Label: POLYMAST- hetacillin potassium gel
- NDC Code(s): 0010-4722-01, 0010-4722-02, 0010-4722-03
- Packager: Boehringer Ingelheim Animal Health USA Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated October 11, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Caution:
-
Description:
POLYMAST (hetacillin potassium) is a broad-spectrum agent which provides bactericidal activity against a wide range of common gram-positive and gram-negative bacteria. It is derived from 6-aminopenicillanic acid and is chemically related to ampicillin.
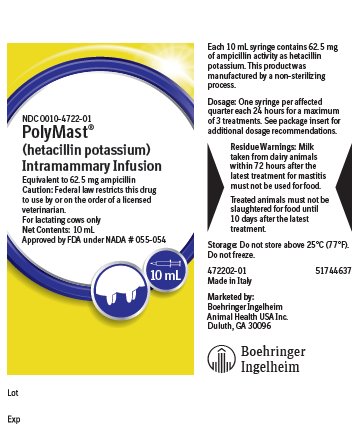
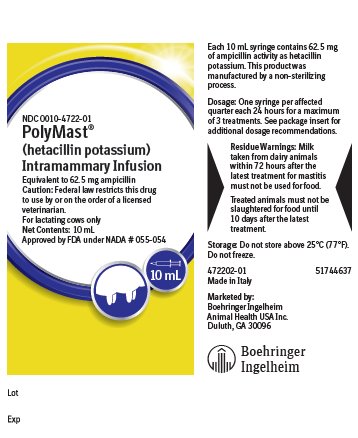
Each 10 mL disposable syringe contains hetacillin potassium equivalent to 62.5 mg ampicillin activity in a stable peanut oil gel. This product was manufactured by a non-sterilizing process.
- Action:
-
Indications:
For the treatment of acute, chronic or subclinical bovine mastitis. POLYMAST should be used at the first signs of inflammation or at the first indication of any alteration in the milk. Subclinical infections should be treated immediately upon determining, by C.M.T. or other tests, that the leukocyte count is elevated, or that a susceptible pathogen has been cultured from the milk.
POLYMAST has been shown to be efficacious in the treatment of mastitis in lactating cows caused by susceptible strains of Streptococcus agalactiae,Streptococcus dysgalactiae,Staphylococcus aureus and Escherichia coli.
Polycillin® (ampicillin) Susceptibility Test Discs, 10 mcg, should be used to estimate the in vitro susceptibility of bacteria to hetacillin.
-
Dosage and Administration:
Infuse the entire contents of one syringe (10 mL) into each infected quarter. Repeat at 24-hour intervals until a maximum of three treatments has been given.
If definite improvement is not noted within 48 hours after treatment, the causal organism should be further investigated.
Wash the udder and teats thoroughly with warm water containing a suitable dairy antiseptic and dry, preferably using individual paper towels. Carefully scrub the teat end and orifice with 70% alcohol, using a separate swab for each teat. Allow to dry.
POLYMAST is packaged with the Opti-Sert® Protective Cap.
For partial insertion: Twist off upper portion of the Opti-Sert Protective Cap to expose 3–4 mm of the syringe tip.
For full insertion: Remove protective cap to expose the full length of the syringe tip.
Insert syringe tip into the teat canal and expel the entire contents of one syringe into each infected quarter. Withdraw the syringe and gently massage the quarter to distribute the medication.
Do not infuse contents of the mastitis syringe into the teat canal if the Opti-Sert Protective Cap is broken or damaged.
- Residue Warnings:
-
Precautions
Because it is a derivative of 6-aminopenicillanic acid, POLYMAST has the potential for producing allergic reactions. Such reactions are rare; however, should they occur, treatment should be discontinued and the subject treated with antihistamines, pressor amines, such as epinephrine or corticosteroids.
The drug does not resist destruction by penicillinase and, hence, is not effective against strains of staphylococcus resistant to penicillin G.
- Storage:
- How Supplied:
-
SPL UNCLASSIFIED SECTION
Opti-Sert is a registered trademark of Zoetis W LLC - used under license.
POLYMAST is a trademark of Boehringer Ingelheim Vetmedica GmbH, used under license.
© 2020 Boehringer Ingelheim Animal Health USA Inc. All rights reserved.
Made in Italy
472201-01 51744638
Marketed by:
Boehringer Ingelheim Animal Health USA Inc.
Duluth, GA 30096
- Principal Display Panel – 10 mL syringe label
- Principal Display Panel – 12 x 10 mL syringe display carton
-
INGREDIENTS AND APPEARANCE
POLYMAST
hetacillin potassium gelProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0010-4722 Route of Administration INTRAMAMMARY Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HETACILLIN POTASSIUM (UNII: 95PFX5932Y) (AMPICILLIN - UNII:7C782967RD) AMPICILLIN 62.5 mg in 10 mL Inactive Ingredients Ingredient Name Strength PEANUT OIL (UNII: 5TL50QU0W4) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0010-4722-02 12 in 1 CARTON 1 NDC:0010-4722-01 10 mL in 1 SYRINGE, PLASTIC 2 NDC:0010-4722-03 144 in 1 PAIL 2 NDC:0010-4722-01 10 mL in 1 SYRINGE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA055054 09/28/2016 Labeler - Boehringer Ingelheim Animal Health USA Inc. (007134091)