Label: LEVOBUNOLOL HYDROCHLORIDE solution/ drops
- NDC Code(s): 24208-505-05, 24208-505-10, 24208-505-15
- Packager: Bausch & Lomb Incorporated
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 24, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Levobunolol hydrochloride ophthalmic solution USP, 0.5% is a noncardioselective beta-adrenoceptor blocking agent for ophthalmic use.
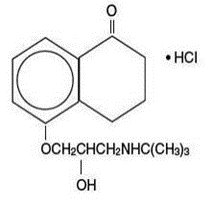
Levobunolol hydrochloride is represented by the following structural formula:
Mol. Formula C 17H 25NO 3•HCl
Mol. Wt. 327.85
Chemical Name:(–)-5-[3-( tert-Butylamino)-2-hydroxypropoxy]-3,4-dihydro-1(2 H)- naphthalenone hydrochloride.
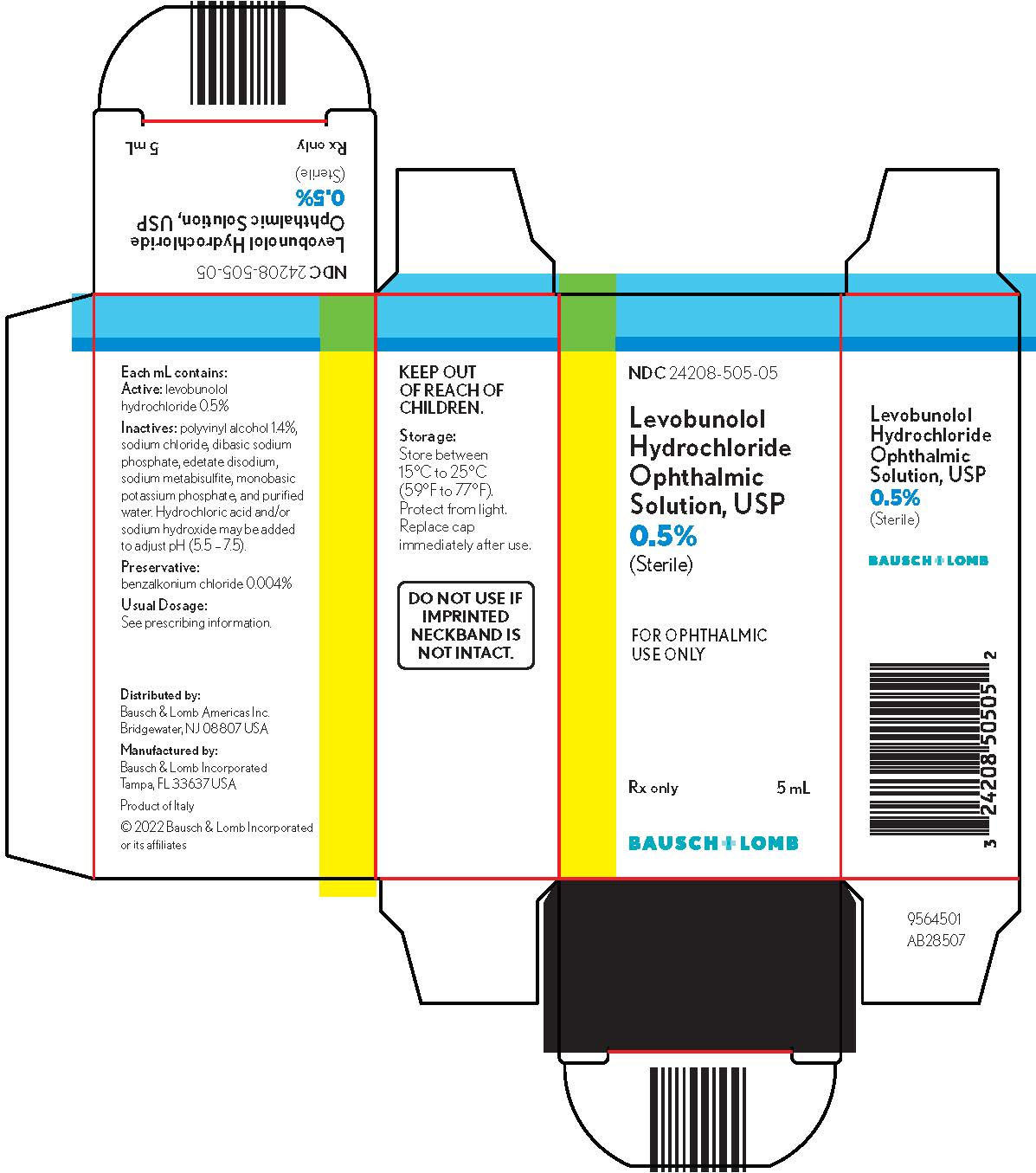
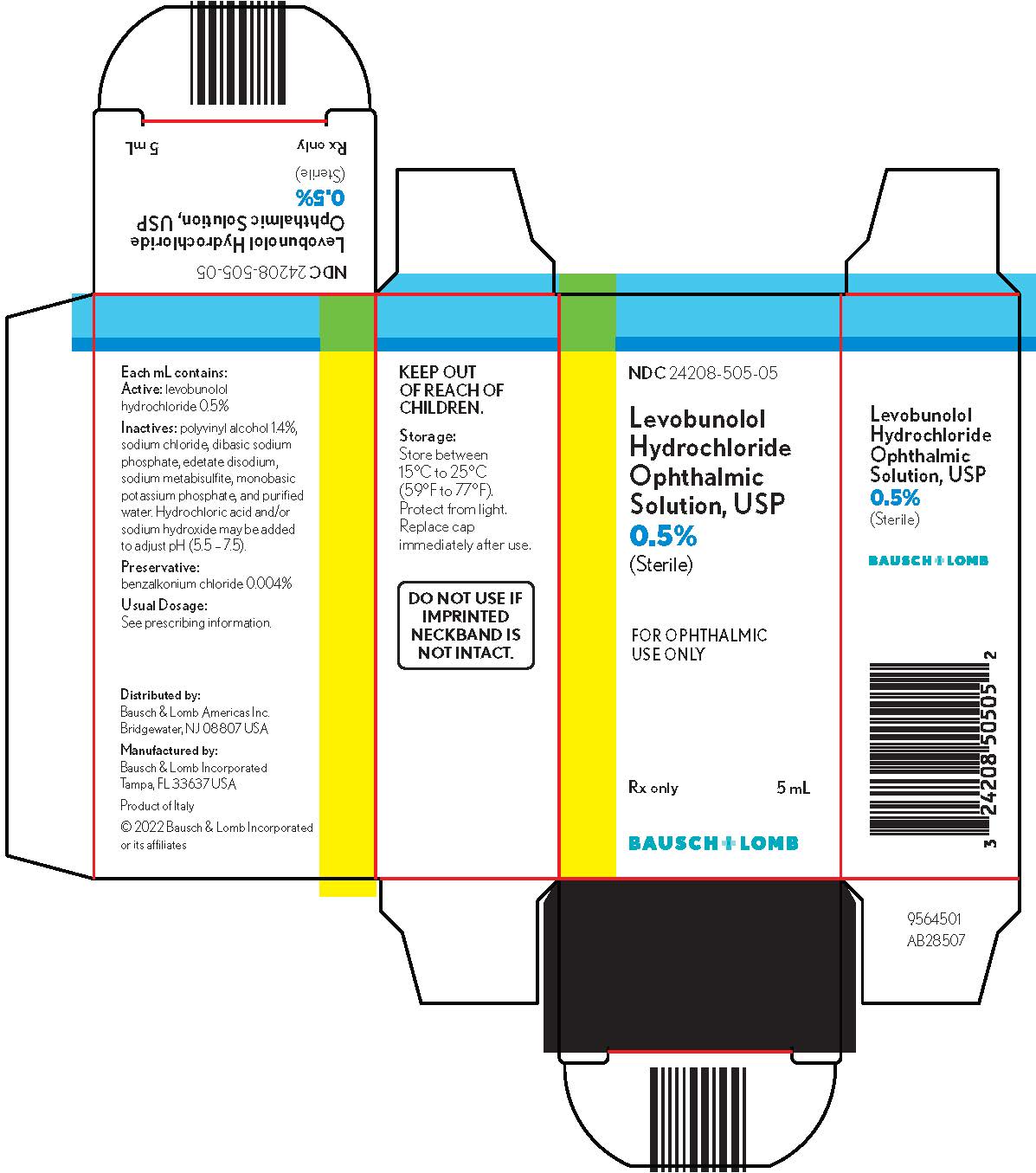
Each mL of 0.5% contains:
Active:levobunolol hydrochloride 0.5%; Inactives:polyvinyl alcohol 1.4%, sodium chloride, dibasic sodium phosphate, edetate disodium, sodium metabisulfite, monobasic potassium phosphate, and purified water. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH (5.5 - 7.5); Preservative:benzalkonium chloride (0.004%).
-
CLINICAL PHARMACOLOGY
Levobunolol hydrochloride is a noncardioselective beta-adrenoceptor blocking agent, equipotent at both beta 1and beta 2receptors. Levobunolol hydrochloride is greater than 60 times more potent than its dextro isomer in its beta-blocking activity, yet equipotent in its potential for direct myocardial depression. Accordingly, the levo isomer, levobunolol hydrochloride, is used. Levobunolol hydrochloride does not have significant local anesthetic (membrane-stabilizing) or intrinsic sympathomimetic activity.
Beta-adrenergic receptor blockade reduces cardiac output in both healthy subjects and patients with heart disease. In patients with severe impairment of myocardial function, beta-adrenergic receptor blockade may inhibit the stimulatory effect of the sympathetic nervous system necessary to maintain adequate cardiac function.
Beta-adrenergic receptor blockade in the bronchi and bronchioles results in increased airway resistance from unopposed parasympathetic activity. Such an effect in patients with asthma or other bronchospastic conditions is potentially dangerous.
Levobunolol hydrochloride ophthalmic solution, USP has been shown to be an active agent in lowering elevated as well as normal intraocular pressure (IOP) whether or not accompanied by glaucoma. Elevated IOP presents a major risk factor in glaucomatous field loss. The higher the level of IOP, the greater the likelihood of optic nerve damage and visual field loss.
The onset of action with one drop of levobunolol hydrochloride ophthalmic solution can be detected within one hour after treatment, with maximum effect seen between 2 and 6 hours.
A significant decrease in IOP can be maintained for up to 24 hours following a single dose.
In controlled clinical studies of approximately two years duration, intraocular pressure was well-controlled in approximately 80% of subjects treated with levobunolol hydrochloride ophthalmic solution 0.5% twice a day. The mean IOP decrease from baseline was between 7 mm Hg and 8 mm Hg. No significant effects on pupil size, tear production or corneal sensitivity were observed. Levobunolol hydrochloride ophthalmic solution at the concentrations tested, when applied topically, decreased heart rate and blood pressure in some patients. The IOP-lowering effect of levobunolol hydrochloride ophthalmic solution was well maintained over the course of these studies.
In a three-month clinical study, a single daily application of 0.5% levobunolol hydrochloride ophthalmic solution controlled the IOP of 72% of subjects achieving an overall mean decrease in IOP of 7.0 mm Hg.
The primary mechanism of the ocular hypotensive action of levobunolol hydrochloride in reducing IOP is most likely a decrease in aqueous humor production. Levobunolol hydrochloride ophthalmic solution reduces IOP with little or no effect on pupil size or accommodation.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Levobunolol hydrochloride ophthalmic solution is contraindicated in those individuals with bronchial asthma, or with a history of bronchial asthma, or severe chronic obstructive pulmonary disease (see WARNINGS); sinus bradycardia; second and third degree atrioventricular block; overt cardiac failure (see WARNINGS); cardiogenic shock; or hypersensitivity to any component of these products.
-
WARNINGS
As with other topically applied ophthalmic drugs, levobunolol hydrochloride ophthalmic solution may be absorbed systemically. The same adverse reactions found with systemic administration of beta-adrenergic blocking agents may occur with topical administration. For example, severe respiratory reactions and cardiac reactions, including death due to bronchospasm in patients with asthma, and rarely death in association with cardiac failure, have been reported with topical application of beta-adrenergic blocking agents (see CONTRAINDICATIONS). Additionally, ophthalmic beta-blockers may impair compensatory tachycardia and increase risk of hypotension.
Cardiac Failure
Sympathetic stimulation may be essential for support of the circulation in individuals with diminished myocardial contractility, and its inhibition by beta-adrenergic receptor blockade may precipitate more severe failure.
In Patients Without a History of Cardiac Failure
Continued depression of the myocardium with beta-blocking agents over a period of time can, in some cases, lead to cardiac failure. At the first sign or symptom of cardiac failure, levobunolol hydrochloride ophthalmic solution should be discontinued (see CONTRAINDICATIONS).
Potentiation of Vascular Insufficiency
Levobunolol hydrochloride ophthalmic solution may potentiate syndromes associated with vascular insufficiency (i.e. Raynaud’s phenomenon), and therefore, should be used with caution in these patients.
Obstructive Pulmonary Disease
PATIENTS WITH CHRONIC OBSTRUCTIVE PULMONARY DISEASE (e.g., CHRONIC BRONCHITIS, EMPHYSEMA) OF MILD OR MODERATE SEVERITY, BRONCHOSPASTIC DISEASE OR A HISTORY OF BRONCHOSPASTIC DISEASE (OTHER THAN BRONCHIAL ASTHMA OR A HISTORY OF BRONCHIAL ASTHMA, IN WHICH LEVOBUNOLOL HYDROCHLORIDE OPHTHALMIC SOLUTION IS CONTRAINDICATED, SEE CONTRAINDICATIONS), SHOULD IN GENERAL NOT RECEIVE BETA BLOCKERS, INCLUDING LEVOBUNOLOL HYDROCHLORIDE OPHTHALMIC SOLUTION. However, if levobunolol hydrochloride ophthalmic solution is deemed necessary in such patients, then it should be administered cautiously since it may block bronchodilation produced by endogenous and exogenous catecholamine stimulation of beta 2receptors.
Major Surgery
The necessity or desirability of withdrawal of beta-adrenergic blocking agents prior to major surgery is controversial. Beta-adrenergic receptor blockade impairs the ability of the heart to respond to beta-adrenergically mediated reflex stimuli. This may augment the risk of general anesthesia in surgical procedures. Some patients receiving beta-adrenergic receptor blocking agents have been subject to protracted severe hypotension during anesthesia. Difficulty in restarting and maintaining the heartbeat has also been reported. For these reasons, in patients undergoing elective surgery, gradual withdrawal of beta-adrenergic blocking agents may be appropriate.
If necessary during surgery, the effects of beta-adrenergic blocking agents may be reversed by sufficient doses of such agonists as isoproterenol, dopamine, dobutamine or levarterenol (see OVERDOSAGE).
Diabetes Mellitus
Beta-adrenergic blocking agents should be administered with caution in patients subject to spontaneous hypoglycemia or to diabetic patients (especially those with labile diabetes) who are receiving insulin or oral hypoglycemic agents. Beta-adrenergic blocking agents may mask the signs and symptoms of acute hypoglycemia.
Thyrotoxicosis
Beta-adrenergic blocking agents may mask certain clinical signs (e.g., tachycardia) of hyperthyroidism. Patients suspected of developing thyrotoxicosis should be managed carefully to avoid abrupt withdrawal of beta-adrenergic blocking agents which might precipitate a thyroid storm.
Choroidal Detachment
Choroidal detachment after filtration procedures has been reported with the administration of aqueous suppressant therapy.
These products contain sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
-
PRECAUTIONS
General
Levobunolol hydrochloride ophthalmic solution, USP should be used with caution in patients with known hypersensitivity to other beta-adrenoceptor blocking agents.
Use with caution in patients with known diminished pulmonary function.
Levobunolol hydrochloride ophthalmic solution should be used with caution in patients who are receiving a beta-adrenergic blocking agent orally, because of the potential for additive effects on systemic beta-blockade or on intraocular pressure. Patients should not typically use two or more topical ophthalmic beta-adrenergic blocking agents simultaneously.
Because of the potential effects of beta-adrenergic blocking agents on blood pressure and pulse rates, these medications must be used cautiously in patients with cerebrovascular insufficiency. Should signs or symptoms develop that suggest reduced cerebral blood flow while using levobunolol hydrochloride ophthalmic solution, alternative therapy should be considered.
In patients with angle-closure glaucoma, the immediate objective of treatment is to reopen the angle. This requires, in most cases, constricting the pupil with a miotic. Levobunolol hydrochloride ophthalmic solution has little or no effect on the pupil. When levobunolol hydrochloride ophthalmic solution is used to reduce elevated intraocular pressure in angle-closure glaucoma, it should be followed with a miotic and not alone.
The preservative in levobunolol hydrochloride ophthalmic solution, benzalkonium chloride, may be absorbed by soft contact lenses. Patients wearing soft (hydrophilic) contact lenses should be instructed to remove contact lenses before administration of the solution and wait at least 15 minutes after instilling levobunolol hydrochloride ophthalmic solution before reinserting soft contact lenses.
Muscle Weakness
Beta-adrenergic blockade has been reported to potentiate muscle weakness consistent with certain myasthenic symptoms (e.g., diplopia, ptosis and generalized weakness).
Drug Interactions
Although levobunolol hydrochloride ophthalmic solution used alone has little or no effect on pupil size, mydriasis resulting from concomitant therapy with levobunolol hydrochloride ophthalmic solution and epinephrine may occur.
Close observation of the patient is recommended when a beta-blocker is administered to patients receiving catecholamine-depleting drugs such as reserpine, because of possible additive effects and the production of hypotension and/or marked bradycardia, which may produce vertigo, syncope, or postural hypotension.
Patients receiving beta-adrenergic blocking agents along with either oral or intravenous calcium antagonists should be monitored for possible atrioventricular conduction disturbances, left ventricular failure and hypotension. In patients with impaired cardiac function, simultaneous use should be avoided altogether.
The concomitant use of beta-adrenergic blocking agents with digitalis and calcium antagonists may have additive effects on prolonging atrioventricular conduction time.
Phenothiazine-related compounds and beta-adrenergic blocking agents may have additive hypotensive effects due to the inhibition of each other’s metabolism.
Risk of Anaphylactic Reaction
While taking beta-blockers, patients with a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated challenge, either accidental, diagnostic, or therapeutic. Such patients may be unresponsive to the usual doses of epinephrine used to treat allergic reactions.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a lifetime oral study in mice, there were statistically significant (p≤0.05) increases in the incidence of benign leiomyomas in female mice at 200 mg/kg/day (14,000 times the recommended human dose for glaucoma), but not at 12 or 50 mg/kg/day (850 and 3,500 times the human dose). In a two year oral study of levobunolol hydrochloride in rats, there was a statistically significant (p≤0.05) increase in the incidence of benign hepatomas in male rats administered 12,800 times the recommended human dose for glaucoma. Similar differences were not observed in rats administered oral doses equivalent to 350 times to 2,000 times the recommended human dose for glaucoma.
Levobunolol did not show evidence of mutagenic activity in a battery of microbiological and mammalian in vitroand in vivoassays.
Reproduction and fertility studies in rats showed no adverse effect on male or female fertility at doses up to 1,800 times the recommended human dose for glaucoma.
Pregnancy
Fetotoxicity (as evidenced by a greater number of resorption sites) has been observed in rabbits when doses of levobunolol hydrochloride equivalent to 200 and 700 times the recommended dose for the treatment of glaucoma were given. No fetotoxic effects have been observed in similar studies with rats at up to 1,800 times the human dose for glaucoma. Teratogenic studies with levobunolol in rats at doses up to 25 mg/kg/day (1,800 times the recommended human dose for glaucoma) showed no evidence of fetal malformations. There were no adverse effects on postnatal development of offspring. It appears when results from studies using rats and studies with other beta-adrenergic blockers are examined, that the rabbit may be a particularly sensitive species. There are no adequate and well-controlled studies in pregnant women. Levobunolol hydrochloride ophthalmic solution should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
-
ADVERSE REACTIONS
In clinical trials the use of levobunolol hydrochloride ophthalmic solution has been associated with transient ocular burning and stinging in up to 1 in 3 patients, and with blepharoconjunctivitis in up to 1 in 20 patients. Decreases in heart rate and blood pressure have been reported (see CONTRAINDICATIONSand WARNINGS).
The following adverse reactions have been reported rarely with the use of levobunolol hydrochloride ophthalmic solution: iridocyclitis, headache, transient ataxia, dizziness, lethargy, urticaria, and pruritus.
Decreased corneal sensitivity has been noted in a small number of patients. Although levobunolol has minimal membrane-stabilizing activity, there remains a possibility of decreased corneal sensitivity after prolonged use.
The following additional adverse reactions have been reported either with levobunolol hydrochloride ophthalmic solution or ophthalmic use of other beta-adrenergic receptor blocking agents:
BODY AS A WHOLE:Headache, asthenia, chest pain. CARDIOVASCULAR:Bradycardia, arrhythmia, hypotension, syncope, heart block, cerebral vascular accident, cerebral ischemia, congestive heart failure, palpitation, cardiac arrest. DIGESTIVE:Nausea, diarrhea. PSYCHIATRIC:Depression, confusion, increase in signs and symptoms of myasthenia gravis, paresthesia. SKIN:Hypersensitivity, including localized and generalized rash, alopecia, Stevens-Johnson Syndrome. RESPIRATORY:Bronchospasm (predominantly in patients with pre-existing bronchospastic disease), respiratory failure, dyspnea, nasal congestion. UROGENITAL:Impotence. ENDOCRINE:Masked symptoms of hypoglycemia in insulin-dependent diabetics (see WARNINGS). SPECIAL SENSES:Signs and symptoms of keratitis or eye allergy, blepharoptosis, visual disturbances including refractive changes (due to withdrawal of miotic therapy in some cases), diplopia, ptosis, and foreign body sensation in eye.
Other reactions associated with the oral use of non-selective adrenergic receptor blocking agents should be considered potential effects with ophthalmic use of these agents.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at
1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. -
OVERDOSAGE
No data are available regarding overdosage in humans. Should accidental ocular overdosage occur, flush eye(s) with water or normal saline. If accidentally ingested, efforts to decrease further absorption may be appropriate (gastric lavage). The most common signs and symptoms to be expected with overdosage with administration of a systemic beta-adrenergic blocking agent are symptomatic bradycardia, hypotension, bronchospasm, and acute cardiac failure. Should these symptoms occur, discontinue levobunolol hydrochloride ophthalmic solution therapy and initiate appropriate supportive therapy. The following supportive measures should be considered:
1. Symptomatic bradycardia: Use atropine sulfate intravenously in a dosage of 0.25 mg to 2 mg to induce vagal blockade. If bradycardia persists, intravenous isoproterenol hydrochloride should be administered cautiously. In refractory cases the use of a transvenous cardiac pacemaker should be considered.
2. Hypotension: Use sympathomimetic pressor drug therapy, such as dopamine, dobutamine or levarterenol. In refractory cases the use of glucagon hydrochloride may be useful.
3. Bronchospasm: Use isoproterenol hydrochloride. Additional therapy with aminophylline may be considered.
4. Acute cardiac failure: Conventional therapy with digitalis, diuretics and oxygen should be instituted immediately. In refractory cases the use of intravenous aminophylline is suggested. This may be followed, if necessary, by glucagon hydrochloride which may be useful.
5. Heart block (second or third degree): Use isoproterenol hydrochloride or a transvenous cardiac pacemaker.
-
DOSAGE AND ADMINISTRATION
The recommended starting dose is one to two drops of levobunolol hydrochloride ophthalmic solution, 0.5% in the affected eye(s) once a day. In patients with more severe or uncontrolled glaucoma, levobunolol hydrochloride ophthalmic solution, 0.5% can be administered twice a day. As with any new medication, careful monitoring of patients is advised. Dosages above one drop of levobunolol hydrochloride ophthalmic solution, 0.5% twice a day are not generally more effective. If the patient's IOP is not at a satisfactory level on this regimen, concomitant therapy with other ophthalmic IOP-lowering agents can be instituted. Patients should not typically use two or more topical ophthalmic beta-adrenergic blocking agents simultaneously.
-
HOW SUPPLIED
Levobunolol hydrochloride ophthalmic solution USP, 0.5% is supplied sterile in a plastic bottle with a controlled drop tip in the following sizes:
NDC 24208-505-05 - 5 mL
NDC 24208-505-10 - 10 mL
NDC 24208-505-15 - 15 mL
Storage:Store between 15°C to 25°C (59°F to 77°F). Protect from light. Replace cap immediately after use.
KEEP OUT OF REACH OF CHILDREN.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USAManufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA© 2022 Bausch & Lomb Incorporated or its affiliates
Revised: October 2022
9117404 (Folded)
9117504 (Flat)
- PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LEVOBUNOLOL HYDROCHLORIDE
levobunolol hydrochloride solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24208-505 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOBUNOLOL HYDROCHLORIDE (UNII: O90S49LDHH) (LEVOBUNOLOL - UNII:G6317AOI7K) LEVOBUNOLOL HYDROCHLORIDE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) 0.04 mg in 1 mL POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) 14 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM METABISULFITE (UNII: 4VON5FNS3C) MONOBASIC POTASSIUM PHOSPHATE (UNII: 4J9FJ0HL51) WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24208-505-05 1 in 1 CARTON 03/04/1994 1 5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC:24208-505-10 1 in 1 CARTON 03/04/1994 2 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 3 NDC:24208-505-15 1 in 1 CARTON 03/04/1994 3 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074326 03/04/1994 Labeler - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Bausch & Lomb Incorporated 079587625 manufacture(24208-505)