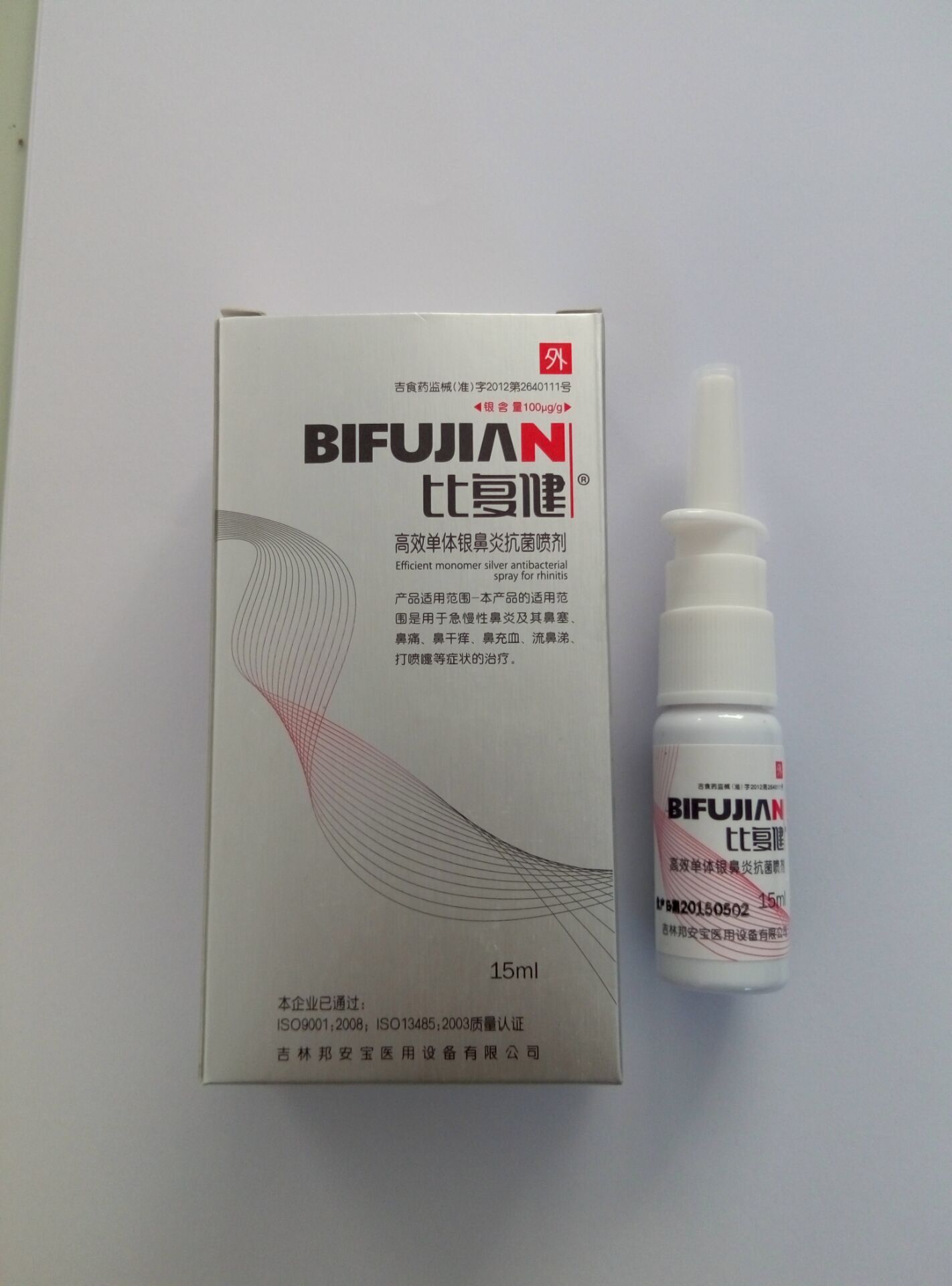
Label: BI FU JIAN HIGH-EFFICIENCY ANTI-BACTERIA MONOMER SILVER SPRAYS FOR RHINITIS- high-efficiency anti-bacteria monomer silver sprays for rhinitis liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 60659-0303-0, 60659-0303-1 - Packager: Bang An Bao Medical Equipment Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 8, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Indcations and usage
- Dosage
-
Warnings
Contraindications:
1, Not for virgins.
2, Do not use if allergic to silver.
3, Pregnant women shall follow doctor advice.
Notes:
1, Do not use if package is broken; do not use with medicine that contains Vaseline, chlorine, iodine or hydrogen peroxide.
2, This product is for external use only; do not take in.
3, Keep away from clothes to prevent contamination. - Out of children
- Inactive ingredient
- Label
-
INGREDIENTS AND APPEARANCE
BI FU JIAN HIGH-EFFICIENCY ANTI-BACTERIA MONOMER SILVER SPRAYS FOR RHINITIS
high-efficiency anti-bacteria monomer silver sprays for rhinitis liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:60659-0303 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILVER (UNII: 3M4G523W1G) (SILVER - UNII:3M4G523W1G) SILVER 1500 ug in 15 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 20000 ug in 15 mL WATER (UNII: 059QF0KO0R) 14978500 ug in 15 mL Product Characteristics Color yellow Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60659-0303-1 1 in 1 CARTON 1 NDC:60659-0303-0 15 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 01/08/2016 Labeler - Bang An Bao Medical Equipment Co., Ltd. (421323212) Registrant - Bang An Bao Medical Equipment Co., Ltd. (421323212) Establishment Name Address ID/FEI Business Operations Bang An Bao Medical Equipment Co., Ltd. 421323212 label(60659-0303) , manufacture(60659-0303)