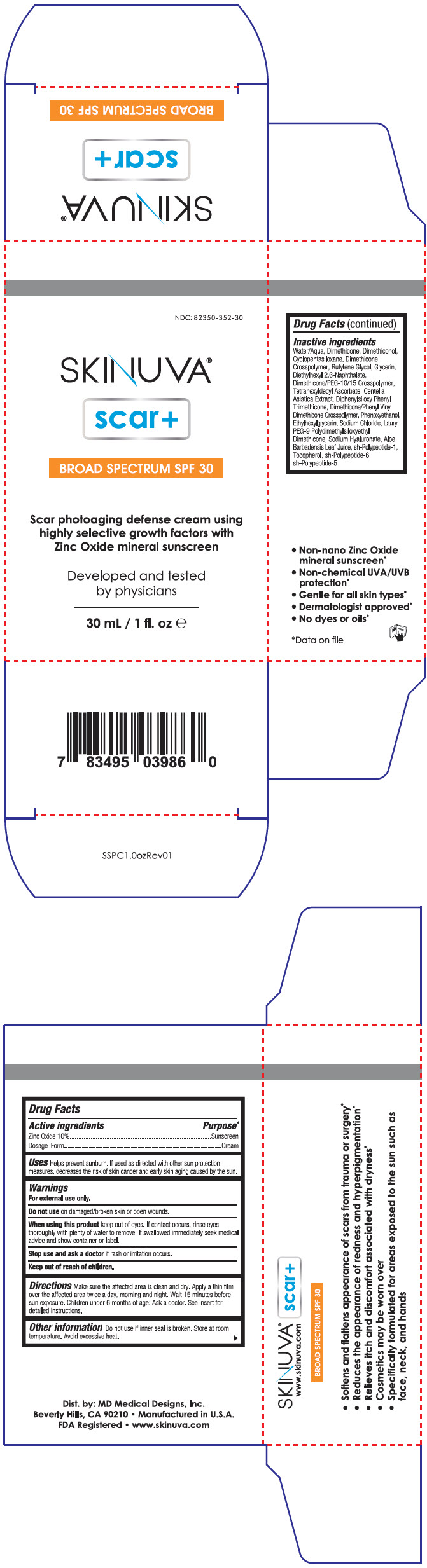
Label: SKINUVA SCAR PLUS- zinc oxide cream
- NDC Code(s): 82350-352-15, 82350-352-30
- Packager: MD Medical Designs, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 12, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
Water/Aqua, Dimethicone, Dimethiconol, Cyclopentasiloxane, Dimethicone Crosspolymer, Butylene Glycol, Glycerin, Diethylhexyl 2,6-Naphthalate, Dimethicone/PEG-10/15 Crosspolymer, Tetrahexyldecyl Ascorbate, Centella Asiatica Extract, Diphenylsiloxy Phenyl Trimethicone, Dimethicone/Phenyl Vinyl Dimethicone Crosspolymer, Phenoxyethanol, Ethylhexylglycerin, Sodium Chloride, Lauryl PEG-9 Polydimethylsiloxyethyl Dimethicone, Sodium Hyaluronate, Aloe Barbadensis Leaf Juice, sh-Polypeptide-1, Tocopherol, sh-Polypeptide-6, sh-Polypeptide-5
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 30 mL Bottle Carton
-
INGREDIENTS AND APPEARANCE
SKINUVA SCAR PLUS
zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82350-352 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Zinc Oxide (UNII: SOI2LOH54Z) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 0.1 g in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Dimethicone (UNII: 92RU3N3Y1O) Cyclomethicone 5 (UNII: 0THT5PCI0R) Butylene Glycol (UNII: 3XUS85K0RA) Glycerin (UNII: PDC6A3C0OX) Diethylhexyl 2,6-Naphthalate (UNII: I0DQJ7YGXM) Dimethicone/PEG-10/15 Crosspolymer (UNII: 21AS8B1BSS) Tetrahexyldecyl Ascorbate (UNII: 9LBV3F07AZ) Centella Asiatica Triterpenoids (UNII: 4YS74Q4G4J) Diphenylsiloxy Phenyl Trimethicone (UNII: I445L28B12) Dimethicone/Phenyl Vinyl Dimethicone Crosspolymer (UNII: ST6ZC4KVH2) Ethylhexylglycerin (UNII: 147D247K3P) Sodium Chloride (UNII: 451W47IQ8X) Lauryl PEG-9 Polydimethylsiloxyethyl Dimethicone (UNII: 25G622K2RA) Hyaluronate Sodium (UNII: YSE9PPT4TH) Aloe Vera Leaf (UNII: ZY81Z83H0X) BASIC FIBROBLAST GROWTH FACTOR (HUMAN) (UNII: S3529G9M9V) Tocopherol (UNII: R0ZB2556P8) Phenoxyethanol (UNII: HIE492ZZ3T) Interleukin-10 (UNII: 9SC4O216V9) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82350-352-15 1 in 1 CARTON 06/30/2022 1 15 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:82350-352-30 1 in 1 CARTON 06/30/2022 2 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph drug M020 06/30/2022 Labeler - MD Medical Designs, Inc. (094586004)