interval (e.g., pentamidine, levomethadyl acetate, methadone).
Drug Interactions
Effects of Other Drugs on Loperamide:
Concomitant use of loperamide hydrochloride capsules with inhibitors of CYP3A4 (e.g., itraconazole) or CYP2C8 (e.g., gemfibrozil) or inhibitors of P-glycoprotein (e.g., quinidine, ritonavir) can increase exposure to loperamide. The increased systemic exposure to loperamide may increase a risk for cardiac adverse reactions especially in patients who are taking multiple CYP enzyme inhibitors, or in patients with underlying cardiac conditions (see WARNINGS). Monitor patients for cardiac adverse reactions.
CYP3A4 Inhibitors:
Itraconazole:
Concomitant administration of multiple doses of 100 mg itraconazole twice daily, an inhibitor of both CYP3A4 and P-glycoprotein, with a single 4 mg dose of loperamide hydrochloride increased the peak plasma concentration and the systemic exposure to loperamide by 2.9-fold and 3.8-fold, respectively.
CYP2C8 Inhibitors:
Gemfibrozil:
When a single 4 mg dose of loperamide hydrochloride was coadministered with 600 mg gemfibrozil, a strong inhibitor of CYP2C8, on day 3 of a 5-day treatment with gemfibrozil twice daily, the mean peak plasma concentration and the systemic exposure to loperamide was increased by 1.6-fold and 2.2-fold, respectively.
CYP3A4 and CYP2C8 Inhibitors:
When multiple doses of both 100 mg itraconazole and 600 mg gemfibrozil twice daily were administered with a single 4 mg dose of loperamide hydrochloride, the mean peak plasma concentration and the systemic exposure to loperamide was increased by 4.2-fold and 12.6-fold, respectively.
P-glycoprotein Inhibitors:
Concomitant administration of a 16 mg single dose of loperamide hydrochloride with a 600 mg single dose of quinidine or ritonavir, both of which are P-glycoprotein inhibitors, resulted in a 2- to 3-fold increase in loperamide plasma concentrations. Due to the potential for enhanced CNS adverse reactions when loperamide is coadministered with quinidine and with ritonavir, caution should be exercised when loperamide hydrochloride capsules are administered at the recommended dosages (2 mg, up to 16 mg maximum daily dose) with P-glycoprotein inhibitors.
Effects of Loperamide on Other Drugs:
Saquinavir:
When a single 16 mg dose of loperamide hydrochloride is coadministered with a 600 mg single dose of saquinavir, loperamide decreased saquinavir exposure by 54%, which may be of clinical relevance due to reduction of therapeutic efficacy of saquinavir. The effect of saquinavir on loperamide is of less clinical significance. Therefore, when loperamide hydrochloride capsules are given with saquinavir, the therapeutic efficacy of saquinavir should be closely monitored.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
In an 18-month rat study with oral loperamide hydrochloride doses up to 40 mg/kg/day (21 times the maximum human dose of 16 mg/day, based on a body surface area comparison), there was no evidence of carcinogenesis.
Loperamide was not genotoxic in the Ames test, the SOS chromotest in E. coli, the dominant lethal test in female mice, or the mouse embryo cell transformation assay.
Fertility and reproductive performance was evaluated in rats using oral doses of 2.5 mg/kg/day, 10 mg/kg/day, and 40 mg/kg/day (females only) in a second study. Oral administration of 20 mg/kg/day (approximately 11 times the human dose based on a body surface area comparison) and higher, produced a strong impairment of female fertility. Treatment of female rats with up to 10 mg/kg/day (approximately 5 times the human dose based on a body surface area comparison) had no effect on fertility. Treatment of male rats with oral doses of 40 mg/kg/day (approximately 21 times the human dose based on a body surface area comparison) produced impairment of male fertility, whereas administration of up to 10 mg/kg/day (approximately 5 times the human dose based on a body surface area comparison) had no effect.
Pregnancy:
Teratogenic Effects. Pregnancy Category C:
Teratology studies have been performed in rats using oral loperamide hydrochloride doses of 2.5 mg/kg/day, 10 mg/kg/day, and 40 mg/kg/day, and in rabbits using oral doses of 5 mg/kg/day, 20 mg/kg/day, and 40 mg/kg/day. These studies have revealed no evidence of impaired fertility or harm to the fetus at doses up to 10 mg/kg/day in rats (5 times the human dose based on body surface area comparison) and 40 mg/kg/day in rabbits (43 times the human dose based on body surface area comparison). Treatment of rats with oral doses of 40 mg/kg/day (21 times the human dose based on a body surface area comparison) produced marked impairment of fertility. The studies produced no evidence of teratogenic activity. There are no adequate and well controlled studies in pregnant women. Loperamide hydrochloride capsules should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers:
Small amounts of loperamide may appear in human breast milk. Therefore, loperamide hydrochloride capsules are not recommended during breast-feeding.
Pediatric Use:
Loperamide hydrochloride capsules are contraindicated in pediatric patients less than 2 years of age due to the risks of respiratory depression and serious cardiac adverse reactions (see CONTRAINDICATIONS). Postmarketing cases of cardiac arrest, syncope, and respiratory depression have been reported in pediatric patients less than 2 years of age (see WARNINGS). Pediatric patients may be more sensitive to CNS effects, such as altered mental status, somnolence, and respiratory depression, than adults. There have been rare reports of paralytic ileus associated with abdominal distention. Most of these reports occurred in the setting of acute dysentery, overdose, and with pediatric patients less than two years of age.
Loperamide hydrochloride capsules should be used with special caution in pediatric patients because of their greater variability of response (see WARNINGS). Dehydration, particularly in pediatric patients less than 6 years of age, may further influence the variability of response to loperamide hydrochloride capsules.
The safety and effectiveness of loperamide hydrochloride capsules in pediatric patients with chronic diarrhea have not been established. Although loperamide hydrochloride capsules have been studied in a limited number of pediatric patients with chronic diarrhea; the therapeutic dose for the treatment of chronic diarrhea in a pediatric population has not been established.
In case of accidental overdosage of loperamide hydrochloride capsules by pediatric patients, see OVERDOSAGE for suggested treatment.
-
ADVERSE REACTIONS:
Clinical Trial Experience:
The adverse effects reported during clinical investigations of loperamide hydrochloride capsules are difficult to distinguish from symptoms associated with the diarrheal syndrome. Adverse experiences recorded during clinical studies with loperamide hydrochloride capsules were generally of a minor and self-limiting nature. They were more commonly observed during the treatment of chronic diarrhea.
The adverse events reported are summarized irrespective of the causality assessment of the investigators.
1) Adverse events from 4 placebo-controlled studies in patients with acute diarrhea
The adverse events with an incidence of 1.0% or greater, which were reported at least as often in patients on loperamide hydrochloride as on placebo, are presented in the table below.
Acute Diarrhea Loperamide
HydrochloridePlacebo No. of treated patients 231 236 Gastrointestinal AE%
Constipation
2.6%
0.8%
The adverse events with an incidence of 1.0% or greater, which were more frequently reported in patients on placebo than on loperamide hydrochloride, were: dry mouth, flatulence, abdominal cramp and colic.
2) Adverse events from 20 placebo-controlled studies in patients with chronic diarrhea
The adverse events with an incidence of 1.0% or greater, which were reported at least as often in patients on loperamide hydrochloride as on placebo, are presented in the table below.
Chronic Diarrhea Loperamide
HydrochloridePlacebo No. of treated patients 285 277 Gastrointestinal AE%
Constipation
5.3%
0.0%
Central and peripheral nervous system AE%
Dizziness
1.4%
0.7%
The adverse events with an incidence of 1.0% or greater, which were more frequently reported in patients on placebo than on loperamide hydrochloride were: nausea, vomiting, headache, meteorism, abdominal pain, abdominal cramp and colic.
3) Adverse events from 76 controlled and uncontrolled studies in patients with acute or chronic diarrhea
The adverse events with an incidence of 1.0% or greater in patients from all studies are given in the table below.
Acute
DiarrheaChronic
DiarrheaAll
Studies*No. of treated patients 1913 1371 3740 - *
- All patients in all studies, including those in which it was not specified if the adverse events occurred in patients with acute or chronic diarrhea.
Gastrointestinal AE%
Nausea
0.7%
3.2%
1.8%
Constipation
1.6%
1.9%
1.7%
Abdominal cramps
0.5%
3.0%
1.4%
Postmarketing Experience:
The following adverse events have been reported:
Cardiac Disorders: QT/QTc-interval prolongation, Torsades de Pointes, other ventricular arrhythmias, cardiac arrest, syncope, and death (see WARNINGS and OVERDOSAGE).
Skin and Subcutaneous Tissue Disorders: Rash, pruritus, urticaria, angioedema, and extremely rare cases of bullous eruption including erythema multiforme, Stevens-Johnson syndrome and Toxic Epidermal Necrolysis have been reported with use of loperamide hydrochloride capsules.
Immune System Disorders: Isolated occurrences of allergic reactions and in some cases severe hypersensitivity reactions including anaphylactic shock and anaphylactoid reactions have been reported with the use of loperamide hydrochloride capsules.
Gastrointestinal Disorders: Dry mouth, abdominal pain, distention or discomfort, nausea, vomiting, flatulence, dyspepsia, constipation, paralytic ileus, megacolon; including toxic megacolon (see CONTRAINDICATIONS and WARNINGS).
Renal and Urinary Disorders: Urinary retention
Nervous System Disorders: Drowsiness, dizziness
General Disorders and Administrative Site Conditions: TirednessA number of the adverse events reported during the clinical investigations and postmarketing experience with loperamide are frequent symptoms of the underlying diarrheal syndrome (abdominal pain/discomfort, nausea, vomiting, dry mouth, tiredness, drowsiness, dizziness, constipation, and flatulence). These symptoms are often difficult to distinguish from undesirable drug effects.
-
DRUG ABUSE AND DEPENDENCE:
Abuse
Loperamide is a mu-opioid agonist. A human abuse potential study of loperamide hydrochloride at single doses up to 60 mg (3.75 times the recommended maximum adult dosage of 16 mg per day) was compared, in a double-blind cross-over design using nine subjects who had been active opiate users, to a threshold dose of codeine sulfate at 120 mg (96 mg base) or placebo. This resulted in one subject (11%) feeling a drug on placebo and identifying it as "dope" (heroin) and liking it slightly. Codeine was felt by 56% of subjects and identified as "dope" by 44%. Loperamide was felt by 44% of subjects and identified as "dope" by 11% and possibly dope mixed with some other kind of drug by another 22%. Loperamide abuse and misuse have been reported, especially at doses of 60 mg or greater. Loperamide can have greater CNS opioid effects at higher doses or with coadministration of drugs that increase systemic exposure and/or increase CNS penetration of loperamide (through inhibition of the CYP450 enzyme system or inhibition of P-glycoprotein). Loperamide is primarily being misused for relief from opioid withdrawal, and abused by a few users who obtain some (reportedly mild-moderate) level of euphoria.
Dependence:
In animals, parenteral administration of loperamide hydrochloride can cause physical dependence, cross-tolerance to opioids, and all the other pharmacologic effects typical of mu-opioid agonists.
Studies in morphine-dependent monkeys demonstrated that loperamide hydrochloride at doses above those recommended for humans prevented signs of morphine withdrawal.
-
OVERDOSAGE:
The use of higher than recommended loperamide hydrochloride capsules doses may result in life-threatening cardiac, CNS and respiratory adverse reactions.
If over-exposure occurs, call your Poison Control Center at 1-800-222-1222 for current information on the management of poisoning or overdosage.
Cardiac Effects:
Symptoms:
Cases of overdosage with loperamide hydrochloride (chronic ingestion of doses ranging from 70 mg to 1600 mg daily; 4 to 100 times the recommended dose) have resulted in life-threatening cardiac adverse reactions, including QT/QTc and QRS interval prolongation, Torsades de Pointes, Brugada syndrome and other ventricular arrhythmias, syncope, cardiac arrest, and death. Cases include patients who were abusing (using supratherapeutic doses in place of opioids to induce euphoria) or misusing (taking higher than recommended doses to control diarrhea or to prevent opioid withdrawal) loperamide. The following are representative cases that included cardiac adverse reactions:
- 25 year old abused loperamide and presented to the hospital on multiple occasions with symptoms of syncope, nausea, vomiting, bradycardia, hypotensive shock. The patient also experienced ventricular tachycardia, a prolonged QTc of 527 ms and QRS interval of 170 ms, frequent premature ventricular contractions, and subsequent cardiac arrest and death (elevated loperamide blood concentration of 32 ng/ml).
- 54 year old misused loperamide hydrochloride (up to 144 mg per day) as a self-treatment for chronic diarrhea for over 2 years. Signs of cardiac toxicity included syncope, prolonged QT of 500 ms, sinus arrest with junctional escape rhythm, and polymorphic ventricular tachycardia, which required cardioversion and implantable cardioverter-defibrillator (ICD) management.
- 26 year old, with prior opioid abuse, presented to the hospital with recurrent syncope and developed Torsades de Pointes requiring electrical cardioversion. An ECG revealed a sinus rhythm with a heart rate of 85 bpm and a markedly prolonged QTc interval of greater than 700 ms. The patient reported ingesting 100 mg to 250 mg of loperamide hydrochloride with 400 mg of cimetidine daily for several months to simulate the euphoric sensation associated with opioids.
Management:
Consider loperamide as a possible cause of cardiac arrhythmias in patients who may have a history of opioid abuse or recent ingestion of unknown drugs and in the differential diagnosis of unstable arrhythmias, prolonged QTc or QRS intervals, and Torsades de Pointes.
If loperamide-induced cardiac toxicity is suspected, promptly discontinue the drug and initiate therapy to manage and prevent cardiac arrhythmias and serious outcomes.
In many cases of loperamide overdosage, anti-arrhythmic medications (e.g., magnesium sulfate) were ineffective in resolving the arrhythmias and preventing further episodes of Torsades de Pointes. Electrical cardioversion and overdrive pacing, and isoproterenol continuous infusion were reported to manage QTc prolongation in the setting of overdose.
CNS and Respiratory Depression:
Symptoms:
Cases of loperamide overdose (including relative overdose due to hepatic dysfunction), may cause opioid toxic effects including CNS depression (e.g., altered mental status, stupor, coordination disorders, somnolence, miosis, muscular hypertonia, respiratory depression), hypotension, urinary retention, and paralytic ileus. Pediatric patients may be more sensitive to CNS effects, including respiratory depression, than adults.
Management:
Loperamide non-cardiac arrhythmia overdosages should be treated as opioid overdosages. Naloxone may reverse the opioid-related toxicity, including CNS and respiratory depression, and hypotension, associated with loperamide overdosage.
In adults and pediatric patients, naloxone may be administered intravenously. Appropriate doses of naloxone, via intranasal, intramuscular, intraosseus, or subcutaneous administration may be necessary if the intravenous route is not available. If the desired degree of opioid-related toxicity counteraction and improvement are not obtained, naloxone may be repeated at two- to three-minute intervals. If no response in opioid-related effects is observed after naloxone has been administered, then diagnosis of opioid-induced toxicity should be questioned.
Refer to the naloxone prescribing information for complete information on initial and subsequent dosages.
For patients whose adverse reactions are responsive to naloxone, monitor vital signs, neurologic and cardiopulmonary status for recurrence of opioid overdose symptoms for at least 24 hours after the last dose of naloxone, due to the prolonged intestinal retention of loperamide and the short duration (one to three hours) of naloxone. Patients with severe CNS or respiratory depression, and those who require multiple doses of naloxone to reverse symptoms, should be admitted to the hospital and may require intensive care.
-
DOSAGE AND ADMINISTRATION:
Loperamide hydrochloride capsules are contraindicated in pediatric patients less than 2 years of age due to the risks of respiratory depression and serious cardiac adverse reactions (see CONTRAINDICATIONS).
Avoid loperamide hydrochloride capsules dosages higher than recommended in adult or pediatric patients 2 years of age and older due to the risk of serious cardiac adverse reactions (see WARNINGS and OVERDOSAGE).
(1 capsule = 2 mg): Patients should receive appropriate fluid and electrolyte replacement as needed.
Acute Diarrhea:
Adults and Pediatric Patients 13 Years and Older:
The recommended initial dose is 4 mg (two capsules) followed by 2 mg (one capsule) after each unformed stool. The maximum daily dose is 16 mg (eight capsules). Clinical improvement is usually observed within 48 hours.
Pediatric Patients 2 to 12 Years of Age:
In pediatric patients 2 to 5 years of age (20 kg or less), the non-prescription liquid formulation of loperamide (1 mg/5 mL) should be used; for ages 6 to 12, either loperamide hydrochloride capsules or the non-prescription liquid formulation of loperamide may be used. For pediatric patients 2 to 12 years of age, the following schedule for capsules or liquid will usually fulfill initial dosage requirements:
Recommended First Day Dosage Schedule
Two to five years (13 kg to 20 kg): 1 mg three times daily (3 mg total daily dosage)
Six to eight years (20 kg to 30 kg): 2 mg twice daily (4 mg total daily dosage)
Eight to twelve years (greater than 30 kg): 2 mg three times daily (6 mg total daily dosage)
Recommended Subsequent Daily Dosage
Following the first treatment day, it is recommended that subsequent loperamide hydrochloride capsules doses (1 mg/10 kg body weight) be administered only after a loose stool. Total daily dosage should not exceed recommended dosages for the first day.
Chronic Diarrhea
Adults:
The recommended initial dose is 4 mg (two capsules) followed by 2 mg (one capsule) after each unformed stool until diarrhea is controlled, after which the dosage of loperamide hydrochloride capsules should be reduced to meet individual requirements. When the optimal daily dosage has been established, this amount may then be administered as a single dose or in divided doses.
The average daily maintenance dosage in clinical trials was 4 mg to 8 mg (two to four capsules per day). The maximum daily dosage is 16 mg (eight capsules per day). If clinical improvement is not observed after treatment with 16 mg per day for at least 10 days, symptoms are unlikely to be controlled by further administration. Loperamide hydrochloride capsules administration may be continued if diarrhea cannot be adequately controlled with diet or specific treatment.
Elderly:
No formal pharmacokinetic studies were conducted in elderly subjects. However, there were no major differences reported in the drug disposition in elderly patients with diarrhea relative to young patients. No dose adjustment is required for the elderly.
In general, elderly patients may be more susceptible to drug-associated effects of the QT interval. Avoid loperamide hydrochloride capsules in elderly patients taking drugs that can result in prolongation of the QT interval (for example, Class IA or III antiarrhythmics) or in patients with risk factors for Torsades de Pointes (see WARNINGS).
Renal Impairment:
No pharmacokinetic data are available in patients with renal impairment. Since the metabolites and the unchanged drug are mainly excreted in the feces, no dosage adjustment is required for patients with renal impairment (see PRECAUTIONS).
Hepatic Impairment:
The pharmacokinetics of loperamide have not been studied in patients with hepatic impairment. Use loperamide hydrochloride capsules with caution in such patients because the systemic exposure may be increased due to reduced metabolism (see PRECAUTIONS).
-
HOW SUPPLIED:
Loperamide Hydrochloride Capsules, USP are available containing 2 mg of loperamide hydrochloride, USP.
The capsule is a hard-shell gelatin capsule with a light brown opaque cap and a light brown opaque body filled with white to off-white powder blend. The capsule is axially printed with MYLAN over 2100 in black ink on both the cap and the body. They are available as follows:
NDC 0615-8087-39
blistercards of 30 capsulesStore at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Revised: 9/2022
VPL:LOP:R1 - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LOPERAMIDE HYDROCHLORIDE
loperamide hydrochloride capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0615-8087(NDC:0378-2100) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 2 mg Inactive Ingredients Ingredient Name Strength FERROSOFERRIC OXIDE (UNII: XM0M87F357) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) FERRIC OXIDE RED (UNII: 1K09F3G675) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) Product Characteristics Color brown (light brown opaque) Score no score Shape CAPSULE Size 14mm Flavor Imprint Code MYLAN;2100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0615-8087-39 30 in 1 BLISTER PACK; Type 0: Not a Combination Product 03/28/2017 10/31/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA072741 09/18/1991 10/31/2024 Labeler - NCS HealthCare of KY, LLC dba Vangard Labs (050052943) Establishment Name Address ID/FEI Business Operations NCS HealthCare of KY, LLC dba Vangard Labs 050052943 repack(0615-8087)


