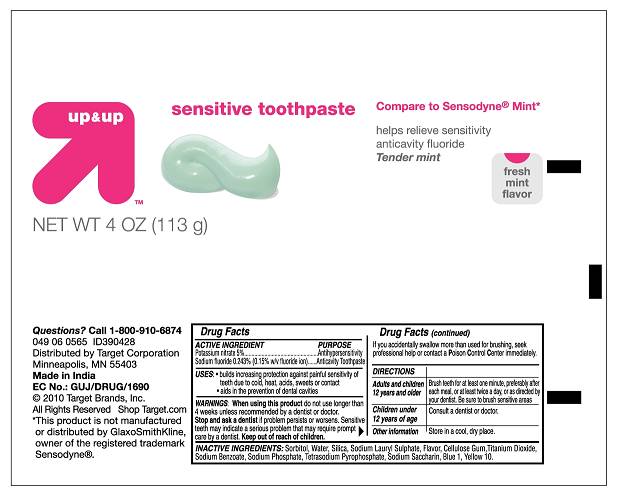
Label: UP AND UP SENSITIVE TOOTHPASTE- potassium nitrate and sodium fluoride paste
-
Contains inactivated NDC Code(s)
NDC Code(s): 67659-078-01, 67659-078-02 - Packager: Team Technologies, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 25, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- USES
-
WARNINGS
When using this product do not use longer than 4 weeks unless recommended by a dentist or doctor. Stop and ask a dentist if problems persists or worsens. Sensitive teeth may indicate a serious problem that may require prompt care by a dentist. Keep out of reach of children. If you accidentally swallow more that used for brushing seek professional help or contact a Poison Control Center immediately.
- Directions
- Inactive ingredients:
- Principal Display Panel
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
-
INGREDIENTS AND APPEARANCE
UP AND UP SENSITIVE TOOTHPASTE
potassium nitrate and sodium fluoride pasteProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67659-078 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POTASSIUM NITRATE (UNII: RU45X2JN0Z) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM NITRATE 5 mg in 1 g SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) SODIUM FLUORIDE 2.43 mg in 1 g Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) WATER (UNII: 059QF0KO0R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM PHOSPHATE (UNII: SE337SVY37) SODIUM PYROPHOSPHATE (UNII: O352864B8Z) SACCHARIN SODIUM (UNII: SB8ZUX40TY) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) Product Characteristics Color white Score Shape Size Flavor MINT (Fresh Mint Flavor) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67659-078-02 1 in 1 CARTON 1 NDC:67659-078-01 113 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 08/25/2010 Labeler - Team Technologies, Inc (192339703)