Label: ATNAA ATROPINE AND PRALIDOXIME CHLORIDE AUTO-INJECTOR- atropine and pralidoxime chloride kit
- NDC Code(s): 11704-777-01
- Packager: Meridian Medical Technologies, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated July 18, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ATNAA safely and effectively. See full prescribing information for ATNAA.
ATNAA (atropine and pralidoxime chloride injection), for intramuscular use
Initial U.S. Approval: 2002INDICATIONS AND USAGE
ATNAA, a combination of atropine, a cholinergic muscarinic antagonist, and pralidoxime chloride, a cholinesterase reactivator, is indicated for the treatment of poisoning by susceptible organophosphorus nerve agents having anticholinesterase activity in adults. (1)
DOSAGE AND ADMINISTRATION
- ATNAA is intended as an initial treatment as soon as symptoms appear; definitive medical care should be sought immediately. (2.1)
- Dosage for Mild Symptoms: If a service member experiences some or all of the mild symptoms, they should self-administer one injection intramuscularly into the lateral thigh muscle or buttocks. If, at any time after the first dose, the service member develops any of the severe symptoms or if the mild symptoms are not relieved, a buddy should administer two additional injections intramuscularly in rapid succession. (2.2)
- Dosage for Severe Symptoms: If a service member has any of the severe symptoms, immediately buddy-administer three injections intramuscularly into the service member's lateral thigh muscle or buttocks in rapid succession. (2.2)
DOSAGE FORMS AND STRENGTHS
Injection: Each single-dose ATNAA autoinjector contains atropine (2.1 mg/0.7 mL) plus pralidoxime chloride (600 mg/2 mL). (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Cardiovascular (CV) Risks: Tachycardia, palpitations, premature ventricular contractions, flutter, fibrillation, etc. Use caution in individuals with known CV disease or conduction problems. (5.1)
- Heat Injury: May inhibit sweating and lead to hyperthermia; avoid excessive exercising and heat exposure. (5.2)
- Acute Glaucoma: May precipitate in susceptible individuals. (5.3)
- Urinary Retention: Administer with caution in individuals with bladder outflow obstruction. (5.4)
- Pyloric Stenosis: May convert into complete obstruction. (5.5)
- Exacerbation of Chronic Lung Disease: Atropine may cause inspiration of bronchial secretions and formation of dangerous viscid plugs in individuals with chronic lung disease; monitor respiratory status. (5.6)
ADVERSE REACTIONS
Common adverse reactions of atropine include dryness of mouth, blurred vision, dry eyes, photophobia, confusion, headache, and dizziness among others. (6.1) The common adverse reactions of pralidoxime chloride include changes in vision, dizziness, headache, drowsiness, nausea, tachycardia, increased blood pressure, muscular weakness, dry mouth, emesis, rash, dry skin, hyperventilation, decreased renal function, excitement, manic behavior, and transient elevation of liver enzymes and creatine phosphokinase. (6.2)
To report SUSPECTED ADVERSE REACTIONS, contact Meridian Medical Technologies®, LLC at 1-833-739-0945 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Succinylcholine and Mivacurium: Accelerated reversal of neuromuscular blocking effects may occur; monitor with concomitant administration. (7.1)
USE IN SPECIFIC POPULATIONS
- Geriatric individuals may be more susceptible to the effects of atropine. (8.5)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Information
2.2 Dosage Information
2.3 Administration Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Cardiovascular Risks
5.2 Heat Injury
5.3 Acute Glaucoma
5.4 Urinary Retention
5.5 Pyloric Stenosis
5.6 Exacerbation of Chronic Lung Disease
6 ADVERSE REACTIONS
6.1 Atropine
6.2 Pralidoxime Chloride
6.3 Injection Site
6.4 Inadvertent Injection
7 DRUG INTERACTIONS
7.1 Succinylcholine and Mivacurium
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
10.1 Symptoms
10.2 Treatment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Information
- Three (3) ATNAA single-dose autoinjectors should be available for use by each service member at risk for organophosphorus nerve agent poisoning; one (1) for mild symptoms plus two (2) more for severe symptoms [see Dosage and Administration (2.2)]. Note that individuals may not have all symptoms included under the mild or severe symptom category.
- For optimal reactivation of organophosphorus-inhibited cholinesterase, the ATNAA should be administered as soon as possible after appearance of symptoms of organophosphorus nerve agent poisoning.
- ATNAA should be self- or buddy–administered by service members after donning protective mask and hood at the first sign of a chemical attack, and only if some or all of the mild symptoms of organophosphorus nerve agent exposure are present.
- Only administer ATNAA to service members experiencing symptoms of organophosphorus nerve agent poisoning in a situation where exposure is known or suspected. The ATNAA autoinjector is intended as an initial treatment of the symptoms of organophosphorus nerve agent poisoning as soon as symptoms appear; definitive medical care should be sought immediately.
- Close supervision of all treated service members is indicated for at least 48 to 72 hours.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit [see Dosage Forms and Strengths (3)].
2.2 Dosage Information
Dosage for Mild Symptoms
First Dose: If you experience some or all of the mild symptoms of nerve agent exposure listed in Table 1, self-administer one (1) ATNAA injection intramuscularly into the lateral thigh muscle or buttocks.
Wait 10 to 15 minutes for ATNAA to take effect. If, after 10 to 15 minutes, the symptoms of organophosphorus nerve agent poisoning are not relieved, seek someone else to check your symptoms. Another service member must administer the second and third injections.
Additional Doses: If you encounter a service member suffering from severe symptoms of organophosphorus nerve agent poisoning listed in Table 1 and one ATNAA has been self-administered, administer two (2) additional ATNAA injections intramuscularly in rapid succession into the casualty's lateral thigh muscle or buttocks.
Dosage for Severe Symptoms
Casualties with severe symptoms may experience most or all of the mild symptoms of organophosphorus nerve agent poisoning, plus most or all of the severe symptoms listed in Table 1.
If a service member is encountered suffering from severe signs of organophosphorus nerve agent poisoning and ATNAA self-aid has not been administered, immediately administer in rapid succession three (3) ATNAA injections into the casualty's lateral thigh muscle or buttocks.
Table 1. Common Symptoms of Organophosphorus Nerve Agent Exposure Mild Symptoms Severe Symptoms - Unexplained runny nose
- Unexplained sudden headache
- Sudden drooling
- Difficulty in seeing (dimness of vision and miosis)
- Tightness of chest or difficulty in breathing
- Wheezing and coughing
- Localized sweating and muscular twitching in the area of contaminated skin
- Stomach cramps
- Nausea, with or without vomiting
- Tachycardia followed by bradycardia
- Strange or confused behavior
- Increased wheezing and increased difficulty in breathing
- Severely pinpointed pupils
- Red eyes with tearing
- Vomiting
- Severe muscular twitching and general weakness
- Involuntary urination and defecation
- Convulsions
- Unconsciousness
- Respiratory failure
- Bradycardia
2.3 Administration Instructions
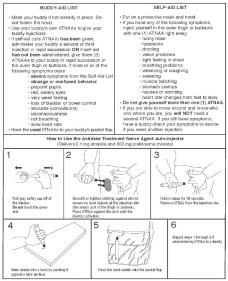
The following instructions should be given to service members for the administration of ATNAA single-dose injections.
Self-Aid
- Administer one (1) ATNAA into your lateral thigh muscle or buttocks as follows:
- Remove gray safety cap from back end.
- Place front end on outer thigh and push hard until injector functions. Hold firmly in place for ten seconds.
- Using a hard surface, bend needle into hook. Push ejected needle through a pocket flap (or other thick and conspicuous part of outer clothing).
- Wait 10 to 15 minutes for the antidote to take effect. If you are able to ambulate, know who you are, and where you are, you will NOT need a second injection.
Giving yourself a second set of injections may cause an overdose of the ATNAA which could result in incapacitation. - If symptoms of organophosphorus nerve agent poisoning are not relieved after administering one injection, seek someone else to check your symptoms. A buddy must administer the second and third injections, if needed.
Buddy-Aid
If you encounter a service member suffering from any of the severe symptoms of organophosphorus nerve agent poisoning, render the following aid:
- Mask the casualty, if necessary. Do not fasten the hood.
- If self-aid (one ATNAA injection) has been administered, administer in rapid succession two (2) additional ATNAA injections into the casualty's lateral thigh muscle or buttocks.
Use the casualty's own ATNAAs when providing aid. Do not use your own autoinjectors on a casualty. If you do, you may not have any antidote available when needed for self-aid. - If self-aid (one ATNAA injection) has not been administered, administer in rapid succession three (3) ATNAA injections into the casualty's lateral thigh muscle or buttocks.
-
3 DOSAGE FORMS AND STRENGTHS
Injection: Each single-dose ATNAA autoinjector contains the following in two separate chambers:
- front chamber (visible): a clear, colorless to yellow, sterile solution of atropine (2.1 mg/0.7 mL)
- back chamber (not visible): a clear, colorless to yellow, sterile solution of pralidoxime chloride (600 mg/2 mL) equivalent to pralidoxime (476.6 mg/2 mL)
When activated, ATNAA sequentially administers both drugs intramuscularly through a single needle in one injection.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Cardiovascular Risks
Cardiovascular adverse reactions reported in the literature for atropine include, but are not limited to, sinus tachycardia, palpitations, premature ventricular contractions, atrial flutter, atrial fibrillation, ventricular flutter, ventricular fibrillation, cardiac syncope, asystole, and myocardial infarction. In individuals with a recent myocardial infarction and/or severe coronary artery disease, there is a possibility that atropine-induced tachycardia may cause ischemia, extend or initiate myocardial infarcts, and stimulate ventricular ectopy and fibrillation. ATNAA should be used with caution in individuals with known cardiovascular disease or cardiac conduction problems.
5.2 Heat Injury
Atropine may inhibit sweating which, in a warm environment or with excessive exercise, can lead to hyperthermia and heat injury. To the extent feasible, avoid excessive exercise and heat exposure [see Overdosage (10.2)].
5.3 Acute Glaucoma
Atropine should be administered with caution in individuals at risk for acute glaucoma.
5.4 Urinary Retention
Atropine should be administered with caution in individuals with clinically significant bladder outflow obstruction because of the risk of urinary retention.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
- Cardiovascular Risks [see Warnings and Precautions (5.1)]
- Heat Injury [see Warnings and Precautions (5.2)]
- Acute Glaucoma [see Warnings and Precautions (5.3)]
- Urinary Retention [see Warnings and Precautions (5.4)]
- Pyloric Stenosis [see Warnings and Precautions (5.5)]
- Exacerbation of Chronic Lung Disease [see Warnings and Precautions (5.6)]
The following adverse reactions associated with the use of atropine and pralidoxime chloride were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Atropine
Because ATNAA contains pralidoxime chloride, which may potentiate the effect of atropine, signs of atropinization may occur earlier than might be expected when atropine is used alone.
Common adverse reactions of atropine can be attributed to its antimuscarinic action. These include dryness of the mouth, blurred vision, dry eyes, photophobia, confusion, headache, dizziness, tachycardia, palpitations, flushing, urinary hesitancy or retention, constipation, abdominal pain, abdominal distention, nausea and vomiting, loss of libido, and impotence. Anhidrosis may produce heat intolerance and impairment of temperature regulation in a hot environment. Dysphagia, paralytic ileus, acute angle closure glaucoma, maculopapular rash, petechial rash, and scarletiniform rash have also been reported. Adverse cardiac reactions, including arrhythmias and myocardial infarction, have been reported with atropine [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2)].
Larger doses of atropine may produce central nervous system effects such as restlessness, tremor, fatigue, locomotor difficulties, delirium, and hallucinations [see Overdosage (10.1)].
Hypersensitivity reactions will occasionally occur; these are usually seen as skin rashes, and may progress to exfoliation. Anaphylactic reaction and laryngospasm are rare.
6.2 Pralidoxime Chloride
Pralidoxime chloride can cause blurred vision, diplopia and impaired accommodation, dizziness, headache, drowsiness, nausea, tachycardia, increased systolic and diastolic blood pressure [see Clinical Pharmacology 12.2)], muscular weakness, dry mouth, emesis, rash, dry skin, hyperventilation, decreased renal function, and decreased sweating when given parenterally to normal adult volunteers who have not been exposed to anticholinesterase poisons.
In several cases of organophosphorus poisoning, excitement and manic behavior have occurred immediately following recovery of consciousness, in either the presence or absence of pralidoxime chloride administration. However, similar behavior has not been reported in subjects given pralidoxime chloride in the absence of organophosphorus poisoning.
Elevations in AST and/or ALT enzyme levels were observed in 1 of 6 normal adult volunteers given 1200 mg of pralidoxime chloride intramuscularly, and in 4 of 6 adult volunteers given 1800 mg intramuscularly. Levels returned to normal in about two weeks. Transient elevations in creatine kinase were observed in all normal volunteers given the drug.
6.4 Inadvertent Injection
In cases where ATNAA is inadvertently administered to service members who are not poisoned with susceptible organophosphorus nerve agents having anticholinesterase activity, the following effects on their ability to function normally may occur.
Atropine 2 mg IM, roughly the equivalent of one ATNAA injection, when given to healthy male volunteers, is associated with minimal effects on visual, motor, and mental functions, though unsteadiness walking and difficulty concentrating may occur. Atropine reduces body sweating and increases body temperature, particularly with exercise and under hot conditions.
Atropine 4 mg IM, roughly the equivalent of two ATNAA injections, when given to healthy male volunteers, is associated with impaired visual acuity, visual near point accommodation, logical reasoning, digital recall, learning, and cognitive reaction time. Ability to read is reduced or lost. Subjects are unsteady and need to concentrate on walking. These effects begin about 15 minutes to one hour or more post-dose.
Atropine 6 mg IM, roughly the equivalent of three ATNAA injections, when given to healthy male volunteers, is associated with the effects described above plus additional central effects including poor coordination, poor attention span, and visual hallucinations (colored flashes) in many subjects. Frank visual hallucinations, auditory hallucinations, disorientation, and ataxia occur in some subjects. Skilled and labor-intense tasks are performed more slowly and less efficiently. Decision making takes longer and is sometimes impaired.
It is unclear if the above data, obtained from studies of healthy male subjects, can be extrapolated to other populations. In the elderly and individuals with co-morbid conditions, the effects of ≥2 mg atropine on the ability to see, walk, and think properly are unstudied; effects may be greater in susceptible populations.
Service members who are mistakenly injected with ATNAA should avoid potentially dangerous overheating, avoid vigorous physical activity, and seek medical attention as soon as feasible.
-
7 DRUG INTERACTIONS
7.1 Succinylcholine and Mivacurium
Since pralidoxime chloride in ATNAA reactivates cholinesterases and succinylcholine and mivacurium are metabolized by cholinesterases, service members poisoned by susceptible organophosphorus nerve agents having anticholinesterase activity who have received ATNAA may exhibit accelerated reversal of the neuromuscular blocking effects of succinylcholine and mivacurium (relative to poisoned service member who has not received pralidoxime). Monitor for neuromuscular effects with concomitant administration.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Atropine readily crosses the placental barrier and enters fetal circulation. There are no adequate data on the developmental risk associated with the use of atropine, pralidoxime chloride, or the combination in pregnant women. Adequate animal reproduction studies have not been conducted with atropine, pralidoxime chloride, or the combination. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
8.2 Lactation
Risk Summary
Atropine has been reported to be excreted in human milk. It is not known whether pralidoxime chloride is excreted in human milk. There are no data on the effects of atropine or pralidoxime chloride on the breastfed infant or the effects of the drugs on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ATNAA and any potential adverse effects on the breastfed infant from ATNAA or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of ATNAA in pediatric patients have not been established.
8.5 Geriatric Use
Geriatric individuals may be more susceptible to the effects of atropine [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Pralidoxime chloride can cause decreased renal function [see Adverse Reactions (6.2)]. Individuals with severe renal impairment may require less frequent doses after the initial dose.
-
10 OVERDOSAGE
10.1 Symptoms
Atropine
Manifestations of atropine overdose are dose-related and include flushing, dry skin and mucous membranes, tachycardia, widely dilated pupils that are poorly responsive to light, blurred vision, and fever (which can sometimes be dangerously elevated). Locomotor difficulties, disorientation, hallucinations, delirium, confusion, agitation, coma, and central depression can occur and may last 48 hours or longer. In instances of severe atropine intoxication, respiratory depression, coma, circulatory collapse, and death may occur.
Pralidoxime Chloride
It may be difficult to differentiate adverse events caused by pralidoxime chloride from those caused by organophosphorus nerve agent poisoning. Symptoms of pralidoxime chloride overdose may include dizziness, blurred vision, diplopia, headache, impaired accommodation, nausea, and tachycardia. Transient hypertension caused by pralidoxime chloride may last several hours.
10.2 Treatment
For atropine overdose, supportive treatment should be administered. If respiration is depressed, artificial respiration with oxygen is necessary. Ice bags, a hypothermia blanket, or other methods of cooling may be required to reduce atropine-induced fever, especially in pediatric patients [see Use in Specific Populations (8.4)]. Catheterization may be necessary if urinary retention occurs. Since atropine elimination largely takes place through the kidney, urinary output must be maintained and increased if possible; intravenous fluids may be indicated. Because of atropine-induced photophobia, the room should be darkened.
A benzodiazepine may be needed to control marked excitement and convulsions. However, large doses for sedation should be avoided because the central nervous system depressant effect may coincide with the depressant effect occurring late in severe atropine poisoning. Barbiturates are potentiated by the anticholinesterases; therefore, barbiturates should be used cautiously in the treatment of convulsions. Central nervous system stimulants are not recommended.
-
11 DESCRIPTION
Each prefilled ATNAA is a single-dose autoinjector that provides an intramuscular dose of atropine, a cholinergic muscarinic antagonist, and pralidoxime chloride, a cholinesterase reactivator, in a self-contained unit, specifically designed for automatic self- or buddy-administration by military personnel.
When activated, each ATNAA injection delivers the following:
- 2.1 mg of atropine in 0.7 mL of sterile, pyrogen-free solution containing 12.47 mg glycerin, not more than 2.8 mg phenol, 3.05 mg sodium citrate dihydrate, 3.5 mg citric acid monohydrate, and Water for Injection. The pH range is 4.0 – 5.0.
- 600 mg of pralidoxime chloride equivalent to 476.6 mg of pralidoxime in 2 mL of sterile, pyrogen-free solution containing 40 mg benzyl alcohol, 22.5 mg glycine, and Water for Injection. The pH is adjusted with hydrochloric acid. The pH range is 2.0 – 3.0.
After ATNAA has been activated, the empty autoinjector should be disposed of properly [see Dosage and Administration (2.2)]. It cannot be refilled, nor can the protruding needle be retracted.
Atropine occurs as white crystals, usually needle-like, or as a white, crystalline powder. It is slightly soluble in water with a molecular weight of 289.38. Atropine, a naturally occurring belladonna alkaloid, is a racemic mixture of equal parts of d- and l-hyoscyamine, with activity due almost entirely to the levo isomer of the drug.
Chemically, atropine is designated as 1αH,5αH-Tropan-3α-ol (±)-tropate. Its empirical formula is C17H23NO3 and its structural formula is as follows:
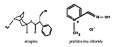
Pralidoxime chloride is an odorless, white to pale-yellow crystalline powder, freely soluble in water, with a molecular weight of 172.61. Chemically, pralidoxime chloride is designated as 2-formyl-1-methylpyridinium chloride oxime. Its empirical formula is C7H9CIN2O and its structural formula is indicated above.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Atropine
Atropine competitively blocks the effects of acetylcholine, including excess acetylcholine due to organophosphorus nerve agent poisoning, at muscarinic cholinergic receptors on smooth muscle, cardiac muscle, secretory gland cells, and in peripheral autonomic ganglia and the central nervous system.
Pralidoxime Chloride
Pralidoxime chloride reactivates acetylcholinesterase which has been inactivated by phosphorylation due to susceptible organophosphorus nerve agents having anticholinesterase activity . However, pralidoxime chloride does not reactivate acetylcholinesterase inactivated by all organophosphorus nerve agents (e.g. soman). Pralidoxime chloride cannot reactivate phosphorylated acetylcholinesterases that have undergone a further chemical reaction known as "aging." Reactivated acetylcholinesterase hydrolyzes excess acetylcholine resulting from organophosphorus nerve agent poisoning to help restore impaired cholinergic neural function. Reactivation is clinically important because only a small proportion of active acetylcholinesterase is needed to maintain vital functions.
12.2 Pharmacodynamics
Atropine
Atropine reduces secretions in the mouth and respiratory passages, relieves airway constriction, and may reduce centrally-mediated respiratory paralysis. In severe organophosphorus nerve agent poisoning, a fully atropinized patient may develop or continue to have respiratory failure and may require artificial respiration and suctioning of airway secretions. Atropine may cause thickening of secretions.
Atropine-induced parasympathetic inhibition may be preceded by a transient phase of stimulation, especially on the heart where small doses first slow the rate before characteristic tachycardia develops due to paralysis of vagal control. Atropine increases heart rate and reduces atrioventricular conduction time. Adequate atropine doses can prevent or abolish bradycardia or asystole produced by organophosphorus nerve agents.
Atropine may decrease the degree of partial heart block which can occur after organophosphorus poisoning. In some patients with complete heart block, atropine may accelerate the idioventricular rate; in others, the rate is stabilized. In some patients with conduction defects, atropine may cause paradoxical atrioventricular (A-V) block and nodal rhythm.
Atropine will not act on the neuromuscular junction and has no effect on muscle paralysis or weakness, fasciculations or tremors; pralidoxime is intended to treat these symptoms.
Systemic doses of atropine slightly raise systolic and lower diastolic pressures and can produce significant postural hypotension. Such doses also slightly increase cardiac output and decrease central venous pressure. Atropine can dilate cutaneous blood vessels, particularly the "blush" area (atropine flush), and may inhibit sweating, thereby causing hyperthermia, particularly in a warm environment or with exercise [see Warnings and Precautions(5.2)].
Pralidoxime Chloride
Pralidoxime chloride has its most critical effect in relieving respiratory muscle paralysis. Because pralidoxime chloride is less effective in relieving depression of the respiratory center, atropine is always required concomitantly to block the effect of accumulated acetylcholine at this site. Pralidoxime chloride has a minor role in relieving muscarinic signs and symptoms, such as salivation or bronchospasm.
ATNAA temporarily increases blood pressure, a known effect of pralidoxime chloride. In a study of 24 healthy young adults administered a single dose of atropine and pralidoxime chloride auto-injector intramuscularly (approximately 9 mg/kg pralidoxime chloride), diastolic blood pressure increased from baseline by 11 ± 14 mm Hg (mean ± SD), and systolic blood pressure increased by 16 ± 19 mm Hg, at 15 minutes post-dose. Blood pressures remained elevated at these approximate levels through one hour post-dose, began to decrease at two hours post-dose, and were near pre-dose baseline at four hours post-dose.
12.3 Pharmacokinetics
Atropine
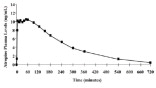
Atropine is well absorbed after intramuscular administration. Single dose ATNAA pharmacokinetic data for atropine are shown in Figure 1. The intramuscular injection site was the antero-lateral thigh.
Mean atropine plasma concentrations shown in Figure 1 indicate a plateau beginning at about 5 minutes post-dose and extending through 60 minutes post-dose.
Figure 1. Mean Atropine Plasma Concentrations After a Single ATNAA Intramuscular Injection in 24 Healthy Adult Subjects [Men (N=12), Women (N=12)]
Pralidoxime Chloride
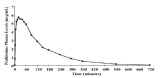
Pralidoxime chloride is well absorbed after intramuscular injection. ATNAA single dose pharmacokinetic data for pralidoxime chloride 600 mg are provided in Figure 2.
Figure 2. Mean Pralidoxime Plasma Concentrations After a Single ATNAA Intramuscular Injection in 24 Healthy Adult Subjects [Men (N=12), Women (N=12)]
The pharmacokinetic properties of the components of ATNAA are presented in Table 2.
Table 2. Pharmacokinetic Properties of the Components of ATNAA Following Intramuscular Administration in Healthy Subjects Pharmacokinetics related to: Atropine Pralidoxime Absorption Cmax (mean ± standard deviation) 13 ± 3 ng/mL 7 ± 3 mcg/mL Tmax (mean ± standard deviation) 31 ± 30 minutes 28 ± 15 minutes Distribution Protein binding 14 to 22% to plasma proteins Not appreciably bound to serum proteins Elimination T½ 2.4 ± 0.3 hours 2 ± 1 hours Major route of excretion Urinary Urinary Percentage of dose excreted in urine 50 to 60% as unchanged drug. About 17 to 28% eliminated in the first 100 minutes. 72 to 94% as unchanged drug. About 57 to 70% eliminated in the first 30 minutes, partly as metabolite. Specific Populations
Renal and Hepatic Impairment
The pharmacokinetics of atropine or pralidoxime have not been evaluated in subjects with renal or hepatic impairment.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
ATNAA is indicated for short-term emergency use only, and no adequate studies regarding the carcinogenic potential of atropine or pralidoxime chloride have been conducted.
Mutagenesis
Studies to assess the mutagenic potential of atropine or pralidoxime chloride have not been conducted.
Impairment of Fertility
Atropine:
In studies in which male rats were orally administered atropine (62.5 to 125 mg/kg) for one week prior to mating and throughout a 5-day mating period with untreated females, a dose-related decrease in fertility was observed. A no-effect dose for male reproductive toxicity was not established. The lowest dose tested was 290 times (on a mg/m2 basis) the dose of atropine in a single application of ATNAA (2.1 mg).
Fertility studies of atropine in females have not been conducted.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Each single-dose ATNAA autoinjector contains atropine (2.1 mg/0.7 mL; colorless to yellow solution, visible in front chamber) and pralidoxime chloride (600 mg/2mL, equivalent to pralidoxime 476.6 mg/2 mL; colorless to yellow solution, not visible in rear chamber). ATNAA, NDC-11704-777-01, is supplied through the Directorate of Medical Materiel, Defense Supply Center, Philadelphia.
Each ATNAA is supplied in a pouch that provides protection from light.
-
17 PATIENT COUNSELING INFORMATION
Use by Military Personnel
ATNAA is intended for use by Military Personnel. See the illustrated Instruction Sheet for Military Personnel.
-
SPL UNCLASSIFIED SECTION
Manufactured by:
Meridian Medical Technologies®, LLC
St. Louis, MO 63146Distributed by:
Defense Logistics Agency Troop Support, Philadelphia
Medical Directorate
700 Robbins Ave
Philadelphia, PA 19111Manufactured for:
U.S. Army Medical Research and Development Command
ATTN: FCMR-ORA
1430 Veterans Drive
Fort Detrick, MD 21702-501207/2022
- INSTRUCTIONS FOR USE
-
PRINCIPAL DISPLAY PANEL - Kit Carton Label
ANTIDOTE TREATMENT
NERVE AGENT, AUTO-INJECTORFor Use in Nerve Agent Poisoning Only
(atropine and pralidoxime chloride injection) Each single-dose auto-injector
delivers an intramuscular injection of 2.1 mg/0.7 mL of atropine and
600 mg/2 mL of pralidoxime chloride equivalent to 476.6 mg pralidoxime.Distributed by: Defense
Logistics Agency Troop
Support, Philadelphia
Medical Directorate
Manufactured for: U.S.
Army Medical Research
and Development
Command, by Meridian
Medical Technologies,LLC1
2
3
10 SECONDSATNAA Utilizes
Binaject® Technology
0002163Sterile Solution for Intramuscular Use Only
NSN 6505-01-362-7427
Rx Only
Store at 25°C (77°F); excursions
permitted to 15 - 30°C (59 - 86°F).
Keep From Freezing. Protect From Light. -
INGREDIENTS AND APPEARANCE
ATNAA ATROPINE AND PRALIDOXIME CHLORIDE AUTO-INJECTOR
atropine and pralidoxime chloride kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:11704-777 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11704-777-01 1 in 1 CARTON; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 01/17/2002 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 SYRINGE, GLASS 0.7 mL Part 2 1 SYRINGE, GLASS 2 mL Part 1 of 2 ATNAA ATROPINE AND PRALIDOXIME CHLORIDE AUTO-INJECTOR
atropine injectionProduct Information Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength atropine (UNII: 7C0697DR9I) (atropine - UNII:7C0697DR9I) atropine 2.1 mg in 0.7 mL Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) Citric Acid Monohydrate (UNII: 2968PHW8QP) Phenol (UNII: 339NCG44TV) Water (UNII: 059QF0KO0R) Sodium Citrate, Unspecified Form (UNII: 1Q73Q2JULR) Nitrogen (UNII: N762921K75) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.7 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021175 01/17/2002 Part 2 of 2 ATNAA ATROPINE AND PRALIDOXIME CHLORIDE AUTO-INJECTOR
pralidoxime chloride injectionProduct Information Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength pralidoxime chloride (UNII: 38X7XS076H) (pralidoxime - UNII:P7MU9UTP52) pralidoxime chloride 600 mg in 2 mL Inactive Ingredients Ingredient Name Strength Benzyl Alcohol (UNII: LKG8494WBH) Glycine (UNII: TE7660XO1C) Water (UNII: 059QF0KO0R) Hydrochloric Acid (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021175 01/17/2002 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021175 01/17/2002 Labeler - Meridian Medical Technologies, LLC (049504624) Establishment Name Address ID/FEI Business Operations Meridian Medical Technologies, LLC 049504624 MANUFACTURE(11704-777) , LABEL(11704-777) , PACK(11704-777) , ANALYSIS(11704-777) , STERILIZE(11704-777)