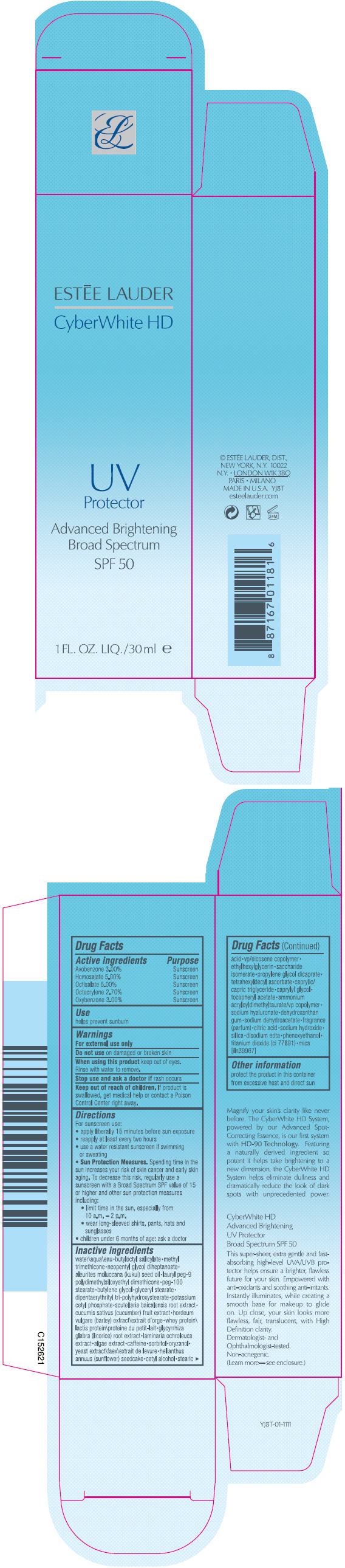
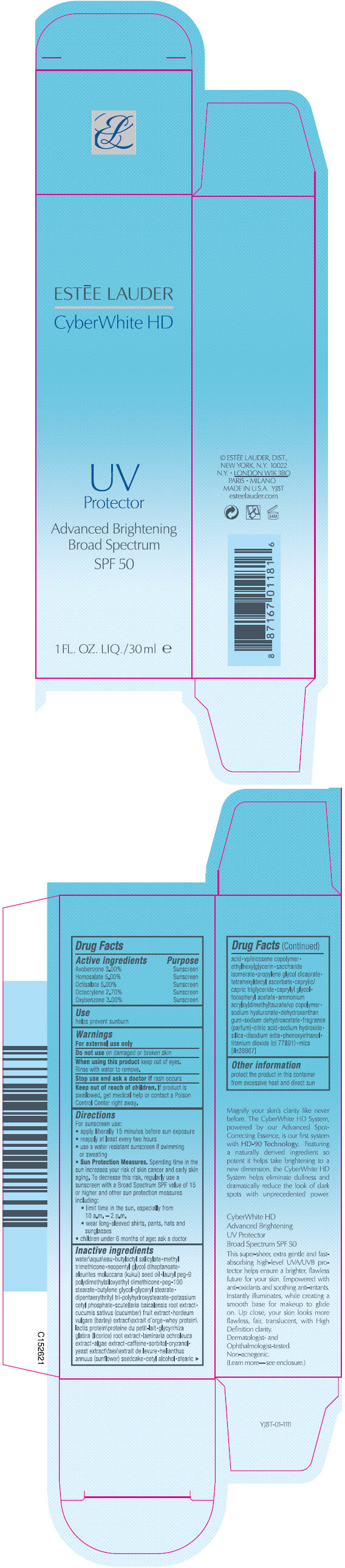
Label: CYBERWHITE HD UV PROTECTOR BROAD SPECTRUM SPF 50- avobenzone, homosalate, octisalate, octocrylene, and oxybenzone lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 11559-037-01 - Packager: ESTEE LAUDER INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 29, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Use
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
-
Inactive ingredients
water\aqua\eau • butyloctyl salicylate • methyl trimethicone • neopentyl glycol diheptanoate • aleurites moluccana (kukui) seed oil • lauryl peg-9 polydimethylsiloxyethyl dimethicone • peg-100 stearate • butylene glycol • glyceryl stearate • dipentaerythrityl tri-polyhydroxystearate • potassium cetyl phosphate • scutellaria baicalensis root extract • cucumis sativus (cucumber) fruit extract • hordeum vulgare (barley) extract\extrait d'orge • whey protein\lactis protein\proteine du petit-lait • glycyrrhiza glabra (licorice) root extract • laminaria ochroleuca extract • algae extract • caffeine • sorbitol • oryzanol • yeast extract\faex\extrait de levure • helianthus annuus (sunflower) seedcake • cetyl alcohol • stearic acid • vp/eicosene copolymer • ethylhexylglycerin • saccharide isomerate • propylene glycol dicaprate • tetrahexyldecyl ascorbate • caprylic/capric triglyceride • caprylyl glycol • tocopheryl acetate • ammonium acryloyldimethyltaurate/vp copolymer • sodium hyaluronate • dehydroxanthan gum • sodium dehydroacetate • fragrance (parfum) • citric acid • sodium hydroxide • silica • disodium edta • phenoxyethanol • titanium dioxide (ci 77891) • mica [iln39967]
- Other information
- PRINCIPAL DISPLAY PANEL - 30ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
CYBERWHITE HD UV PROTECTOR BROAD SPECTRUM SPF 50
avobenzone, homosalate, octisalate, octocrylene, and oxybenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11559-037 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Avobenzone (UNII: G63QQF2NOX) (Avobenzone - UNII:G63QQF2NOX) Avobenzone 0.0309 g in 100 mL Homosalate (UNII: V06SV4M95S) (Homosalate - UNII:V06SV4M95S) Homosalate 0.0515 g in 100 mL Octisalate (UNII: 4X49Y0596W) (Octisalate - UNII:4X49Y0596W) Octisalate 0.0515 g in 100 mL Octocrylene (UNII: 5A68WGF6WM) (Octocrylene - UNII:5A68WGF6WM) Octocrylene 0.0278 g in 100 mL Oxybenzone (UNII: 95OOS7VE0Y) (Oxybenzone - UNII:95OOS7VE0Y) Oxybenzone 0.0309 g in 100 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) butyloctyl salicylate (UNII: 2EH13UN8D3) methyl trimethicone (UNII: S73ZQI0GXM) neopentyl glycol diheptanoate (UNII: 5LKW3C543X) kukui nut oil (UNII: TP11QR7B8R) lauryl peg-9 polydimethylsiloxyethyl dimethicone (UNII: 25G622K2RA) peg-100 stearate (UNII: YD01N1999R) butylene glycol (UNII: 3XUS85K0RA) glyceryl monostearate (UNII: 230OU9XXE4) potassium cetyl phosphate (UNII: 03KCY6P7UT) scutellaria baicalensis root (UNII: 7J95K7ID2S) cucumber (UNII: YY7C30VXJT) whey (UNII: 8617Z5FMF6) glycyrrhiza glabra (UNII: 2788Z9758H) laminaria ochroleuca (UNII: 4R2124HE76) caffeine (UNII: 3G6A5W338E) sorbitol (UNII: 506T60A25R) gamma oryzanol (UNII: SST9XCL51M) yeast (UNII: 3NY3SM6B8U) helianthus annuus seedcake (UNII: 482WYF7XLC) cetyl alcohol (UNII: 936JST6JCN) stearic acid (UNII: 4ELV7Z65AP) ethylhexylglycerin (UNII: 147D247K3P) saccharide isomerate (UNII: W8K377W98I) propylene glycol dicaprate (UNII: U783H9JHWY) tetrahexyldecyl ascorbate (UNII: 9LBV3F07AZ) medium-chain triglycerides (UNII: C9H2L21V7U) caprylyl glycol (UNII: 00YIU5438U) .alpha.-tocopherol acetate (UNII: 9E8X80D2L0) ammonium acryloyldimethyltaurate/vp copolymer (UNII: W59H9296ZG) hyaluronate sodium (UNII: YSE9PPT4TH) sodium dehydroacetate (UNII: 8W46YN971G) citric acid monohydrate (UNII: 2968PHW8QP) sodium hydroxide (UNII: 55X04QC32I) silicon dioxide (UNII: ETJ7Z6XBU4) edetate disodium (UNII: 7FLD91C86K) phenoxyethanol (UNII: HIE492ZZ3T) titanium dioxide (UNII: 15FIX9V2JP) mica (UNII: V8A1AW0880) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11559-037-01 1 in 1 CARTON 1 30 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 10/01/2013 Labeler - ESTEE LAUDER INC (005914387) Establishment Name Address ID/FEI Business Operations ELGC K.K. 712808195 RELABEL(11559-037) , REPACK(11559-037) Establishment Name Address ID/FEI Business Operations Estee Lauder Pennsylvania Distribution Center 2 (PADC 2) 828534516 MANUFACTURE(11559-037) , RELABEL(11559-037) , REPACK(11559-037) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD 244669714 MANUFACTURE(11559-037) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD. 205952385 MANUFACTURE(11559-037) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER N.V. 370151326 MANUFACTURE(11559-037) Establishment Name Address ID/FEI Business Operations LEN-RON MANUFACTURING DIVISION OF ARAMIS INC 809771152 MANUFACTURE(11559-037) Establishment Name Address ID/FEI Business Operations NORTEC KEYSTONE 943871157 MANUFACTURE(11559-037) , RELABEL(11559-037) , REPACK(11559-037) Establishment Name Address ID/FEI Business Operations NORTHTEC BRISTOL 959338336 MANUFACTURE(11559-037) , RELABEL(11559-037) , REPACK(11559-037) Establishment Name Address ID/FEI Business Operations NORTHTEC KEYSTONE 949264774 MANUFACTURE(11559-037) , RELABEL(11559-037) , REPACK(11559-037) Establishment Name Address ID/FEI Business Operations WHITMAN LABORATORIES, LTD. 216866277 MANUFACTURE(11559-037) Establishment Name Address ID/FEI Business Operations Aveda Corporation 071352058 MANUFACTURE(11559-037) Establishment Name Address ID/FEI Business Operations Pennsylvania Logistics Center 078364654 REPACK(11559-037) , RELABEL(11559-037)