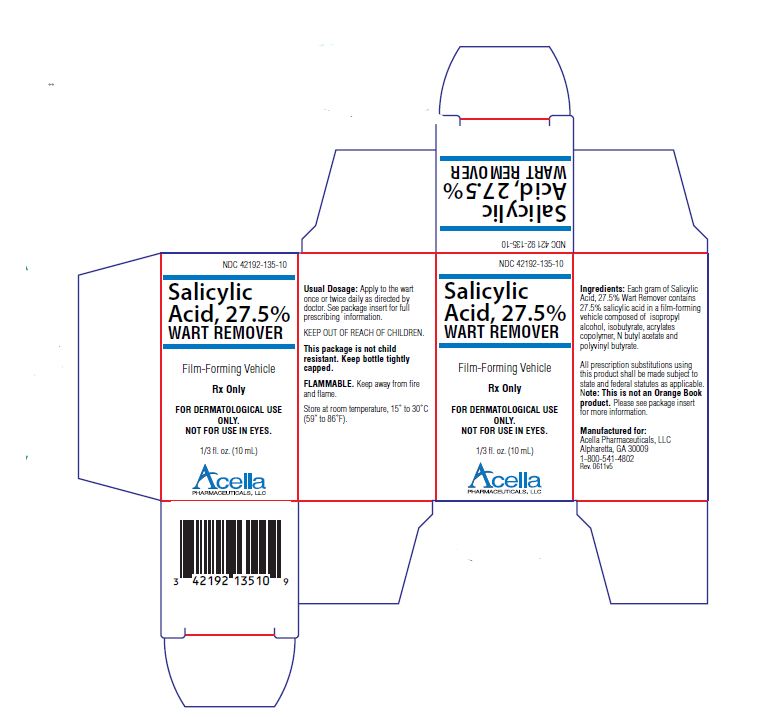
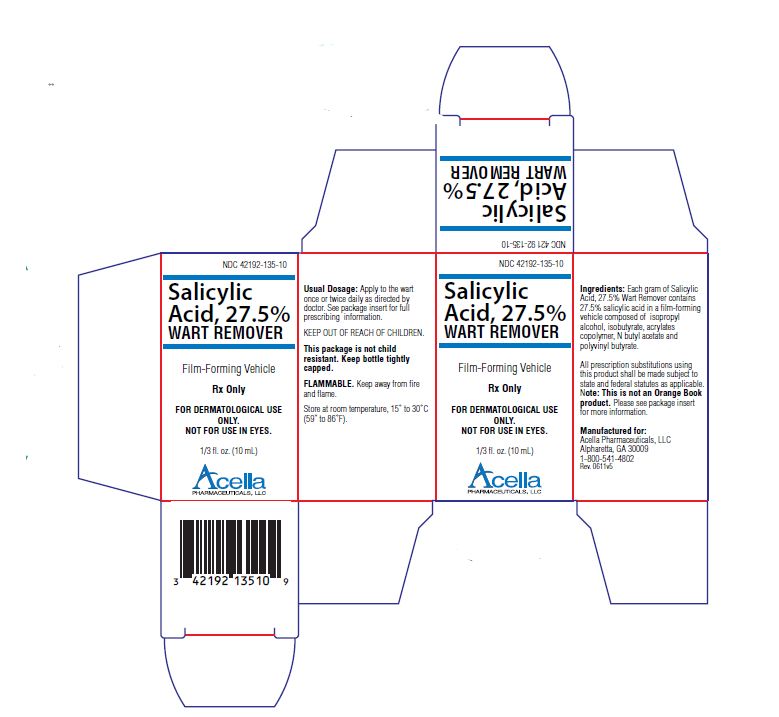
Label: SALICYLIC ACID liquid
- NDC Code(s): 42192-135-10
- Packager: Acella Pharmaceuticals, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 11, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION:
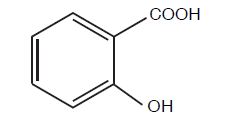
Salicylic Acid, 27.5% Wart Remover is a topical preparation containing 27.5% salicylic acid in a film-forming vehicle composed of isopropyl alcohol, isobutyrate, acrylates copolymer, N butyl acetate and polyvinyl butyrate. The pharmacologic activity of Salicylic Acid, 27.5% Wart Remover is generally attributed to the keratolytic activity of salicylic acid which is incorporated into a film-forming vehicle designed to cover the wart without the need for a bandage. The structural formula of salicylic acid is:
- CLINICAL PHARMACOLOGY:
- INDICATIONS AND USES:
- CONTRAINDICATIONS:
-
PRECAUTIONS:
Salicylic Acid, 27.5% Wart Remover is for external use only. Do not permit Salicylic Acid, 27.5% Wart Remover to contact eyes or mucous membranes. If contact with eyes or mucous membranes occurs, immediately flush with water for 15 minutes. Salicylic Acid, 27.5% Wart Remover should not be allowed to contact normal skin surrounding wart. Treatment should be discontinued if excessive irritation occurs. Salicylic Acid, 27.5% Wart Remover is flammable and should be kept away from fire or flame. Keep bottle tightly capped when not in use.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
- ADVERSE REACTIONS:
-
DOSAGE AND ADMINISTRATION:
Prior to application of Salicylic Acid, 27.5% Wart Remover, soak wart in warm water for five minutes. Remove any loosened tissue by gently rubbing with a brush, wash cloth or emery board. Dry thoroughly. Using the brush applicator supplied, apply twice to entire wart surface, allowing the first application to dry before applying the second. Treatment should be once or twice a day and should continue as directed by physician. Be careful not to apply to surrounding skin.
Clinically visible improvement will normally occur during the first or second week of therapy. Maximum resolution may be expected after four to six weeks of use.
- HOW SUPPLIED:
- STORAGE AND HANDLING
-
PATIENT INSTRUCTIONS:
Your health care provider has prescribed Salicylic Acid, 27.5% Wart Remover, a topical prescription preparation for the treatment of common and plantar warts. In order for Salicylic Acid, 27.5% Wart Remover to work correctly and to ensure maximum benefit, the following instructions should be followed carefully. Of course, as with any medication, always consult your health care provider if you experience any discomfort or unexpected reactions.
- STEP 1: WASH
-
STEP 2: BRUSH
- Using the brush applicator supplied, apply twice to entire wart surface, allowing the first application to dry before applying the second.
- Be careful not to apply to surrounding skin.
- Leave Salicylic Acid, 27.5% Wart Remover on wart area after it dries.
Repeat Step 1 and Step 2 once or twice daily as advised by your health care provider.
Clinically visible improvement will normally occur during the first or second week of therapy.
MANUFACTURED FOR:
Acella Pharmaceuticals, LLC
Alpharetta, GA 30009
1-800-541-4802Rev. 0611
- PRINCIPAL DISPLAY PANEL - 10 mL
-
INGREDIENTS AND APPEARANCE
SALICYLIC ACID
salicylic acid liquidProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:42192-135 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 275 mg in 1 mL Inactive Ingredients Ingredient Name Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) BUTYL ACETATE (UNII: 464P5N1905) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42192-135-10 1 in 1 CARTON 10/10/2011 1 10 mL in 1 BOTTLE, WITH APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/10/2011 Labeler - Acella Pharmaceuticals, LLC (825380939) Registrant - Acella Pharmaceuticals, LLC (825380939) Establishment Name Address ID/FEI Business Operations Acella Pharmaceuticals, LLC 825380939 manufacture(42192-135)