Label: hydrocodone bitartrate and acetaminophen- Hydrocodone Bitartrate and Acetaminophen liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 58177-909-07 - Packager: ETHEX Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CIII
Drug Label Information
Updated March 12, 2007
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- N/A - Section Title Not Found In Database
-
DESCRIPTION
Hydrocodone bitartrate and acetaminophen is supplied in liquid form for oral administration.
WARNING: May be habit forming (see PRECAUTIONS, Information for Patients, and DRUG ABUSE AND DEPENDENCE).
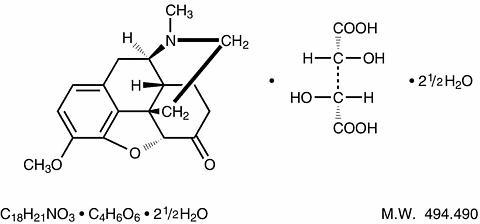
Hydrocodone bitartrate is an opioid analgesic and antitussive and occurs as fine, white crystals or as a crystalline powder. It is affected by light. The chemical name is 4,5α-epoxy-3-methoxy-17-methylmorphinan-6-one tartrate (1:1) hydrate (2:5). It has the following structural formula:
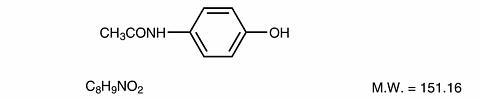
Acetaminophen, 4'-hydroxyacetanilide, a slightly bitter, white, odorless, crystalline powder, is a non-opiate, non-salicylate analgesic and antipyretic. It has the following structural formula:
Hydrocodone bitartrate and acetaminophen oral solution contains:
Per 5mL Per 15mL Hydrocodone Bitartrate………. 2.5 mg 7.5 mg Acetaminophen.. 167 mg 500 mg Alcohol…………. 7% 7% In addition, the liquid contains the following inactive ingredients: ammoniated glyyerrhizin, dibasic sodium phosphate, citric acid, ethyl maltol, polyethylene glycol, propylene glycol, purified water, saccharin sodium, sodium citrate, sodium benzoate, sodium gluconate, sorbitol solution, with D&C Yellow No. 10 and FD&C Yellow No. 6 as coloring, and natural and artificial flavoring. Hydrochloric acid or sodium hydroxide may be added to adjust the pH.
-
CLINICAL PHARMACOLOGY
Hydrocodone is a semisynthetic narcotic analgesic and antitussive with multiple actions qualitatively similar to those of codeine. Most of these involve the central nervous system and smooth muscle. The precise mechanism of action of hydrocodone and other opiates is not known, although it is believed to relate to the existence of opiate receptors in the central nervous system. In addition to analgesia, narcotics may produce drowsiness, changes in mood and mental clouding.
The analgesic action of acetaminophen involves peripheral influences, but the specific mechanism is as yet undetermined. Antipyretic activity is mediated through hypothalamic heat regulating centers. Acetaminophen inhibits prostaglandin synthetase. Therapeutic doses of acetaminophen have negligible effects on the cardiovascular or respiratory systems; however, toxic doses may cause circulatory failure and rapid, shallow breathing.
Pharmacokinetics
The behavior of the individual components is described below.
Hydrocodone
Following a 10 mg oral dose of hydrocodone administered to five adult male subjects, the mean peak concentration was 23.6 ± 5.2 ng/mL. Maximum serum levels were achieved at 1.3 ± 0.3 hours and the half-life was determined to be 3.8 ± 0.3 hours. Hydrocodone exhibits a complex pattern of metabolism including O-demethylation, N-demethylation and 6-keto reduction to the corresponding 6-α- and 6-β-hydroxymetabolites.
See OVERDOSAGE for toxicity information.
Acetaminophen
Acetaminophen is rapidly absorbed from the gastrointestinal tract and is distributed throughout most body tissues. The plasma half-life is 1.25 to 3 hours, but may be increased by liver damage and following overdosage. Elimination of acetaminophen is principally by liver metabolism (conjugation) and subsequent renal excretion of metabolites. Approximately 85% of an oral dose appears in the urine within 24 hours of administration, most as the glucuronide conjugate, with small amounts of other conjugates and unchanged drug.
See OVERDOSAGE for toxicity information.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Respiratory Depression
At high doses or in sensitive patients, hydrocodone may produce dose-related respiratory depression by acting directly on the brain stem respiratory center. Hydrocodone also affects the center that controls respiratory rhythm, and may produce irregular and periodic breathing.
Infants may have increased sensitivity to the respiratory depressant effects of opioids (see PRECAUTIONS, Pediatric Use). If use of hydrocodone bitartrate and acetaminophen oral solution in such patients is contemplated, it should be administered cautiously, in substantially reduced initial doses, by personnel experienced in administering opioids to infants, and with intensive monitoring.
Head Injury and Increased Intracranial Pressure
The respiratory depressant effects of narcotics and their capacity to elevate cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions or a preexisting increase in intracranial pressure. Furthermore, narcotics produce adverse reactions, which may obscure the clinical course of patients with head injuries.
-
PRECAUTIONS
General
Special Risk Patients
As with any narcotic analgesic agent, hydrocodone bitartrate and acetaminophen oral solution should be used with caution in elderly or debilitated patients, and those with severe impairment of hepatic or renal function, hypothyroidism, Addison's disease, prostatic hypertrophy or urethral stricture. The usual precautions should be observed and the possibility of respiratory depression should be kept in mind.
Information for Patients
Hydrocodone, like all narcotics, may impair mental and/or physical abilities required for the performance of potentially hazardous tasks such as driving a car or operating machinery. Such tasks should be avoided while taking this product. Alcohol and other CNS depressants may produce an additive CNS depression, when taken with this combination product, and should be avoided.
Hydrocodone may be habit-forming. Patients should take the drug only for as long as it is prescribed, in the amounts prescribed, and no more frequently than prescribed.
Physicians should instruct patients and caregivers to read the patient information leaflet, which appears as the last section of the labeling.
Laboratory Tests
In patients with severe hepatic or renal disease, effects of therapy should be monitored with serial liver and/or renal function tests.
Drug Interactions
Patients receiving narcotics, antihistamines, antipsychotics, antianxiety agents, or other CNS depressants (including alcohol) concomitantly with hydrocodone bitartrate and acetaminophen oral solution may exhibit an additive CNS depression. When combined therapy is contemplated, the dose of one or both agents should be reduced.
The use of MAO inhibitors or tricyclic antidepressants with hydrocodone preparations may increase the effect of either the antidepressant or hydrocodone.
Drug/Laboratory Test Interactions
Acetaminophen may produce false-positive test results for urinary 5-hydroxyindoleacetic acid.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No adequate studies have been conducted in animals to determine whether hydrocodone has a potential for carcinogenesis, mutagenesis, or impairment of fertility.
Hydrocodone has not demonstrated mutagenic potential using the Ames Salmonella-Microsomal Activation test, the Basc test on Drosophila germ cells, and the Micronucleus test on mouse bone marrow.
No adequate studies have been conducted in animals to determine whether acetaminophen has a potential for carcinogenesis, mutagenesis, or impairment of fertility. Acetaminophen has not demonstrated mutagenic potential using the Ames Salmonella-Microsomal Activation test, the Basc test on Drosophila germ cells, and the Micronucleus test on mouse bone marrow.
Pregnancy
Nonteratogenic Effects
Babies born to mothers who have been taking opioids regularly prior to delivery will be physically dependent. The withdrawal signs include irritability and excessive crying, tremors, hyperactive reflexes, increased respiratory rate, increased stools, sneezing, yawning, vomiting and fever. These signs usually appear during the first few days of life. The intensity of the syndrome does not always correlate with the duration of maternal opioid use or dose. There is no consensus on the best method of managing withdrawal.
Labor and Delivery
Narcotic analgesics cross the placental barrier. The closer to delivery and the larger the dose used, the greater the possibility of respiratory depression in the newborn. Narcotic analgesics should be avoided during labor if delivery of a premature infant is anticipated. If the mother has received narcotic analgesics during labor, newborn infants should be observed closely for signs of respiratory depression. Resuscitation may be required (see OVERDOSAGE). The effect of hydrocodone, if any, on the later growth, development, and functional maturation of the child is unknown.
Nursing Mothers
Acetaminophen is excreted in breast milk in small amounts, but the significance of its effects on nursing infants is not known. It is not known whether hydrocodone is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from hydrocodone and acetaminophen, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness in the pediatric population below the age of two years have not been established. Use of hydrocodone bitartrate and acetaminophen oral solution in the pediatric population is supported by the evidence from adequate and well controlled studies of hydrocodone and acetaminophen combination products in adults with additional data which support the development of metabolic pathways in children two years of age and over (see DOSAGE AND ADMINISTRATION for pediatric dosage information).
Geriatric Use
Clinical studies of hydrocodone bitartrate and acetaminophen oral solution did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Hydrocodone and the major metabolites of acetaminophen are known to be substantially excreted by the kidney. Thus the risk of toxic reactions may be greater in patients with impaired renal function due to the accumulation of the parent compound and/or metabolites in the plasma. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Hydrocodone may cause confusion and over-sedation in the elderly; elderly patients generally should be started on low doses of hydrocodone bitartrate and acetaminophen oral solution and observed closely.
-
ADVERSE REACTIONS
Potential effects of high dosage are also listed in the OVERDOSAGE section.
Cardio-renal: Bradycardia, cardiac arrest, circulatory collapse, renal toxicity, renal tubular necrosis, hypotension.
Central Nervous System/Psychiatric: Anxiety, dizziness, drowsiness, dysphoria, euphoria, fear, general malaise, impairment of mental and physical performance, lethargy, lightheadedness, mental clouding, mood changes, psychological dependence, sedation, somnolence progressing to stupor or coma.
Endocrine: Hypoglycemic coma.
Gastrointestinal System: Abdominal pain, constipation, gastric distress, heartburn, hepatic necrosis, hepatitis, occult blood loss, nausea, peptic ulcer, and vomiting.
Genitourinary System: Spasm of vesical sphincters, ureteral spasm, and urinary retention.
Hematologic: Agranulocytosis, hemolytic anemia, iron deficiency anemia, prolonged bleeding time, thrombocytopenia.
Hypersensitivity: Allergic reactions.
Musculoskeletal: Skeletal muscle flaccidity.
Respiratory Depression: Acute airway obstruction, apnea, dose-related respiratory depression (see OVERDOSAGE), shortness of breath.
Special Senses: Cases of hearing impairment or permanent loss have been reported predominantly in patients with chronic overdose.
Skin: Cold and clammy skin, diaphoresis, pruritus, rash.
-
DRUG ABUSE AND DEPENDENCE
Controlled Substance
Hydrocodone bitartrate and acetaminophen oral solution is classified as a Schedule III controlled substance.
Abuse and Dependence
Hydrocodone can produce drug dependence of the morphine type and, therefore, has the potential for being abused. Psychological dependence, physical dependence, and tolerance may develop upon repeated administration of narcotics; therefore, this product should be prescribed and administered with caution appropriate to the use of other oral narcotic medications. However, psychological dependence is unlikely to develop when hydrocodone bitartrate and acetaminophen oral solution are used for a short time for the treatment of pain.
Physical dependence, the condition in which continued administration of the drug is required to prevent the appearance of a withdrawal syndrome, assumes clinically significant proportions only after several weeks of continued narcotic use, although some mild degree of physical dependence may develop after a few days of narcotic therapy. Tolerance, in which increasingly large doses are required in order to produce the same degree of analgesia, is manifested initially by a shortened duration of analgesic effect, and subsequently by decreases in the intensity of analgesia. The rate of development of tolerance varies among patients.
-
OVERDOSAGE
Following an acute overdosage, toxicity may result from hydrocodone or acetaminophen.
Signs and Symptoms
Toxicity from hydrocodone poisoning includes the opioid triad of loss of consciousness, pinpoint pupils, and respiratory depression (Cheyne-Stokes respiration, cyanosis, decrease in respiratory rate and/or tidal volume). Convulsions may occur.
The toxic dose of acetaminophen for adults is 10 grams. In adults, hepatic toxicity has rarely been reported with acute overdoses of less than 10 grams, or fatalities with less than 15 grams.
Early symptoms following a potentially hepatotoxic overdose of acetaminophen may include diaphoresis, general malaise, nausea, and vomiting. Clinical and laboratory evidence of hepatic toxicity may not be apparent until 48 to 72 hours post-ingestion.
Other signs and symptoms of overdose of this product include bradycardia, cold and clammy skin, extreme somnolence progressing to stupor or coma, hypoglycemic coma, hypotension, renal tubular necrosis, skeletal muscle flaccidity, thrombocytopenia.
In severe overdosage, apnea; circulatory collapse; cardiac arrest; dose-dependent, potentially fatal hepatic necrosis; and death may occur.
Treatment
A single or multiple overdose with hydrocodone and acetaminophen is a potentially lethal polydrug overdose, and consultation with a regional poison control center is recommended.
Immediate treatment includes support of cardiorespiratory function and measures to reduce drug absorption. Vomiting should be induced with syrup of ipecac, if the patient is alert (adequate pharyngeal and laryngeal reflexes). Oral activated charcoal (1 g/kg) should follow gastric emptying. The first dose should be accompanied by an appropriate cathartic. If repeated doses are used, the cathartic might be included with alternate doses as required. Hypotension is usually hypovolemic and should respond to fluids. Vasopressors and other supportive measures should be employed as indicated. A cuffed endo-tracheal tube should be inserted before gastric lavage of the unconscious patient and, when necessary, to provide assisted respiration.
Meticulous attention should be given to maintaining adequate pulmonary ventilation. In severe cases of intoxication, peritoneal dialysis, or preferably hemodialysis may be considered. If hypoprothrombinemia occurs due to acetaminophen overdose, vitamin K should be administered intravenously.
Naloxone, a narcotic antagonist, can reverse respiratory depression and coma associated with opioid overdose. Naloxone hydrochloride 0.4 mg to 2 mg is given parenterally. Since the duration of action of hydrocodone may exceed that of the naloxone, the patient should be kept under continuous surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration. A narcotic antagonist should not be administered in the absence of clinically significant respiratory or cardiovascular depression.
If the dose of acetaminophen may have exceeded 140 mg/kg, acetylcysteine should be administered as early as possible. Serum acetaminophen levels should be obtained, since levels four or more hours following ingestion help predict acetaminophen toxicity. Do not await acetaminophen assay results before initiating treatment. Hepatic enzymes should be obtained initially, and repeated at 24-hour intervals.
Methemoglobinemia over 30% should be treated with methylene blue by slow intravenous administration.
-
DOSAGE AND ADMINISTRATION
Dosage should be adjusted according to severity of pain and response of the patient. However, it should be kept in mind that tolerance to hydrocodone can develop with continued use and that the incidence of untoward effects is dose related.
The usual adult dosage is one tablespoonful every 4 to 6 hours as needed for pain. The total daily dosage should not exceed 6 tablespoonfuls.
The usual dosages for children are given by the table below, and are to be given every 4 to 6 hours as needed for pain. These dosages correspond to an average individual dose of 0.27 mL/kg of hydrocodone bitartrate and acetaminophen oral solution (providing 0.135 mg/kg of hydrocodone bitartrate and 9 mg/kg of acetaminophen). Dosing should be based on weight whenever possible.
BODY WEIGHT APPROXIMATE
AGEDOSE
every 4
to 6 hoursMAXIMUM
TOTAL
DAILY DOSE
(6 doses
per day)12 to 15 kg
27 to 34 lbs.2 to 3 years ¾ teaspoonful
= 3.75 mL4½ teaspoonfuls
= 22.5 mL16 to 22kg
35 to 50 lbs.4 to 6 years 1 teaspoonful
= 5 mL6 teaspoonfuls
= 30 mL23 to 31 kg
51 to 69 lbs.7 to 9 years 1½ teaspoonfuls
= 7.5 mL9 teaspoonfuls
= 45 mL32 to 45 kg
70 lbs. to 100 lbs.10 to 13 years 2 teaspoonfuls
= 10 mL12 teaspoonfuls
= 60 mL46 kg and up
101 lbs. and up14 years to adult 1 Tablespoonful
= 15 mL6 Tablespoonfuls
= 90 mLThe total daily dosage for children should not exceed 6 doses per day. It is of utmost importance that the dose of hydrocodone bitartrate and acetaminophen oral solution be administered accurately. A household teaspoon or tablespoon is not an adequate measuring device, especially when one-half or three-fourths of a teaspoonful is to be measured. Given the inexactitude of the household spoon measure and the possibility of using a tablespoon instead of a teaspoon, which could lead to overdosage, it is strongly recommended that caregivers obtain and use a calibrated measuring device. Healthcare providers should recommend a dropper that can measure and deliver the prescribed dose accurately, and instruct caregivers to use extreme caution in measuring the dosage.
- HOW SUPPLIED
-
Patient Information Leaflet
Hydrocodone Bitartrate and Acetaminophen Oral Solution CIII
7.5 mg/500 mg per 15 mLSummary
Hydrocodone bitartrate and acetaminophen oral solution is used to relieve moderate to moderately severe pain. You should not take hydrocodone bitartrate and acetaminophen oral solution if you are allergic to hydrocodone or acetaminophen. The most common side effects of hydrocodone bitartrate and acetaminophen oral solution are abdominal pain, dizziness, drowsiness, lightheadedness, nausea, shortness of breath, unusual tiredness, and vomiting. Take this medicine as directed by your doctor. Do not take more of it, do not take it more often, and do not take it for a longer time than your doctor ordered. Uses
Hydrocodone bitartrate and acetaminophen oral solution is an analgesic used to relieve moderate to moderately severe pain. Hydrocodone bitartrate and acetaminophen oral solution is a combination product containing hydrocodone (hye-droe-KO-done) bitartrate and acetaminophen (a-seat-a-MIN-oh-fen). Hydrocodone is a narcotic pain reliever and a cough suppressant. Acetaminophen is a non-narcotic pain reliever and fever reducer. A narcotic analgesic and acetaminophen used together may provide better pain relief than either product used alone. If you have any questions, please call your doctor or pharmacist.
General Cautions
- Do not take this drug if you have allergies or unusual reactions to narcotic pain relievers or acetaminophen because it is likely that you may also be allergic to hydrocodone bitartrate and acetaminophen oral solution.
- This product may inhibit your mental and physical abilities required for the performance of potentially hazardous tasks such as driving a car or operating machinery. Such tasks should be avoided while you are taking this product.
- This medicine may not be right for you. Check with your doctor or pharmacist, if you:
- are pregnant.
- are nursing.
- are taking other medications: narcotic pain relievers; allergy medicines; antidepressant medicines; acetaminophen-containing medicines or other medicines that cause central nervous system depression, including alcohol.
- have other medical problems: a history of drug or alcohol abuse; recent head injury; emphysema, asthma, or other chronic lung disease; liver disease; kidney disease; underactive thyroid, Addison’s disease, enlarged prostate or difficulty urinating.
Proper Use
Take this medicine as directed by your doctor. Do not share it with anyone else. This medicine can cause drug dependence and has the potential for abuse. Do not take more of it, do not take it more often, and do not take it for a longer time than your doctor ordered. If you think that this medicine is not working properly after taking it for some time, do not increase the dose. Check with your doctor or pharmacist.
Dosing
The dose of this medication will be different for different patients. Follow the directions provided by your doctor. The following information includes only the average doses of this medication. If your dose is different, do not change doses unless your doctor tells you to do so.
BODY WEIGHT APPROXIMATE
AGEDOSE
Every 4
to 6 hoursMAXIMUM TOTAL
DAILY DOSE
(6 doses per day)12 to 15 kg
27 to 34 lbs.2 to 3 years ¾ teaspoonful
= 3.75 mL4 ½ teaspoonfuls
= 22.5 mL16 to 22 kg
35 to 50 lbs.4 to 6 years 1 teaspoonful
= 5 mL6 teaspoonfuls
= 30 mL23 to 31 kg
51 to 69 lbs.7 to 9 years 1 ½ teaspoonfuls
= 7.5 mL9 teaspoonfuls
= 45 mL32 to 45 kg
70 to 100 lbs.10 to 13 years 2 teaspoonfuls
= 10 mL12 teaspoonfuls
= 60 mL46 kg and up
101 lbs. and up14 years to adult 1 Tablespoonful
= 15 mL6 Tablespoonfuls
= 90 mLIt is very important that hydrocodone bitartrate and acetaminophen oral solution be dosed accurately. A household teaspoon or tablespoon is not an accurate measuring device, especially when one-half or three-fourths of a teaspoonful is to be measured.
Since a household teaspoon is not accurate and can be mixed-up with a tablespoon (which can cause overdosage), it is strongly recommended that you obtain and use a proper measuring device. Ask your doctor or pharmacist for help to find a dropper that can measure the needed dose properly and ask for help if you do not understand how to use the dropper.
Missed Dose
- To avoid a possible overdose, it is important that you do not take more than a single dosage at one time, or that you don't take doses at intervals less than 4 hours apart.
- If you miss taking a dose of hydrocodone bitartrate and acetaminophen oral solution, take it as soon as you remember. However, make sure to wait at least 4 hours before taking your next dose.
- If you missed taking a dose, and it is almost time for your next dose, skip the missed dose and take your medicine as scheduled.
- Do not double the prescribed dose.
Possible Side Effects
Side effects you may experience include abdominal pain, constipation, difficulty urinating, dizziness, drowsiness, fear, fuzzy thinking, general feeling of discomfort or illness, lightheadedness, mood changes, nausea, nervousness, rash, shortness of breath, slower reactions, unusual tiredness, and vomiting.
Call your doctor if these effects continue or are bothersome.
Side effects not listed above may sometimes occur. If you notice any other effects, check with your doctor.
Storage
- Keep out of the reach of children.
- Store at room temperature (protect from heat, do not refrigerate).
- Keep in original labeled bottle.
- Discard medicines that are old or no longer needed.
- Even a single overdose of this medicine may be a life-threatening situation. If you suspect that you or someone else may have taken more than the prescribed dose of this medicine, contact your local poison control center or emergency room immediately. This medicine was prescribed for your particular condition. Do not use if for another condition or give the drug to others.
- This leaflet provides a summary of information about hydrocodone bitartrate and acetaminophen oral solution. If you have any questions or concerns, or want more information about hydrocodone bitartrate and acetaminophen oral solution, contact your doctor or pharmacist. Your pharmacist also has a longer leaflet about hydrocodone bitartrate and acetaminophen oral solution that is written for health professionals that you can ask to read.
Manufactured by
KV Pharmaceutical Co. for
ETHEX Corporation
St. Louis, MO 63044Rev. 08/06
P4218-2
-
INGREDIENTS AND APPEARANCE
HYDROCODONE BITARTRATE AND ACETAMINOPHEN
hydrocodone bitartrate and acetaminophen liquidProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:58177-909 Route of Administration ORAL DEA Schedule CIII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Hydrocodone bitartrate (UNII: NO70W886KK) (hydrocodone - UNII:6YKS4Y3WQ7) 7.5 mg in 15 mL acetaminophen (UNII: 362O9ITL9D) (acetaminophen - UNII:362O9ITL9D) 500 mg in 15 mL Inactive Ingredients Ingredient Name Strength ammoniated glyyerrhizin () dibasic sodium phosphate (UNII: GR686LBA74) citric acid (UNII: 2968PHW8QP) ethyl maltol (UNII: L6Q8K29L05) polyethylene glycol () propylene glycol (UNII: 6DC9Q167V3) water (UNII: 059QF0KO0R) saccharin sodium (UNII: SB8ZUX40TY) sodium citrate (UNII: 1Q73Q2JULR) sodium benzoate (UNII: OJ245FE5EU) sodium gluconate (UNII: R6Q3791S76) sorbitol solution () D&C Yellow No. 10 () FD&C Yellow No. 6 () Natural and artifical flavoring () hydrochloric acid (UNII: QTT17582CB) sodium hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58177-909-07 473 mL in 1 BOTTLE Labeler - ETHEX Corporation
