Label: TRIPLE ANTIBIOTIC PLUS PAIN RELIEF MAXIMUM STRENGTH- bacitracin zinc, neomycin sulfate, polymyxin b sulfate and pramoxine hydrochloride ointment
- NDC Code(s): 68016-715-01
- Packager: Chain Drug Consortium, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 6, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients (in each gram)
- Purposes
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredient
- Questions or comments?
-
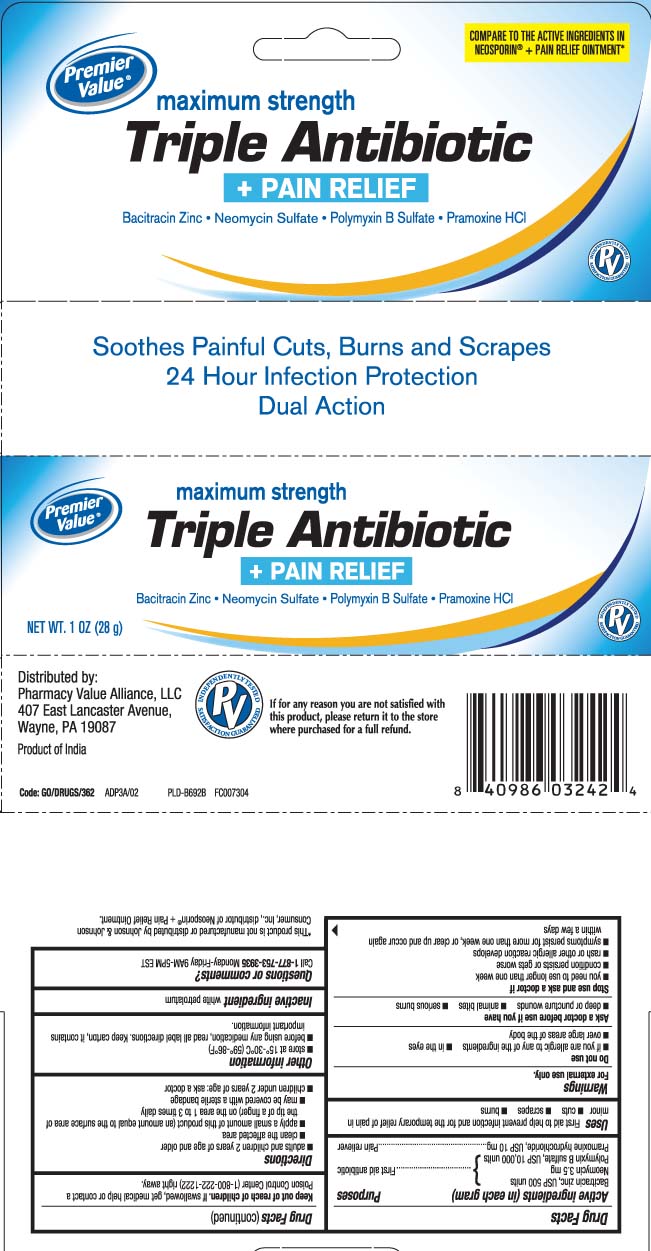
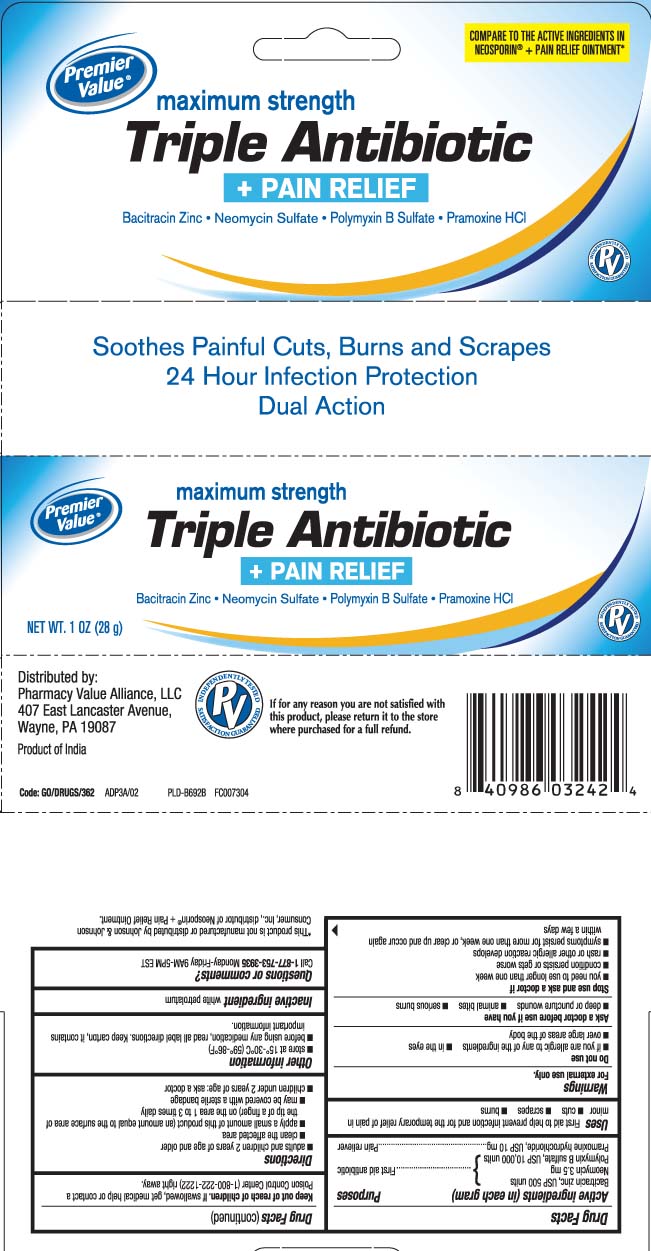
Principal display panel
COMPARE TO THE ACTIVE INGREDIENTS IN NEOSPORIN® + PAIN RELIEF OINTMENT*
maximum strength
Triple Antibiotic
+ PAIN RELIEF
Bacitracin Zinc • Neomycin Sulfate • Polymyxin B Sulfate • Pramoxine HCl
Soothe Painful Cuts, Burns and Scrapes
24 Hour Infection Protection
Dual Action
*This product is not manufactured or distributed by Johnson & Johnson Consumer, Inc., Distributor of Neosporin® + Pain Relief Ointment.
Distributed by:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087
- Package label
-
INGREDIENTS AND APPEARANCE
TRIPLE ANTIBIOTIC PLUS PAIN RELIEF MAXIMUM STRENGTH
bacitracin zinc, neomycin sulfate, polymyxin b sulfate and pramoxine hydrochloride ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-715 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 500 [USP'U] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 10000 [USP'U] in 1 g PRAMOXINE HYDROCHLORIDE (UNII: 88AYB867L5) (PRAMOXINE - UNII:068X84E056) PRAMOXINE HYDROCHLORIDE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-715-01 1 in 1 CARTON 07/10/2015 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 07/10/2015 Labeler - Chain Drug Consortium, LLC (101668460)