Label: ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (PIMIENTA CALIENTE) - BROWN- octinoxate and oxybenzone lipstick
ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON GLAMOUR) - BROWN- octinoxate and oxybenzone lipstick
ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (BORGONA SEXY) - PURPLE- octinoxate and oxybenzone lip .......
ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (BORGONA SEXY EXOTIC) - PURPLE- octinoxate and oxybenzone lipstick
ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON GLAMOUR EXOTIC) - BROWN- octinoxate and oxybenzone lipstick
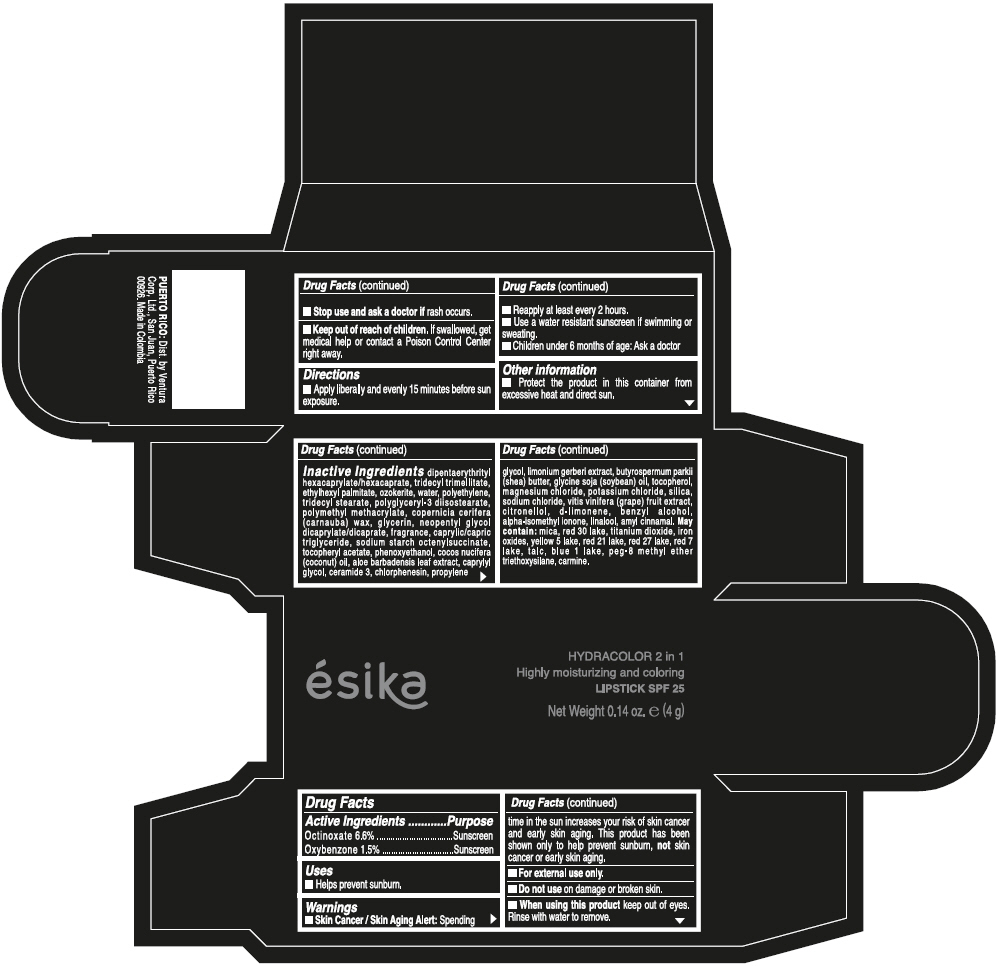
ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25- octinoxate and oxybenzone kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 13537-475-01, 13537-475-02, 13537-476-01, 13537-476-02, view more13537-477-01, 13537-477-02, 13537-478-01, 13537-478-02, 13537-479-01, 13537-479-02, 13537-804-01, 13537-804-02, 13537-805-01, 13537-805-02, 13537-806-01, 13537-806-02, 13537-807-01, 13537-807-02, 13537-808-01, 13537-808-02, 13537-809-01, 13537-809-02, 13537-810-01, 13537-810-02, 13537-811-01, 13537-811-02, 13537-812-01, 13537-812-02, 13537-813-01, 13537-813-02, 13537-814-01, 13537-814-02, 13537-815-01, 13537-815-02, 13537-816-01, 13537-816-02, 13537-817-01, 13537-817-02, 13537-818-01, 13537-818-02, 13537-819-01, 13537-819-02, 13537-820-01, 13537-820-02, 13537-821-01, 13537-821-02, 13537-822-01, 13537-822-02, 13537-823-01, 13537-823-02, 13537-824-01, 13537-824-02, 13537-825-01, 13537-825-02, 13537-826-01, 13537-826-02, 13537-827-01, 13537-827-02, 13537-828-01, 13537-828-02, 13537-829-01, 13537-829-02, 13537-830-01, 13537-830-02, 13537-831-01, 13537-831-02, 13537-832-01, 13537-832-02, 13537-833-01, 13537-833-02, 13537-834-01, 13537-834-02, 13537-835-01, 13537-835-02, 13537-836-01, 13537-836-02, 13537-837-01, 13537-837-02, 13537-838-01, 13537-838-02, 13537-839-01, 13537-839-02, 13537-840-01, 13537-840-02, 13537-880-01, 13537-880-02 - Packager: Ventura Corporation LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 9, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
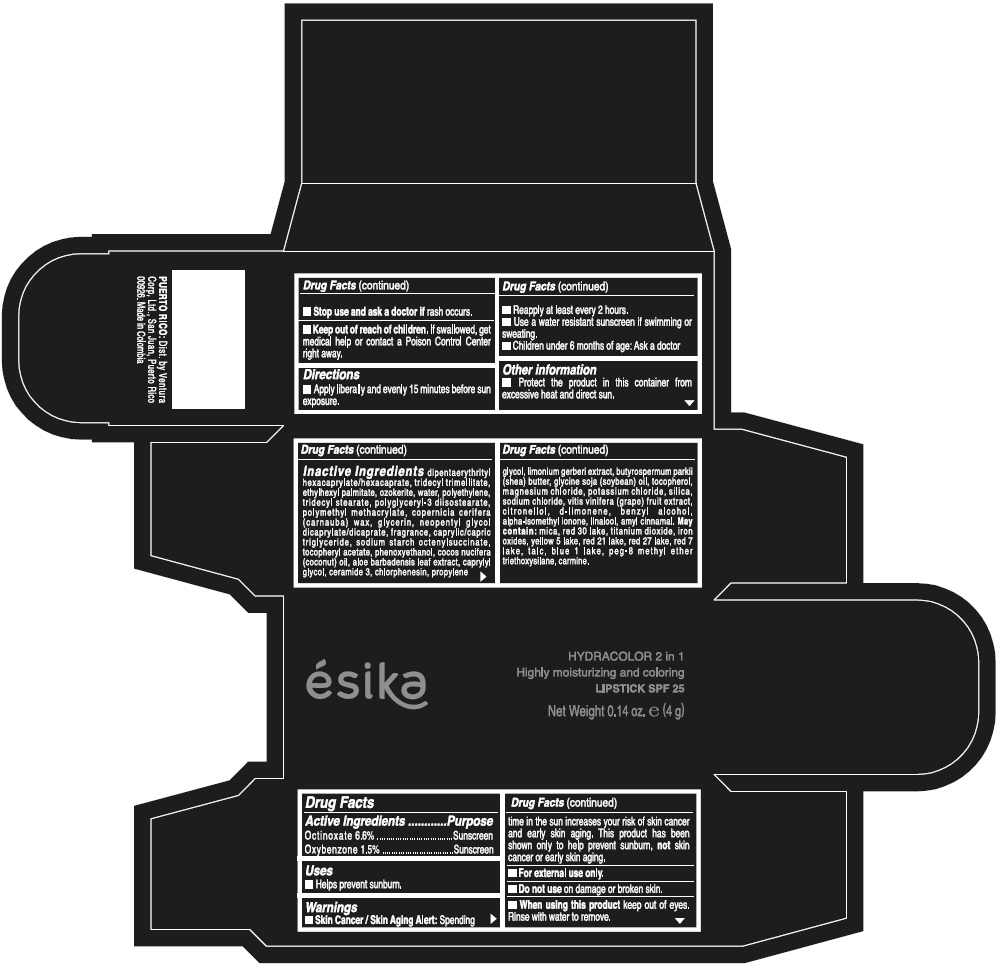
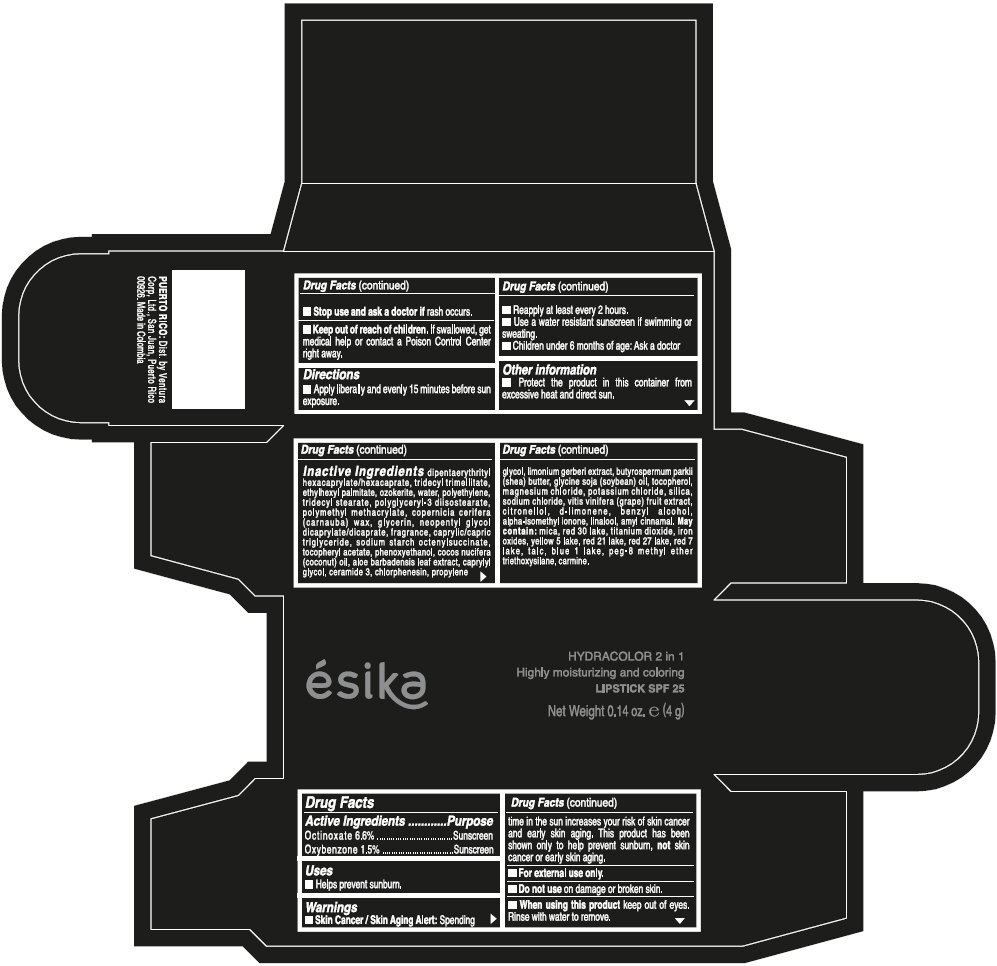
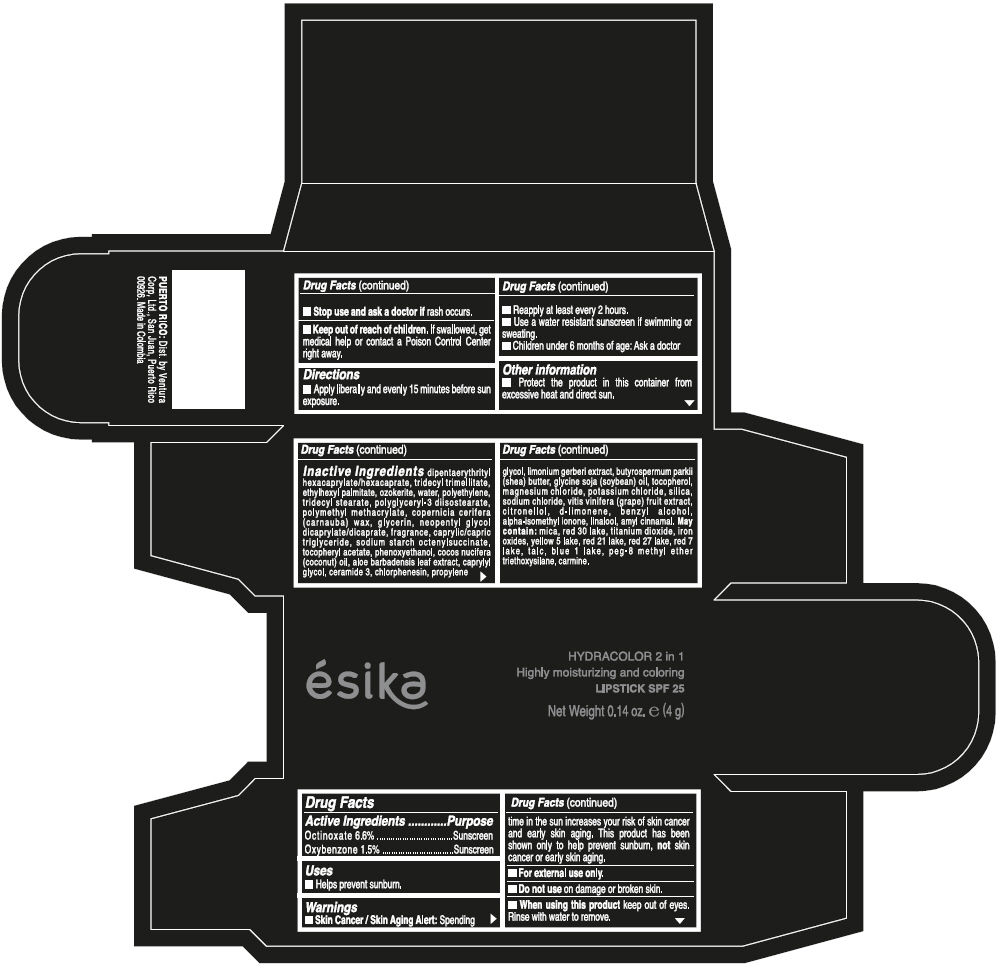
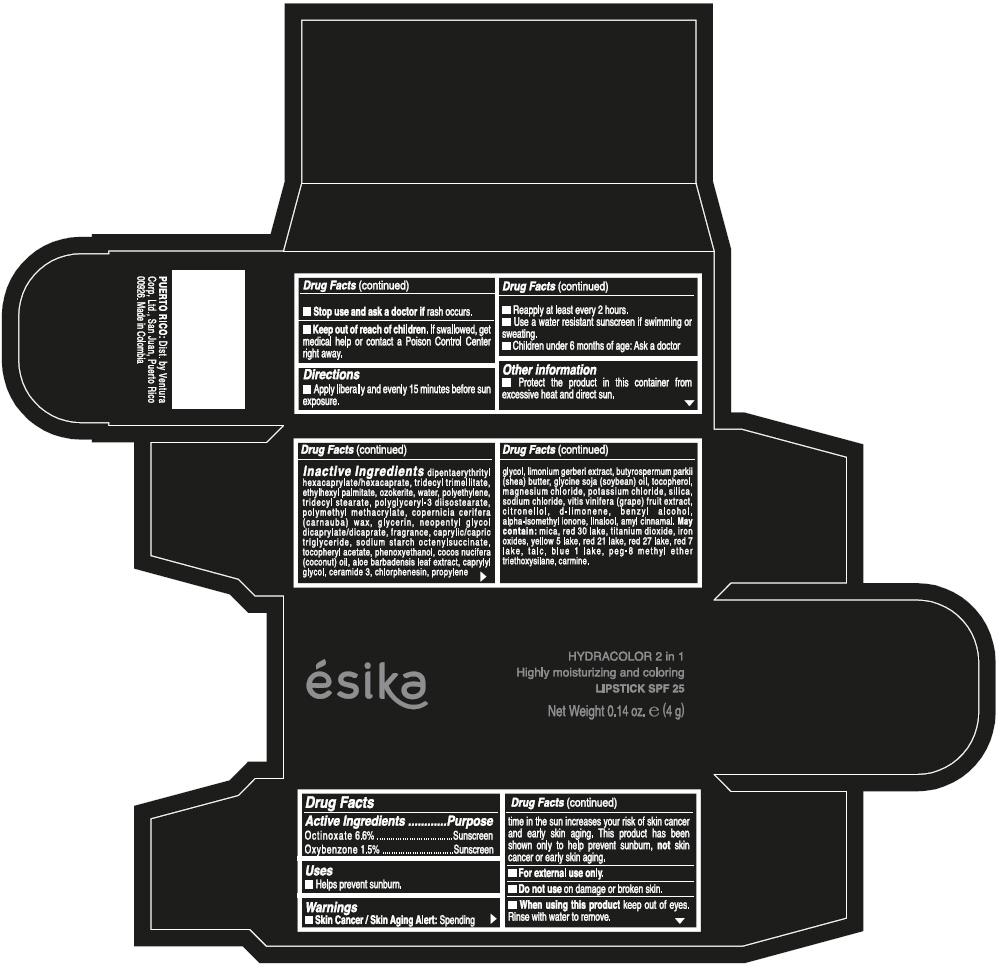
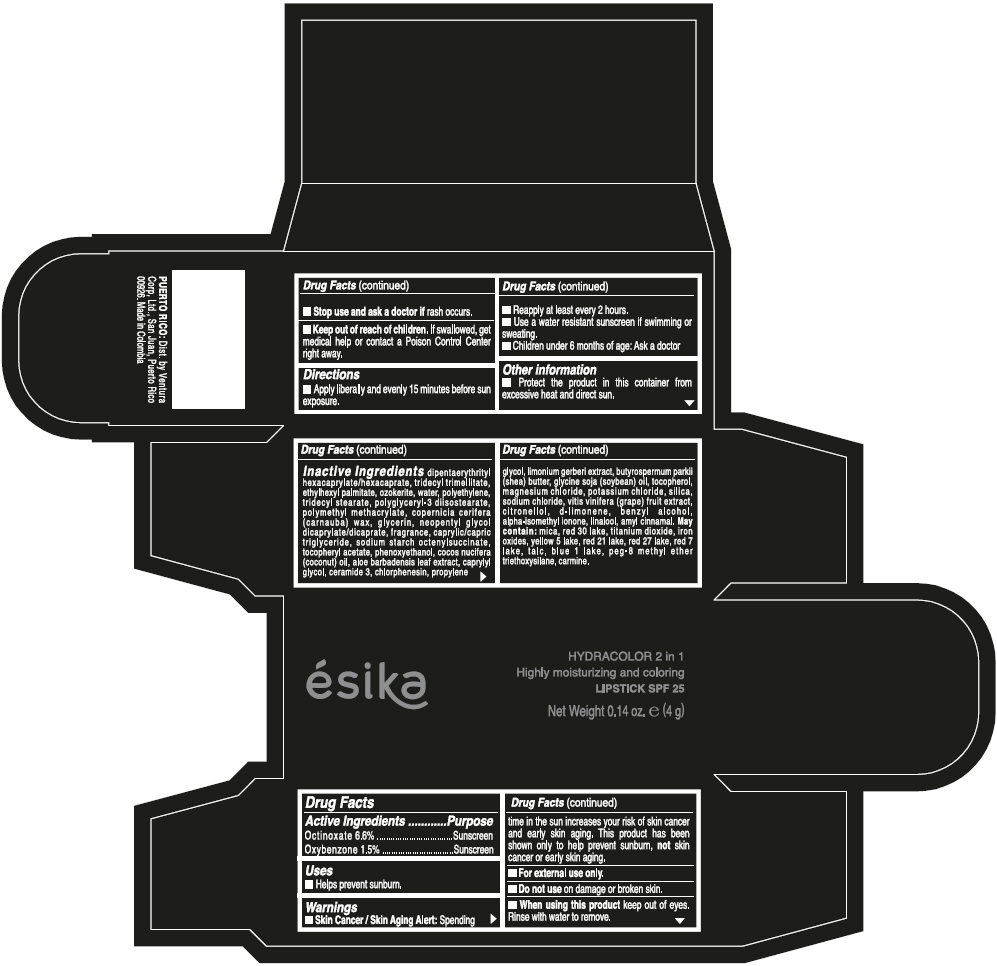
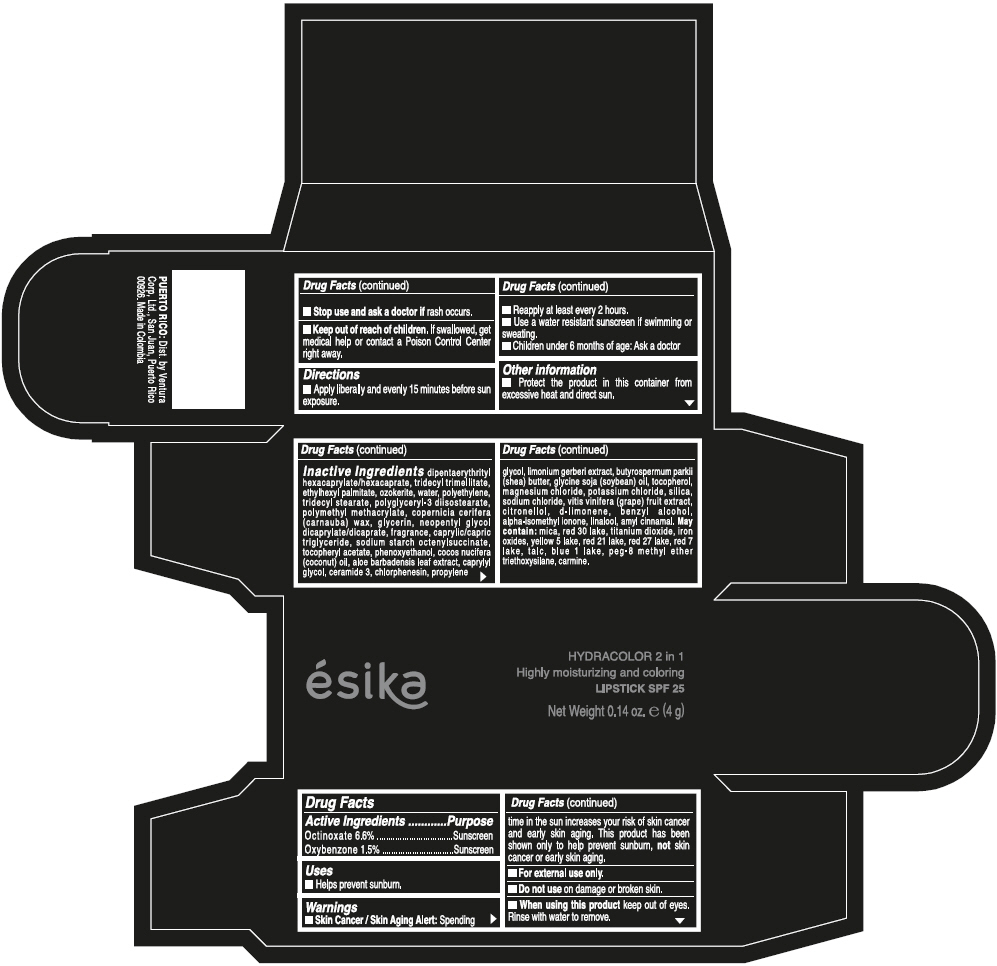
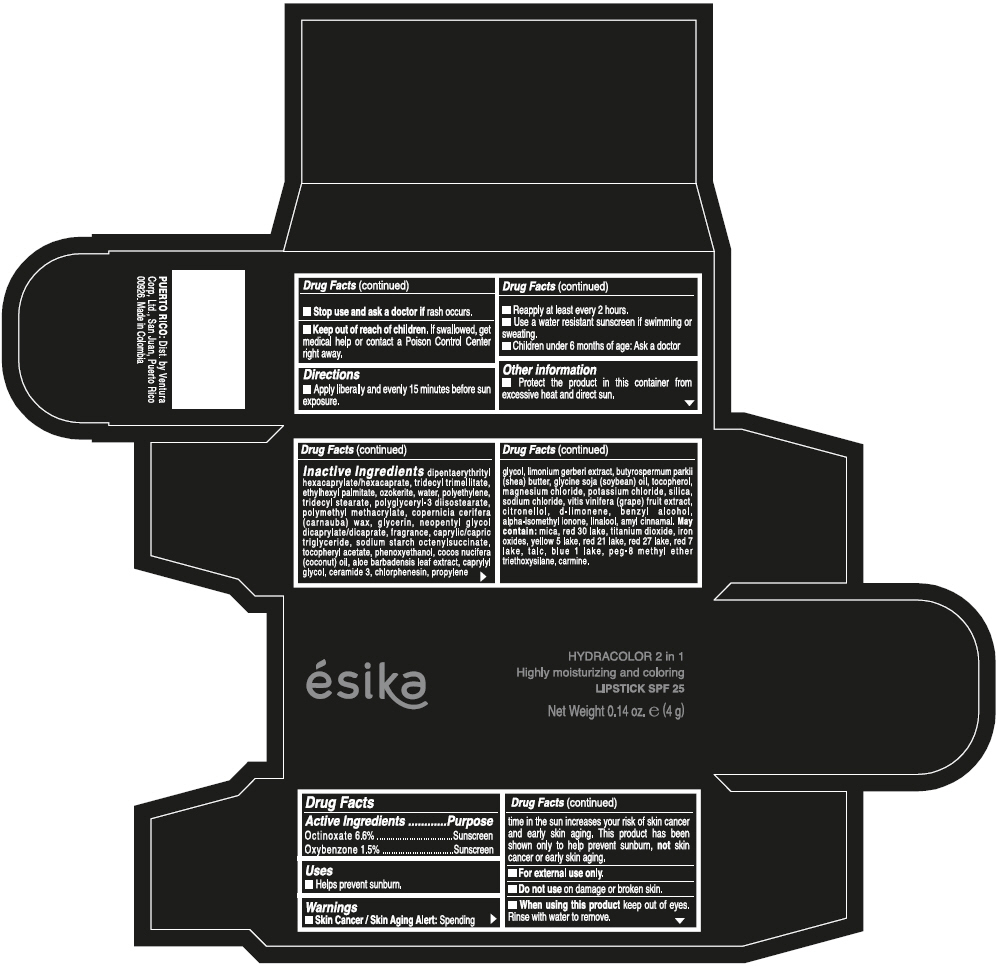
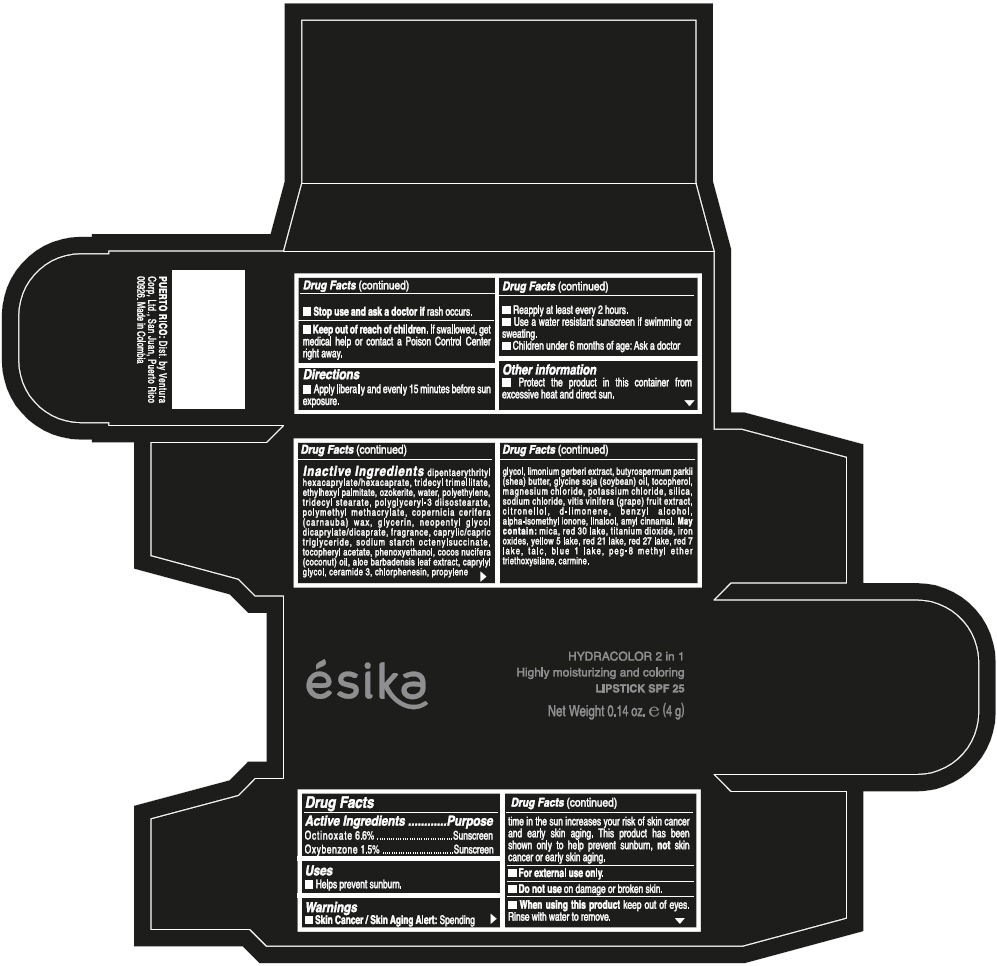
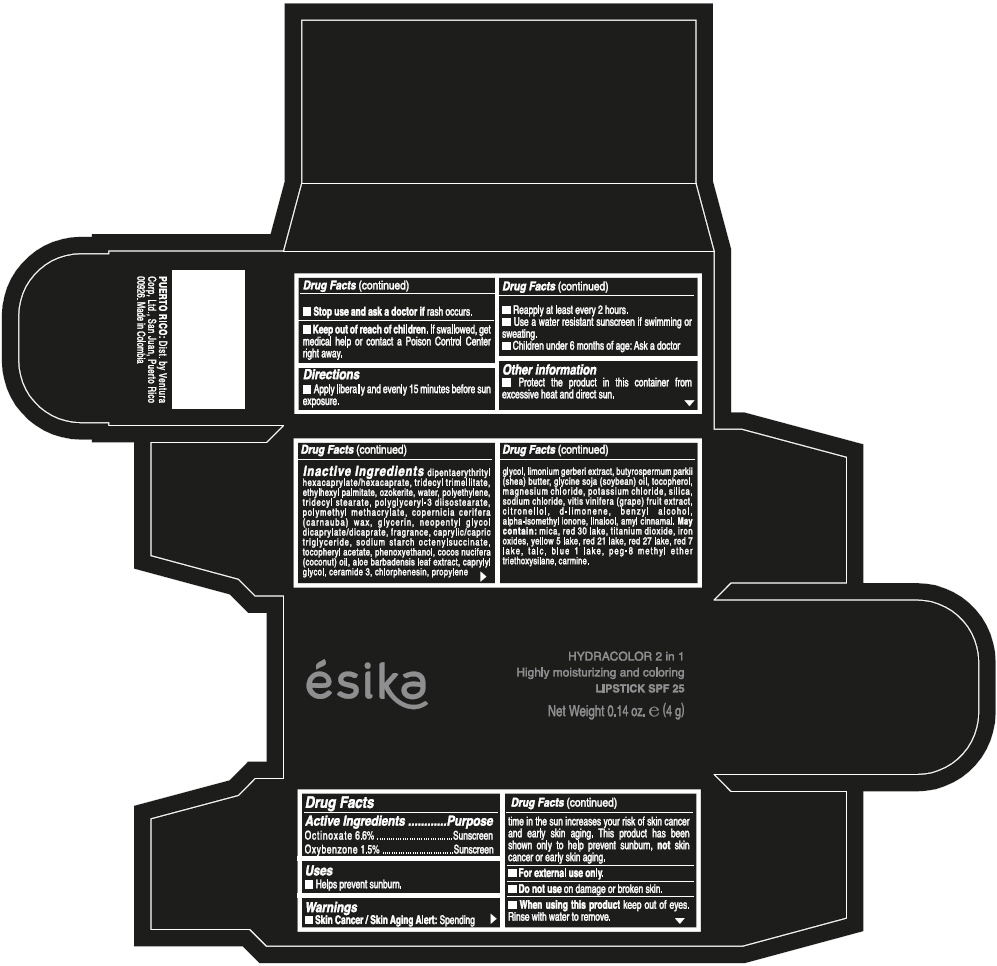
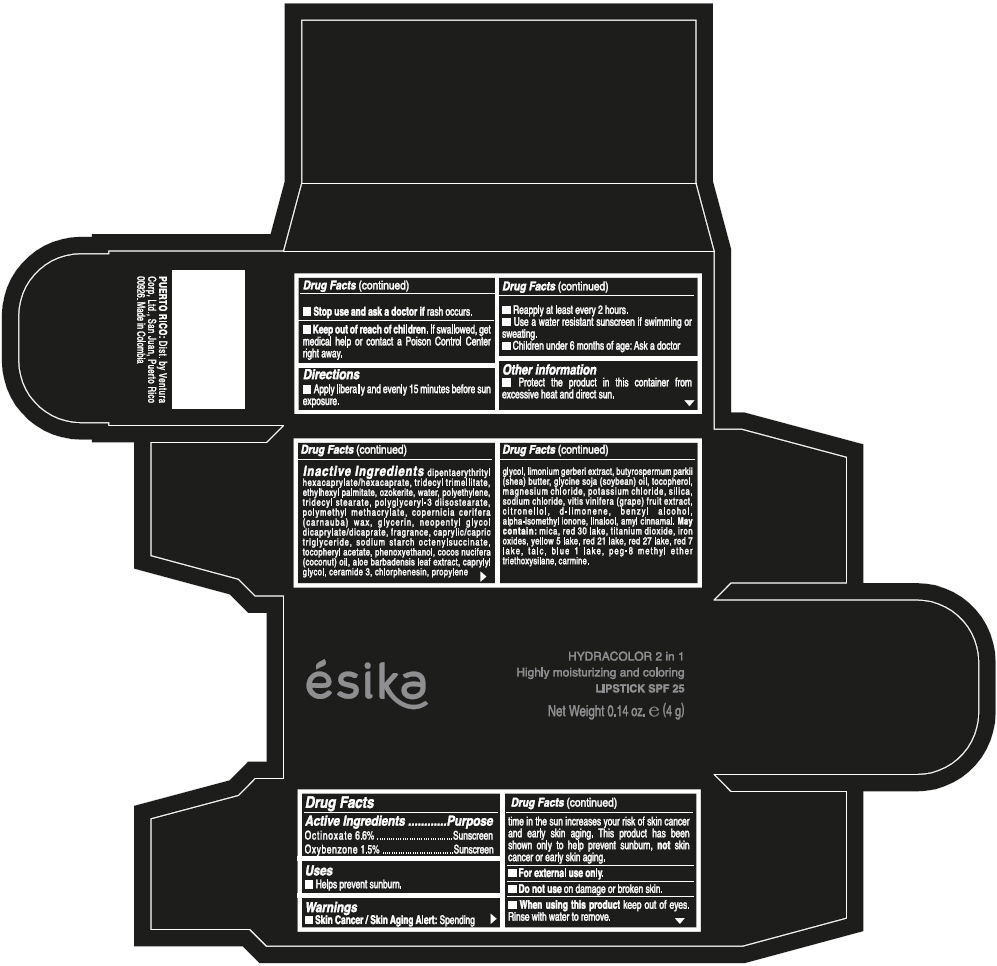
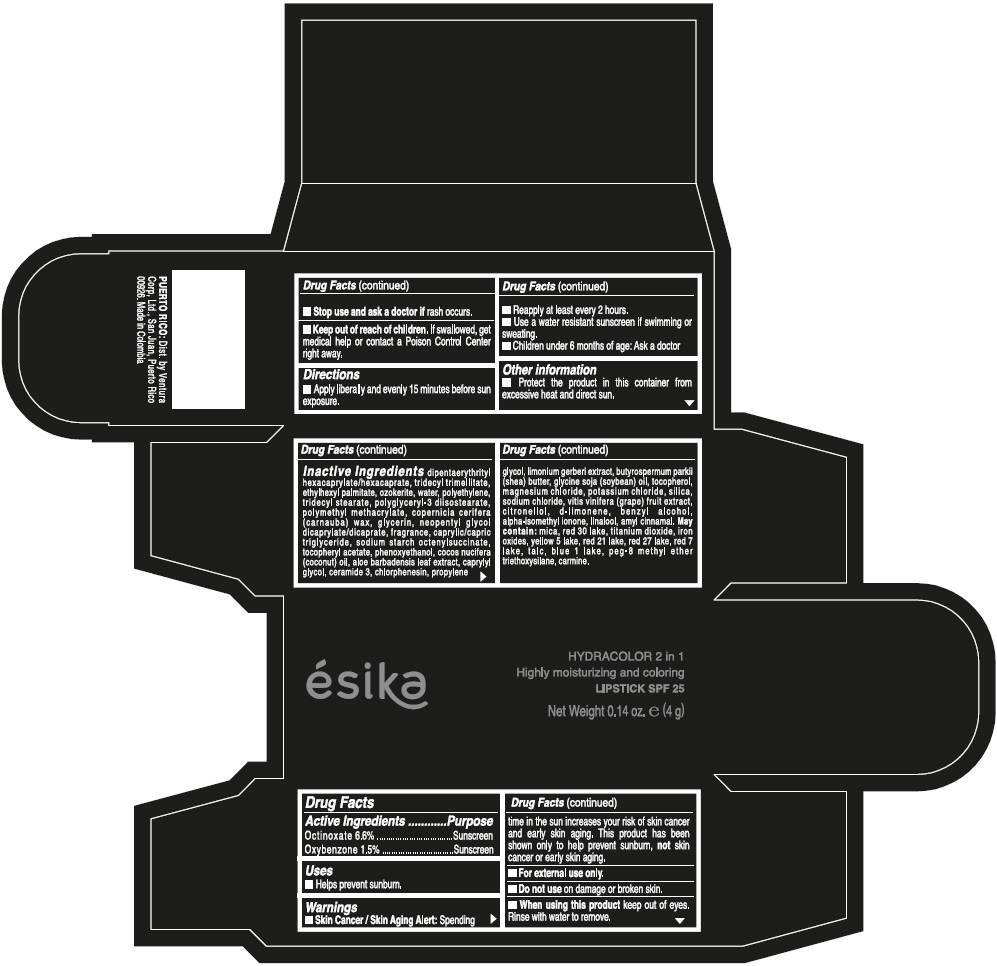
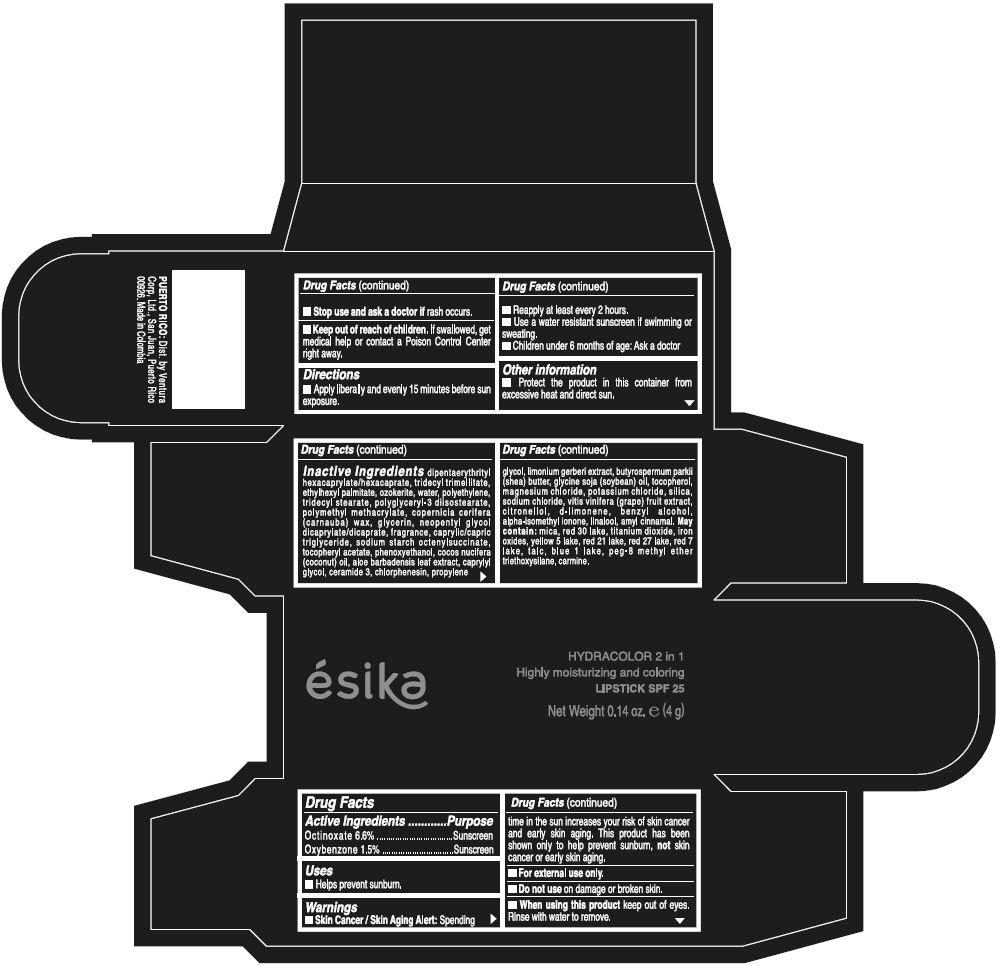
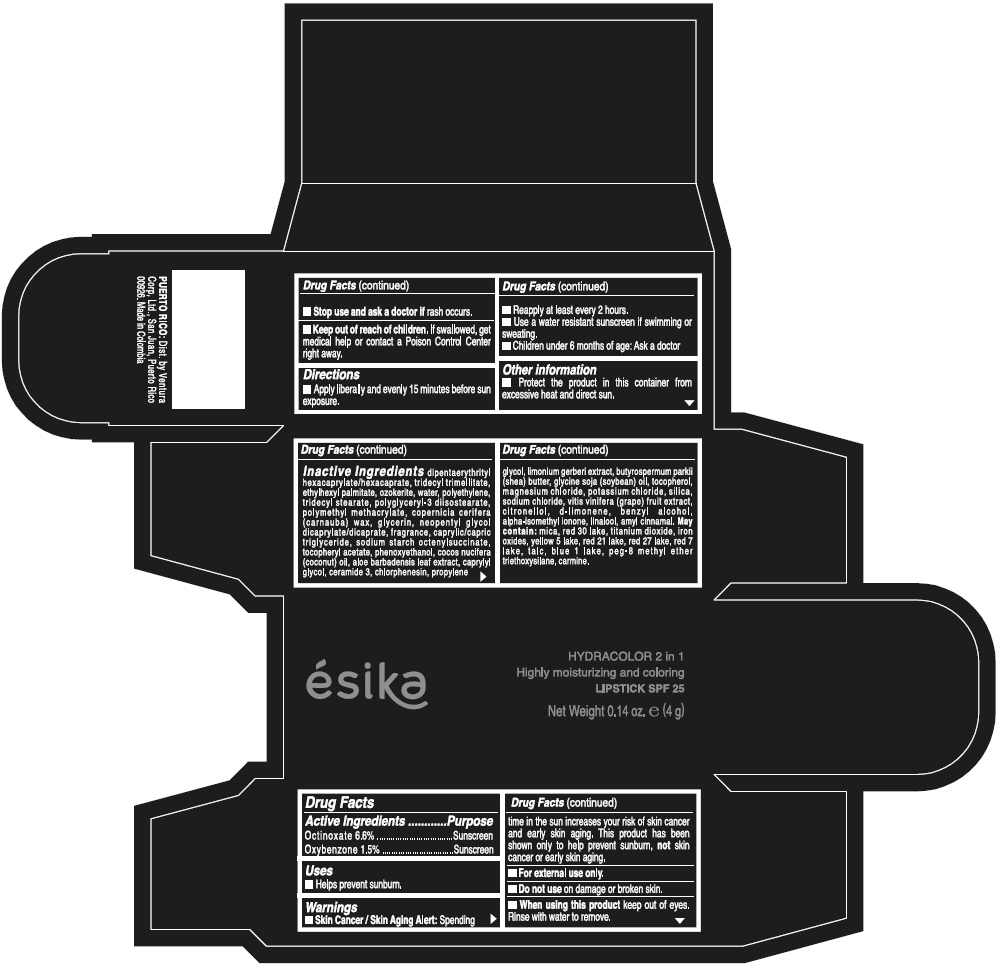
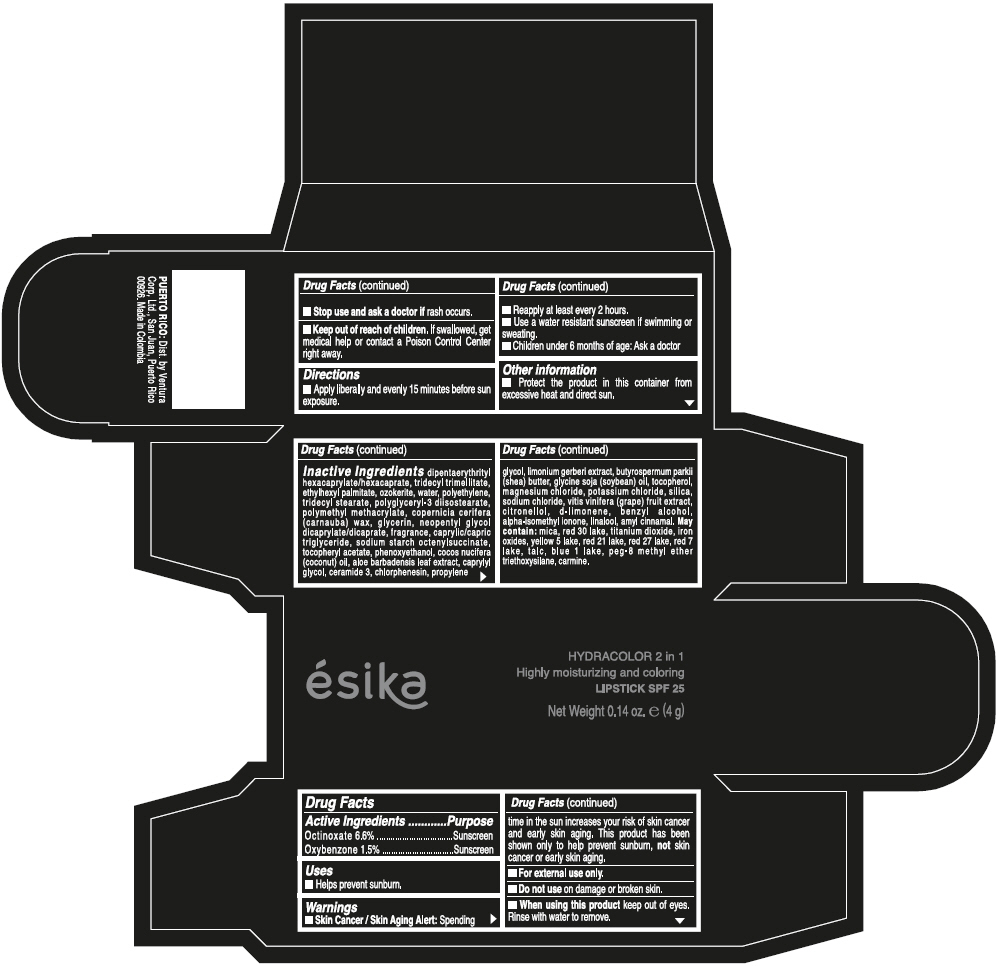
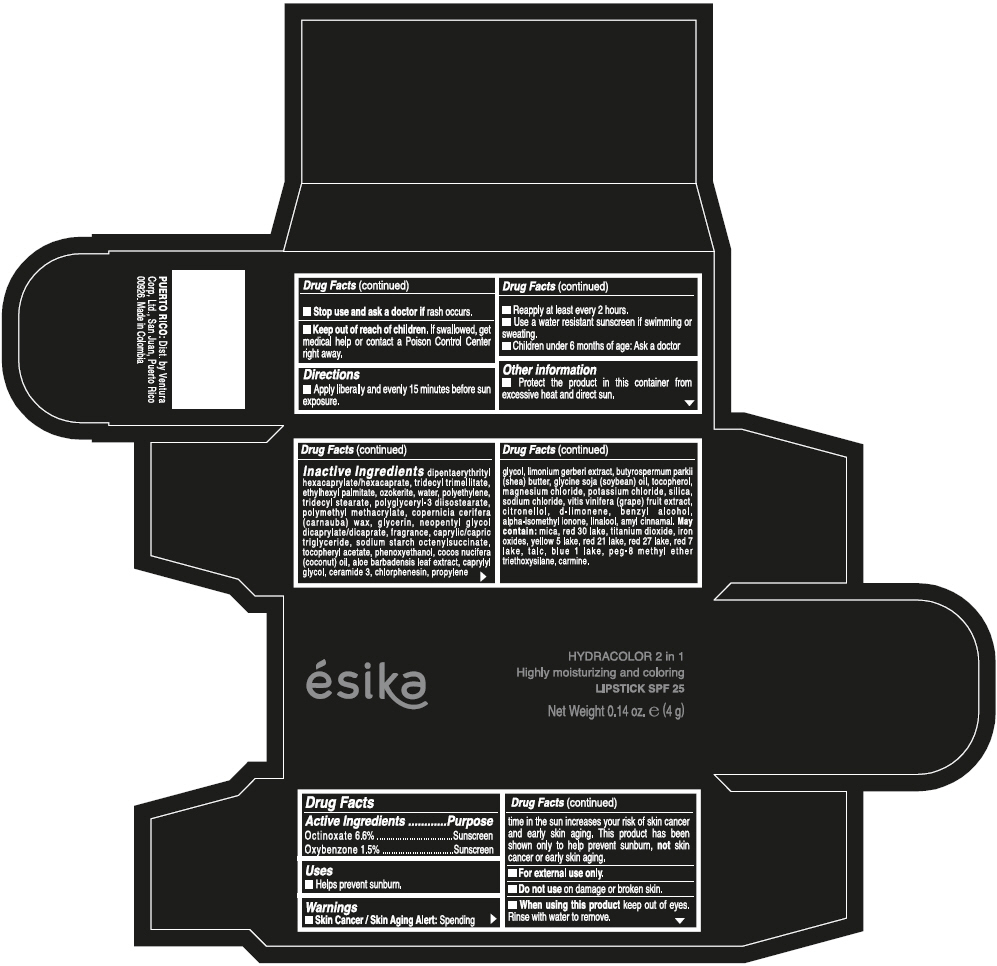
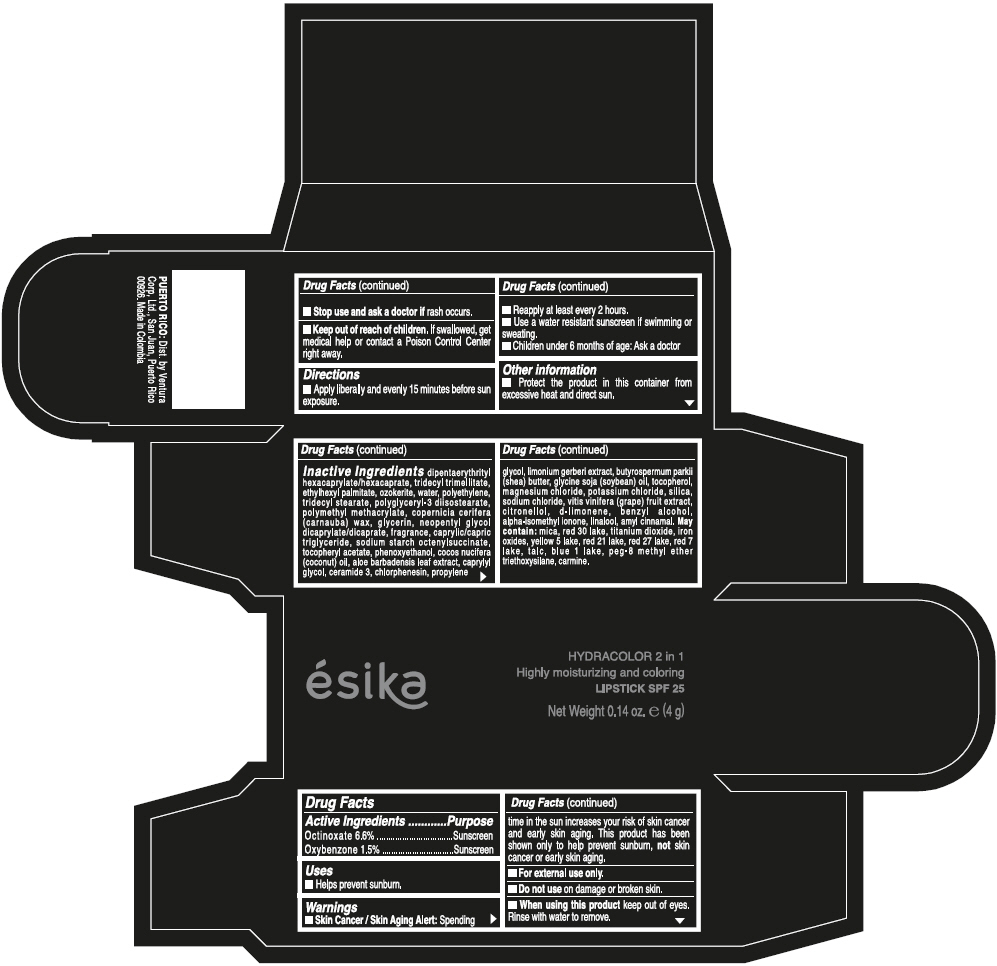
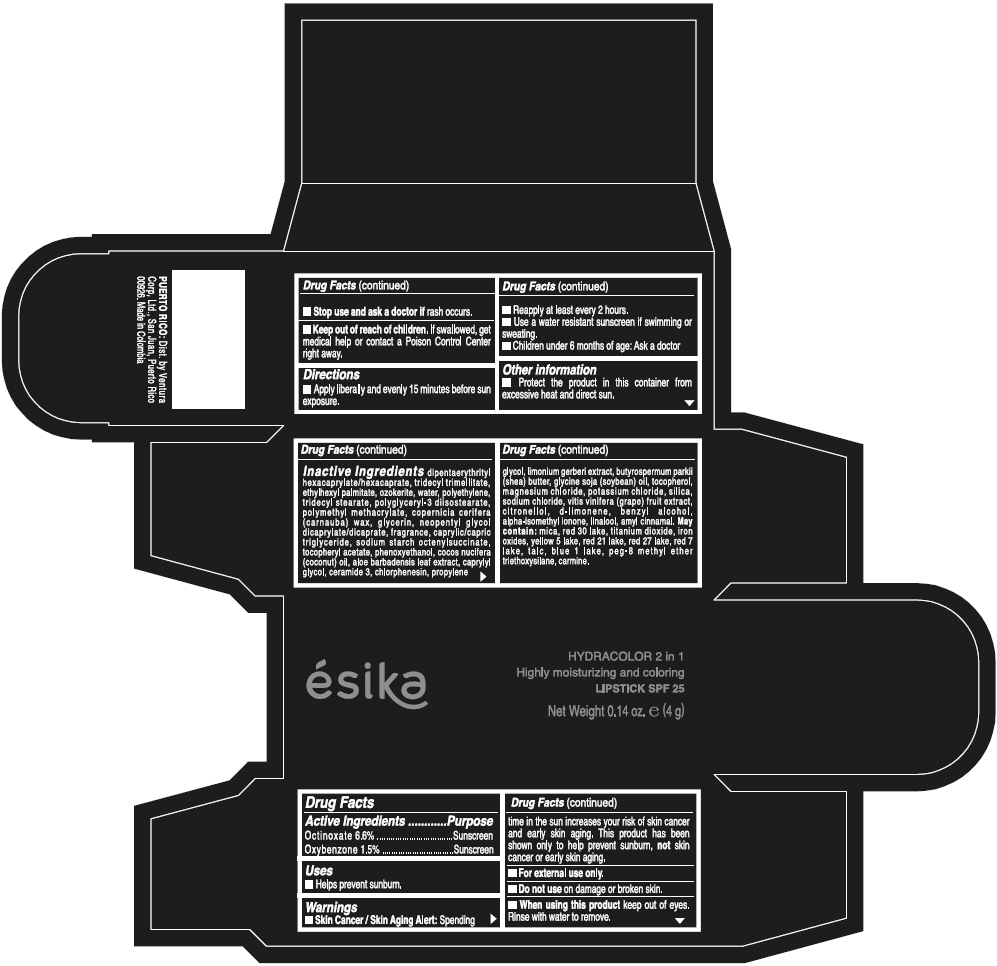
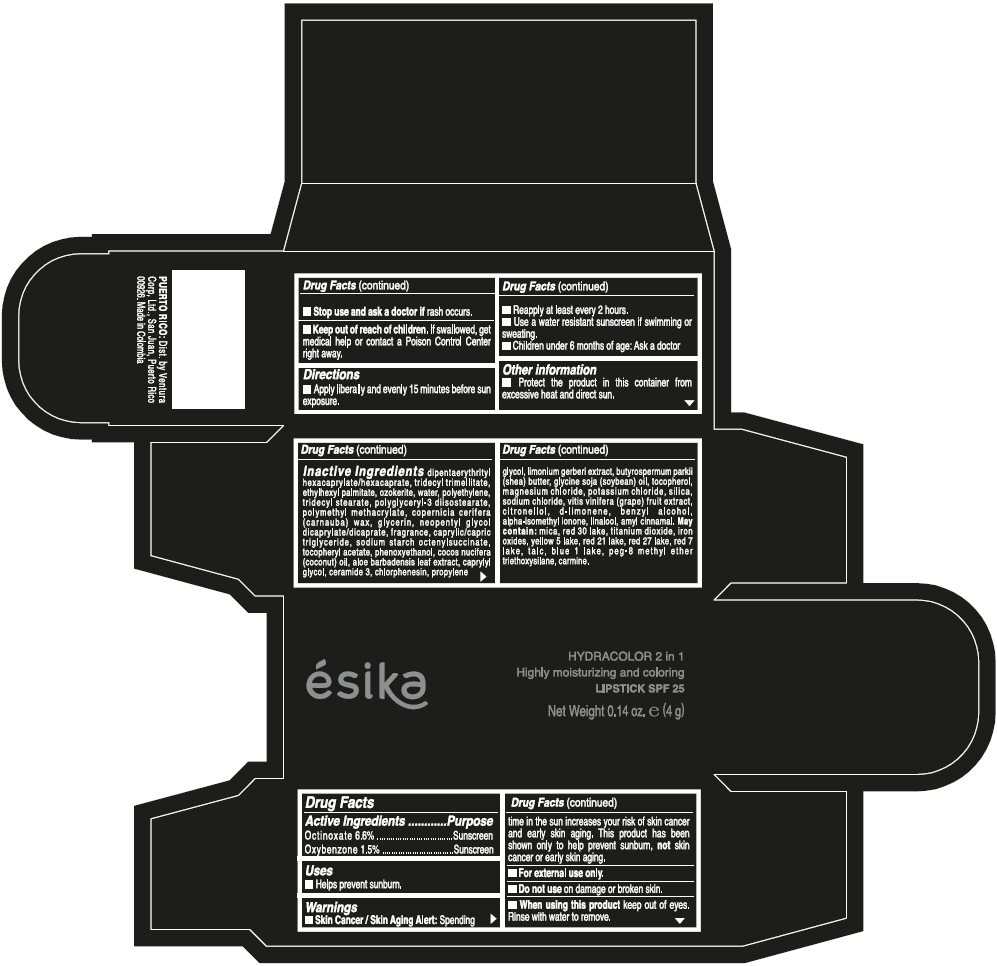
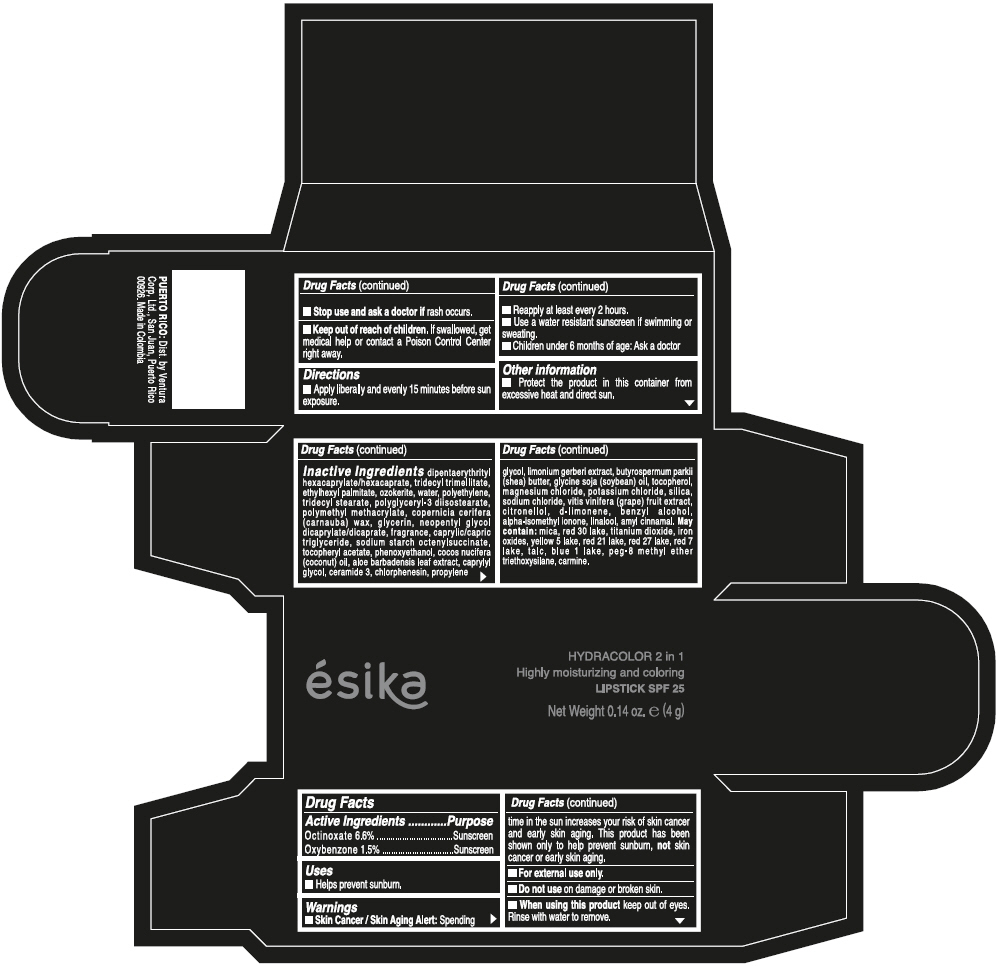
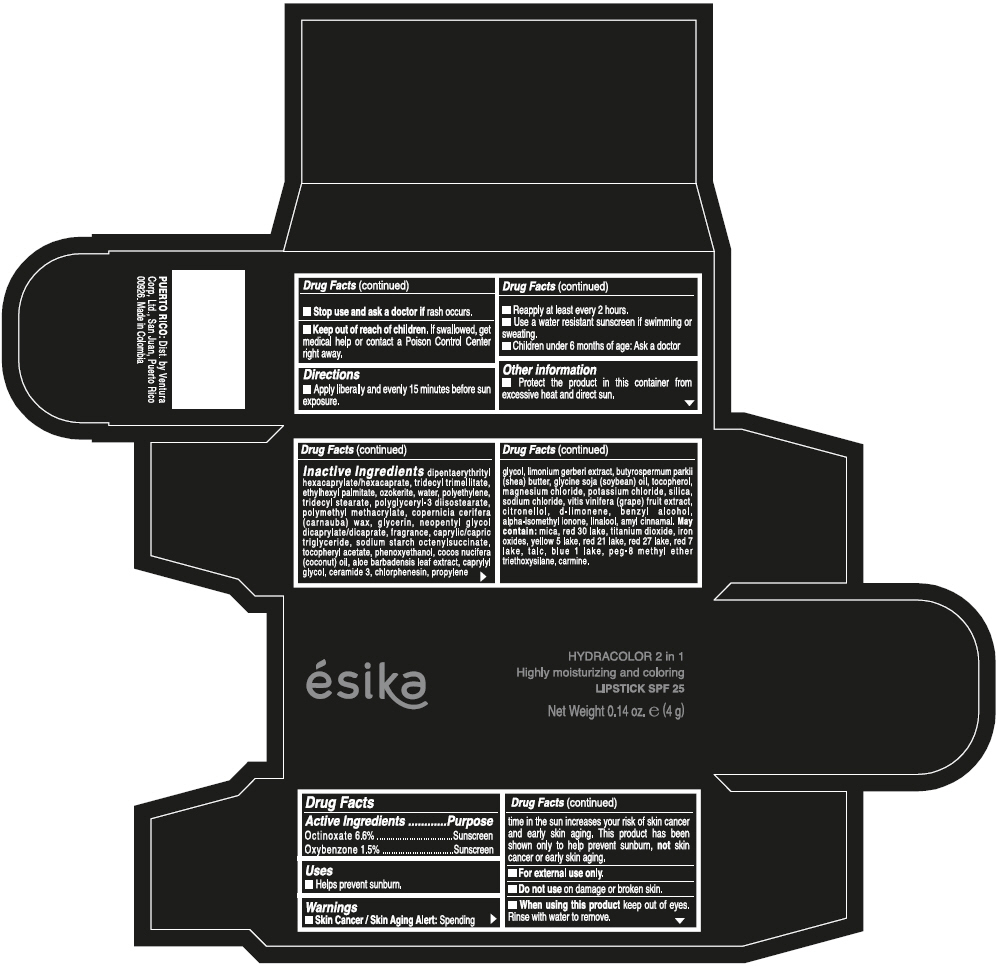
- ACTIVE INGREDIENT
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE, TRIDECYL TRIMELLITATE, ETHYLHEXYL PALMITATE, OZOKERITE, WATER, POLYETHYLENE, TRIDECYL STEARATE, POLYGLYCERYL-3 DIISOSTEARATE, POLYMETHYL METHACRYLATE, COPERNICIA CERIFERA (CARNAUBA) WAX, GLYCERIN, NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE, FRAGRANCE, CAPRYLIC/CAPRIC TRIGLYCERIDE, SODIUM STARCH OCTENYLSUCCINATE, TOCOPHERYL ACETATE, PHENOXYETHANOL, COCOS NUCIFERA (COCONUT) OIL, ALOE BARBADENSIS LEAF EXTRACT, CAPRYLYL GLYCOL, CERAMIDE 3, CHLORPHENESIN, PROPYLENE GLYCOL, LIMONIUM GERBERI EXTRACT, BUTYROSPERMUM PARKII (SHEA) BUTTER, GLYCINE SOJA (SOYBEAN) OIL, TOCOPHEROL, MAGNESIUM CHLORIDE, POTASSIUM CHLORIDE, SILICA, SODIUM CHLORIDE, VITIS VINIFERA (GRAPE) FRUIT EXTRACT, CITRONELLOL, d-LIMONENE, BENZYL ALCOHOL, ALPHA-ISOMETHYL IONONE, LINALOOL, AMYL CINNAMAL.
MAY CONTAIN:
MICA, RED 30 LAKE, TITANIUM DIOXIDE, IRON OXIDES, YELLOW 5 LAKE, RED 21 LAKE, RED 27 LAKE, RED 7 LAKE, TALC, BLUE 1 LAKE, PEG-8 METHYL ETHER TRIETHOXYSILANE, CARMINE. - SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (PIMIENTA CALIENTE) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (MARRON GLAMOUR) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (BORGONA SEXY) - PURPLE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSA DULZURA) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROJO DESEO) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROJO INTRIGANTE) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROJO LUJURIA) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (CORAL CHIC) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSA ENCANTO) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA CAUTIVANTE) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (GRANATE SEDUCTOR) - PURPLE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSA ROMANTICA) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (NARANJA PRIMAVERA) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FRAMBUESA MANIA) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FRESA COQUETA) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (CANELA NUDE) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (MORA GLAM) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (CORAL INTENSE) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSA GEOMETRIC) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA PRISMA) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (NARANJA ESFERA) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (NUDE INFINITO) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROJO ESIKA) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (MARRON TOPACIO) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (CORAL CUARZO) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA AMATISTA) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (NUDE PRISMA) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (DORADO ESENCIAL) - YELLOW
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSA ENCANTO FEMME) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (PIMIENTA CALIENTE FEMME) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (GRANATE SEDUCTOR FEMME) - PURPLE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSA ROMANTICA FEMME) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA CAUTIVANTE FEMME) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROJO INTRIGANTE FEMME) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (PIMIENTA CALIENTE EXOTIC) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSA ENCANTO EXOTIC) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (CORAL CHIC EXOTIC) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA CAUTIVANTE EXOTIC) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROJO DESEO EXOTIC) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FRAMBUESA MANIA EXOTIC) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (BORGONA SEXY EXOTIC) - PURPLE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (MARRON GLAMOUR EXOTIC) - BROWN
- PRINCIPAL DISPLAY PANEL - Kit Carton
-
INGREDIENTS AND APPEARANCE
ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (PIMIENTA CALIENTE) - BROWN
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-475 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-475-02 1 in 1 BOX 11/09/2016 1 NDC:13537-475-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON GLAMOUR) - BROWN
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-476 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-476-02 1 in 1 BOX 11/09/2016 1 NDC:13537-476-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (BORGONA SEXY) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-477 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-477-02 1 in 1 BOX 11/09/2016 1 NDC:13537-477-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA DULZURA) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-478 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-478-02 1 in 1 BOX 11/09/2016 1 NDC:13537-478-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO DESEO) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-479 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-479-02 1 in 1 BOX 11/09/2016 1 NDC:13537-479-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO INTRIGANTE) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-880 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-880-02 1 in 1 BOX 11/09/2016 1 NDC:13537-880-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO LUJURIA) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-804 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-804-02 1 in 1 BOX 11/09/2016 1 NDC:13537-804-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL CHIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-805 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-805-02 1 in 1 BOX 11/09/2016 1 NDC:13537-805-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ENCANTO) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-806 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-806-02 1 in 1 BOX 11/09/2016 1 NDC:13537-806-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA CAUTIVANTE) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-807 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-807-02 1 in 1 BOX 11/09/2016 1 NDC:13537-807-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (GRANATE SEDUCTOR) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-808 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-808-02 1 in 1 BOX 11/09/2016 1 NDC:13537-808-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ROMANTICA) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-809 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-809-02 1 in 1 BOX 11/09/2016 1 NDC:13537-809-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NARANJA PRIMAVERA) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-810 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-810-02 1 in 1 BOX 11/09/2016 1 NDC:13537-810-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FRAMBUESA MANIA) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-811 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-811-02 1 in 1 BOX 11/09/2016 1 NDC:13537-811-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FRESA COQUETA) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-812 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-812-02 1 in 1 BOX 11/09/2016 1 NDC:13537-812-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CANELA NUDE) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-813 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-813-02 1 in 1 BOX 11/09/2016 1 NDC:13537-813-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MORA GLAM) - BROWN
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-814 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-814-02 1 in 1 BOX 11/09/2016 1 NDC:13537-814-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL INTENSE) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-815 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-815-02 1 in 1 BOX 11/09/2016 1 NDC:13537-815-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA GEOMETRIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-816 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-816-02 1 in 1 BOX 11/09/2016 1 NDC:13537-816-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA PRISMA) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-817 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-817-02 1 in 1 BOX 11/09/2016 1 NDC:13537-817-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NARANJA ESFERA) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-818 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-818-02 1 in 1 BOX 11/09/2016 1 NDC:13537-818-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NUDE INFINITO) - BROWN
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-819 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-819-02 1 in 1 BOX 11/09/2016 1 NDC:13537-819-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO ESIKA) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-820 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-820-02 1 in 1 BOX 11/09/2016 1 NDC:13537-820-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON TOPACIO) - BROWN
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-821 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-821-02 1 in 1 BOX 11/09/2016 1 NDC:13537-821-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL CUARZO) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-822 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-822-02 1 in 1 BOX 11/09/2016 1 NDC:13537-822-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA AMATISTA) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-823 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-823-02 1 in 1 BOX 11/09/2016 1 NDC:13537-823-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NUDE PRISMA) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-824 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-824-02 1 in 1 BOX 11/09/2016 1 NDC:13537-824-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (DORADO ESENCIAL) - YELLOW
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-825 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-825-02 1 in 1 BOX 11/09/2016 1 NDC:13537-825-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ENCANTO FEMME) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-826 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-826-02 1 in 1 BOX 11/09/2016 1 NDC:13537-826-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (PIMIENTA CALIENTE FEMME) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-827 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-827-02 1 in 1 BOX 11/09/2016 1 NDC:13537-827-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (GRANATE SEDUCTOR FEMME) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-828 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-828-02 1 in 1 BOX 11/09/2016 1 NDC:13537-828-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ROMANTICA FEMME) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-829 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-829-02 1 in 1 BOX 11/09/2016 1 NDC:13537-829-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA CAUTIVANTE FEMME) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-830 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-830-02 1 in 1 BOX 11/09/2016 1 NDC:13537-830-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO INTRIGANTE FEMME) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-831 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-831-02 1 in 1 BOX 11/09/2016 1 NDC:13537-831-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (PIMIENTA CALIENTE EXOTIC) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-832 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-832-02 1 in 1 BOX 11/09/2016 1 NDC:13537-832-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ENCANTO EXOTIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-833 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-833-02 1 in 1 BOX 11/09/2016 1 NDC:13537-833-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL CHIC EXOTIC) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-834 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-834-02 1 in 1 BOX 11/09/2016 1 NDC:13537-834-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA CAUTIVANTE EXOTIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-835 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-835-02 1 in 1 BOX 11/09/2016 1 NDC:13537-835-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO DESEO EXOTIC) - RED
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-836 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-836-02 1 in 1 BOX 11/09/2016 1 NDC:13537-836-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FRAMBUESA MANIA EXOTIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-837 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-837-02 1 in 1 BOX 11/09/2016 1 NDC:13537-837-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (BORGONA SEXY EXOTIC) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-838 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-838-02 1 in 1 BOX 11/09/2016 1 NDC:13537-838-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON GLAMOUR EXOTIC) - BROWN
octinoxate and oxybenzone lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-839 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-839-02 1 in 1 BOX 11/09/2016 1 NDC:13537-839-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 11/09/2016 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25
octinoxate and oxybenzone kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-840 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-840-02 2 in 1 BOX 11/09/2016 1 NDC:13537-840-01 1 in 1 TUBE Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 8 g Part 2 1 TUBE 8 g Part 3 1 TUBE 8 g Part 4 1 TUBE 8 g Part 5 1 TUBE 8 g Part 6 1 TUBE 8 g Part 7 1 TUBE 8 g Part 8 1 TUBE 8 g Part 9 1 TUBE 8 g Part 10 1 TUBE 8 g Part 11 1 TUBE 8 g Part 12 1 TUBE 8 g Part 13 1 TUBE 8 g Part 14 1 TUBE 8 g Part 15 1 TUBE 8 g Part 16 1 TUBE 8 g Part 17 1 TUBE 8 g Part 18 1 TUBE 8 g Part 19 1 TUBE 8 g Part 20 1 TUBE 8 g Part 21 1 TUBE 8 g Part 22 1 TUBE 8 g Part 23 1 TUBE 8 g Part 24 1 TUBE 8 g Part 25 1 TUBE 8 g Part 26 1 TUBE 8 g Part 27 1 TUBE 8 g Part 28 1 TUBE 8 g Part 29 1 TUBE 8 g Part 30 1 TUBE 8 g Part 31 1 TUBE 8 g Part 32 1 TUBE 8 g Part 33 1 TUBE 8 g Part 34 1 TUBE 8 g Part 35 1 TUBE 8 g Part 36 1 TUBE 8 g Part 37 1 TUBE 8 g Part 38 1 TUBE 8 g Part 39 1 TUBE 8 g Part 40 1 TUBE 8 g Part 41 1 TUBE 8 g Part 42 1 TUBE 8 g Part 1 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (PIMIENTA CALIENTE) - BROWN
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 2 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON GLAMOUR) - BROWN
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 3 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (BORGONA SEXY) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 4 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA DULZURA) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 5 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO DESEO) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 6 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO INTRIGANTE) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 7 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO LUJURIA) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 8 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL CHIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 9 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ENCANTO) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 10 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA CAUTIVANTE) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 11 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (GRANATE SEDUCTOR) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 12 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ROMANTICA) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 13 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NARANJA PRIMAVERA) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 14 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FRAMBUESA MANIA) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 15 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FRESA COQUETA) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 16 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CANELA NUDE) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 17 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MORA GLAM) - BROWN
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 18 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL INTENSE) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 19 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA GEOMETRIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 20 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA PRISMA) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 21 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NARANJA ESFERA) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 22 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NUDE INFINITO) - BROWN
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 23 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO ESIKA) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 24 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON TOPACIO) - BROWN
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 25 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL CUARZO) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 26 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA AMATISTA) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 27 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (NUDE PRISMA) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 28 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (DORADO ESENCIAL) - YELLOW
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 29 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ENCANTO FEMME) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 30 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (PIMIENTA CALIENTE FEMME) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 31 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (GRANATE SEDUCTOR FEMME) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 32 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ROMANTICA FEMME) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 33 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA CAUTIVANTE FEMME) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 34 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO INTRIGANTE FEMME) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 35 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (PIMIENTA CALIENTE EXOTIC) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 36 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROSA ENCANTO EXOTIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 37 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (CORAL CHIC EXOTIC) - ORANGE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 38 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FUCSIA CAUTIVANTE EXOTIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 39 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (ROJO DESEO EXOTIC) - RED
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 40 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (FRAMBUESA MANIA EXOTIC) - PINK
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 41 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (BORGONA SEXY EXOTIC) - PURPLE
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Part 42 of 42 ESIKA HYDRACOLOR 2 IN 1 HIGHLY MOISTURIZING AND COLORING SPF 25 (MARRON GLAMOUR EXOTIC) - BROWN
octinoxate and oxybenzone lipstickProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0664 g in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0151 g in 1 g Inactive Ingredients Ingredient Name Strength DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) ETHYLHEXYL PALMITATE (UNII: 2865993309) CERESIN (UNII: Q1LS2UJO3A) WATER (UNII: 059QF0KO0R) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL STEARATE (UNII: A8OE252M6L) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DICAPRYLATE/DICAPRATE (UNII: VLW429K27K) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PHENOXYETHANOL (UNII: HIE492ZZ3T) COCONUT OIL (UNII: Q9L0O73W7L) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CERAMIDE NP (UNII: 4370DF050B) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHEA BUTTER (UNII: K49155WL9Y) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) WINE GRAPE (UNII: 3GOV20705G) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) LIMONENE, (+)- (UNII: GFD7C86Q1W) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) LINALOOL, (+/-)- (UNII: D81QY6I88E) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) MICA (UNII: V8A1AW0880) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 21 (UNII: 08744Z6JNY) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 7 (UNII: ECW0LZ41X8) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/09/2016 Labeler - Ventura Corporation LTD (602751344) Establishment Name Address ID/FEI Business Operations Bel Star S.A. (Colombia) 880160197 MANUFACTURE(13537-475, 13537-476, 13537-477, 13537-478, 13537-479, 13537-880, 13537-804, 13537-805, 13537-806, 13537-807, 13537-808, 13537-809, 13537-810, 13537-811, 13537-812, 13537-813, 13537-814, 13537-815, 13537-816, 13537-817, 13537-818, 13537-819, 13537-820, 13537-821, 13537-822, 13537-823, 13537-824, 13537-825, 13537-826, 13537-827, 13537-828, 13537-829, 13537-830, 13537-831, 13537-832, 13537-833, 13537-834, 13537-835, 13537-836, 13537-837, 13537-838, 13537-839, 13537-840)