Label: OB COMPLETE DHA- omega-3 fatty acids, calcium ascorbate, calcium threonate, cholecalciferol, .alpha.-tocopherol, d-, thiamine mononitrate, riboflavin, pyridoxine hydrochloride, folic acid, cyanocobalamin, iron pentacarbonyl, ferrous asparto glycinate, zinc oxide, and cupric sulfate capsule, liquid filled
- NHRIC Code(s): 68025-049-60
- Packager: Vertical Pharmaceuticals, LLC
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated January 7, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SUPPLEMENT FACTS
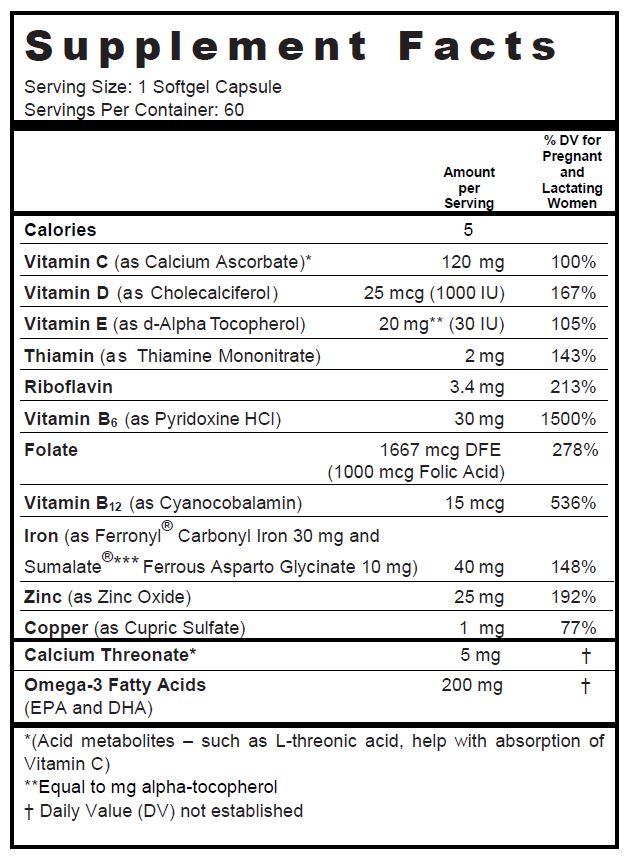
Other Ingredients: Soybean Oil, Gelatin, Glycerin, Yellow Beeswax, Lecithin, Purified Water, Carmine Hydrosoluble, Titanium Dioxide, Ethyl Vanillin, FD&C Red #40, FD&C Blue #1
THIS PRODUCT CONTAINS SOY AND FISH OIL (ANCHOVIES, MACKEREL, SARDINES, TUNA).
GLUTEN-, LACTOSE- AND SUGAR-FREE
OB Complete® with DHA is a prescription multivitamin/multimineral indicated for use in improving the nutritional status of women prior to conception, throughout pregnancy, and in the postnatal period for both lactating and non-lactating mothers.
-
CONTRAINDICATIONS
OB Complete™ with DHA should not be used by patients with a known hypersensitivity to any of the listed ingredients including soy and fish oil.
WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. KEEP THIS PRODUCT OUT OF REACH OF CHILDREN. In case of accidental overdose, call a doctor or poison control center immediately.
-
PRECAUTIONS
Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient. Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
The patient’s medical conditions and consumption of other drugs, herbs, and/or supplements should be considered.
-
DRUG INTERACTIONS
OB Complete® with DHA Prenatal Multivitamin/Multimineral is not recommended for and should not be given to patients receiving levodopa because the action of levodopa is antagonized by pyridoxine. There is a possibility of increased bleeding due to pyridoxine interaction with anticoagulants (e.g., Aspirin, Heparin, Clopidogrel).
-
ADVERSE REACTIONS
Adverse reactions have been reported with specific vitamins and minerals but generally at levels substantially higher than those contained herein. However, allergic and idiosyncratic reactions are possible at lower levels. Iron, even at the usual recommended levels, has been associated with gastrointestinal intolerance in some patients.
- DESCRIPTION
- DIRECTIONS FOR USE
- HOW SUPPLIED
- STORAGE
-
HEALTH CLAIM
KEEP THIS PRODUCT OUT OF REACH OF CHILDREN.
***Sumalate® (Ferrous Asparto Glycinate) is a registered trademark of Albion International, Inc. and is covered by the following patents: U.S. Pat. No. 6,716,814; U.S. Pat. No. 8,007,846 and U.S. Pat. No. 8,425,956.
For use on the order of a healthcare practitioner.
Call your doctor about side effects. To report side effects, call Vertical Pharmaceuticals at 1-770-509-4500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
PLR-OBCDHA-00003-3 Rev. 11/2021
Distributed by:
Vertical Pharmaceuticals, LLC
Alpharetta, GA 300051-770-509-4500
www.verticalpharma.com

- PRINCIPAL DISPLAY PANEL - 60 Softgel Bottle Label
-
INGREDIENTS AND APPEARANCE
OB COMPLETE DHA
omega-3 fatty acids, calcium ascorbate, calcium threonate, cholecalciferol, .alpha.-tocopherol, d-, thiamine mononitrate, riboflavin, pyridoxine hydrochloride, folic acid, cyanocobalamin, iron pentacarbonyl, ferrous asparto glycinate, zinc oxide, and cupric sulfate capsule, liquid filledProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:68025-049 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Omega-3 Fatty Acids (UNII: 71M78END5S) (Omega-3 Fatty Acids - UNII:71M78END5S) Omega-3 Fatty Acids 200 mg Calcium Ascorbate (UNII: 183E4W213W) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 120 mg Calcium Threonate (UNII: HBB4YPO93U) (CALCIUM CATION - UNII:2M83C4R6ZB) Calcium Threonate 5 mg Cholecalciferol (UNII: 1C6V77QF41) (Cholecalciferol - UNII:1C6V77QF41) Cholecalciferol 1000 [iU] .Alpha.-Tocopherol, D- (UNII: N9PR3490H9) (.Alpha.-Tocopherol, D- - UNII:N9PR3490H9) .Alpha.-Tocopherol, D- 30 [iU] Thiamine Mononitrate (UNII: 8K0I04919X) (Thiamine Ion - UNII:4ABT0J945J) Thiamine 2 mg Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 3.4 mg Pyridoxine Hydrochloride (UNII: 68Y4CF58BV) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine Hydrochloride 30 mg Folic Acid (UNII: 935E97BOY8) (Folic Acid - UNII:935E97BOY8) Folic Acid 1 mg Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin 15 ug IRON PENTACARBONYL (UNII: 6WQ62TAQ6Z) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 30 mg Ferrous Asparto Glycinate (UNII: H7426RGB3L) (Ferrous Cation - UNII:GW89581OWR) Ferrous Cation 10 mg Zinc Oxide (UNII: SOI2LOH54Z) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 25 mg Cupric Sulfate (UNII: LRX7AJ16DT) (Cupric Cation - UNII:8CBV67279L) Cupric Cation 1 mg Inactive Ingredients Ingredient Name Strength Soybean Oil (UNII: 241ATL177A) Gelatin (UNII: 2G86QN327L) Glycerin (UNII: PDC6A3C0OX) Yellow Wax (UNII: 2ZA36H0S2V) Water (UNII: 059QF0KO0R) Lecithin, Soybean (UNII: 1DI56QDM62) Pigment Red 48 (UNII: 07XHK4SAV6) Titanium Dioxide (UNII: 15FIX9V2JP) Ethyl Vanillin (UNII: YC9ST449YJ) FD&C Red No. 40 (UNII: WZB9127XOA) FD&C Blue No. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:68025-049-60 60 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 07/01/2011 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color imprint scoring 1 shape size (solid drugs) 15 mm Labeler - Vertical Pharmaceuticals, LLC (173169017)


