Label: PRENATAL PLUS VITAMINS- prenatal with ferrous fumarate and folic acid tablet
- NDC Code(s): 39328-106-10, 39328-106-50
- Packager: Patrin Pharma, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 13, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
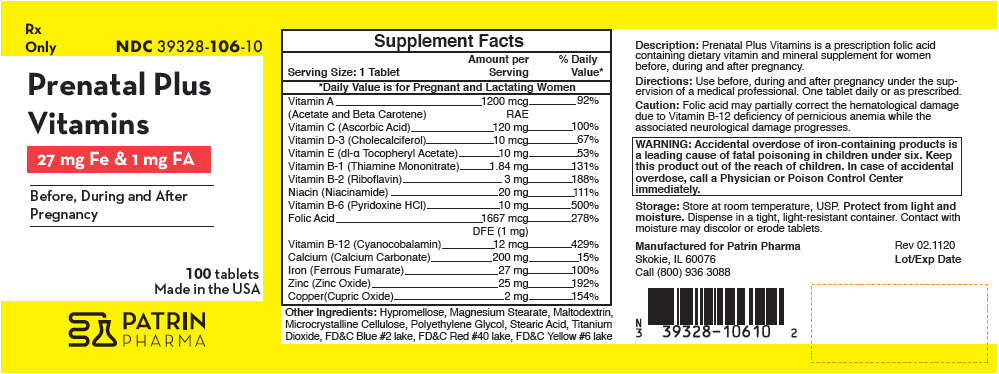
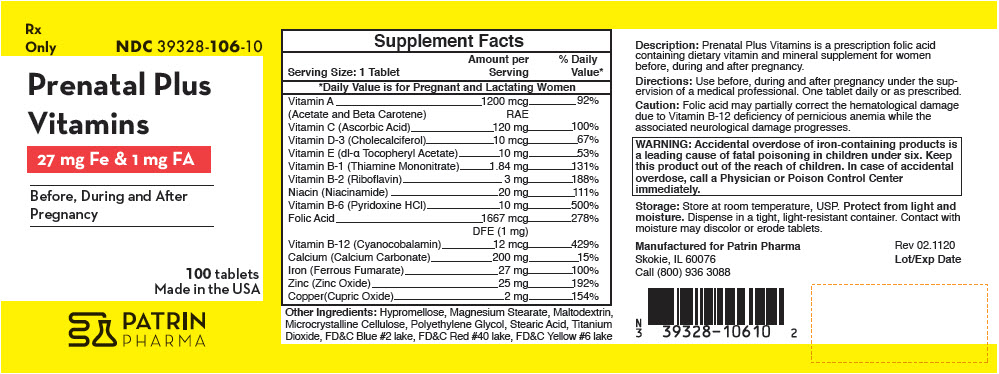
Supplement Facts Serving Size: 1 Tablet Amount per Serving % Daily Value* - *
- Daily Value is for Pregnant and Lactating Women
Vitamin A 1200 mcg 92% (Acetate and Beta Carotene) RAE Vitamin C (Ascorbic Acid) 120 mg 100% Vitamin D-3 (Cholecalciferol) 10 mcg 67% Vitamin E (dl-α Tocopheryl Acetate) 10 mg 53% Vitamin B-1 (Thiamine Mononitrate) 1.84 mg 131% Vitamin B-2 (Riboflavin) 3 mg 188% Niacin (Niacinamide) 20 mg 111% Vitamin B-6 (Pyridoxine HCl) 10 mg 500% Folic Acid 1667 mcg 278% DFE (1 mg) Vitamin B-12 (Cyanocobalamin) 12 mcg 429% Calcium (Calcium Carbonate) 200 mg 15% Iron (Ferrous Fumarate) 27 mg 100% Zinc (Zinc Oxide) 25 mg 192% Copper(Cupric Oxide) 2 mg 154% Other Ingredients: Hypromellose, Magnesium Stearate, Maltodextrin, Microcrystalline Cellulose, Polyethylene Glycol, Stearic Acid, Titanium Dioxide, FD&C Blue #2 lake, FD&C Red #40 lake, FD&C Yellow #6 lake
- Description
- Directions
- Caution
- BOXED WARNING (What is this?)
- Storage
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 100 Tablet Bottle Label
-
INGREDIENTS AND APPEARANCE
PRENATAL PLUS VITAMINS
prenatal with ferrous fumarate and folic acid tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:39328-106 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Vitamin A Acetate (UNII: 3LE3D9D6OY) (Vitamin A - UNII:81G40H8B0T) Vitamin A 924 ug Beta Carotene (UNII: 01YAE03M7J) (Beta Carotene - UNII:01YAE03M7J) Beta Carotene 276 ug Ascorbic Acid (UNII: PQ6CK8PD0R) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 120 mg Cholecalciferol (UNII: 1C6V77QF41) (Cholecalciferol - UNII:1C6V77QF41) Cholecalciferol 10 ug .Alpha.-TocopheroL Acetate, DL- (UNII: WR1WPI7EW8) (.Alpha.-Tocopherol, DL- - UNII:7QWA1RIO01) .Alpha.-Tocopherol, DL- 10 mg Thiamine Mononitrate (UNII: 8K0I04919X) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 1.84 mg Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 3 mg Niacinamide (UNII: 25X51I8RD4) (Niacinamide - UNII:25X51I8RD4) Niacinamide 20 mg Pyridoxine Hydrochloride (UNII: 68Y4CF58BV) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine Hydrochloride 10 mg Folic Acid (UNII: 935E97BOY8) (Folic Acid - UNII:935E97BOY8) Folic Acid 1667 ug Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin 12 ug Calcium Carbonate (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB) Calcium Carbonate 200 mg Ferrous Fumarate (UNII: R5L488RY0Q) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 27 mg Zinc Oxide (UNII: SOI2LOH54Z) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 25 mg Cupric Oxide (UNII: V1XJQ704R4) (CUPRIC CATION - UNII:8CBV67279L) CUPRIC CATION 2 mg Inactive Ingredients Ingredient Name Strength Hypromellose, Unspecified (UNII: 3NXW29V3WO) Magnesium Stearate (UNII: 70097M6I30) Maltodextrin (UNII: 7CVR7L4A2D) Microcrystalline Cellulose (UNII: OP1R32D61U) Polyethylene Glycol 400 (UNII: B697894SGQ) Stearic Acid (UNII: 4ELV7Z65AP) Titanium Dioxide (UNII: 15FIX9V2JP) FD&C Blue NO. 2 (UNII: L06K8R7DQK) FD&C Red NO. 40 (UNII: WZB9127XOA) FD&C Yellow NO. 6 (UNII: H77VEI93A8) Product Characteristics Color BROWN (Beige) Score no score Shape OVAL (OVAL) Size 9mm Flavor Imprint Code P;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:39328-106-10 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/01/2013 2 NDC:39328-106-50 500 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/01/2013 11/04/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 04/01/2013 Labeler - Patrin Pharma, Inc. (806841677)